The Swine Flu Fiasco
 In January 1976, a group of young healthy servicemen fell ill with an unknown respiratory illness at Fort Dix, a US Army training center in New Jersey. Several were hospitalized. One recruit, refusing hospitalization, died. The cause proved to be H1N1, the influenza strain responsible for the 1918 pandemic. It was considered the most dangerous form of influenza, but since 1918 it was mostly limited to those working with pigs. For the first time in 58 years, H1N1 was clearly spreading quickly through human to human contact. Out of 500 young men, 13 became sick over a few weeks, and 1 died.
In January 1976, a group of young healthy servicemen fell ill with an unknown respiratory illness at Fort Dix, a US Army training center in New Jersey. Several were hospitalized. One recruit, refusing hospitalization, died. The cause proved to be H1N1, the influenza strain responsible for the 1918 pandemic. It was considered the most dangerous form of influenza, but since 1918 it was mostly limited to those working with pigs. For the first time in 58 years, H1N1 was clearly spreading quickly through human to human contact. Out of 500 young men, 13 became sick over a few weeks, and 1 died.
It appeared to scientists at the Center for Disease Control (CDC) that an influenza apocalypse was upon us. Armed with technology (vaccination) not available in 1918, or the smaller influenza epidemics of 1957 and 1968, the CDC pressed for mass vaccination. On March 24, 1976, President Gerald Ford, flanked by Drs. Sabin and Salk of polio vaccine fame, announced:
“I have been advised that there is a very real possibility that unless we take effective counteractions, there could be an epidemic of this dangerous disease next fall and winter here in the United States. Let me state clearly at this time: no one knows exactly how serious this threat could be. Nevertheless, we cannot afford to take a chance with the health of our nation. Accordingly, I am today announcing the following actions. I am asking the Congress to appropriate $135 million, prior to their April recess, for the production of sufficient vaccine to inoculate every man, woman, and child in the United States.”1
Field trials for a vaccine began in April. Mass immunization began on October 1st, 1976. Eleven days later, three elderly patients in Pittsburgh died almost immediately after vaccination. The Allegheny County Health Department suspended the vaccination program. In Minnesota, health authorities noticed several cases of Guillain-Barré. This was followed by a rising incidence of unexplained deaths and Guillain-Barré (a complication of both influenza itself and other influenza vaccines).2 When no cases of H1N1 appeared in the winter, the perceived risk/benefit ratio shifted to all risk, and the vaccination program was stopped in December.
New programs were set up following this “Swine Flu Fiasco” of 1976. A considerable irony is those vaccinated in 1976 may have been protected as older adults during the 2009 H1N1 pandemic.3 The 1976 Swine Flu vaccination program also presaged many of the challenges of communicating vaccine benefits vs. vaccine safety that we see with vaccination programs today.
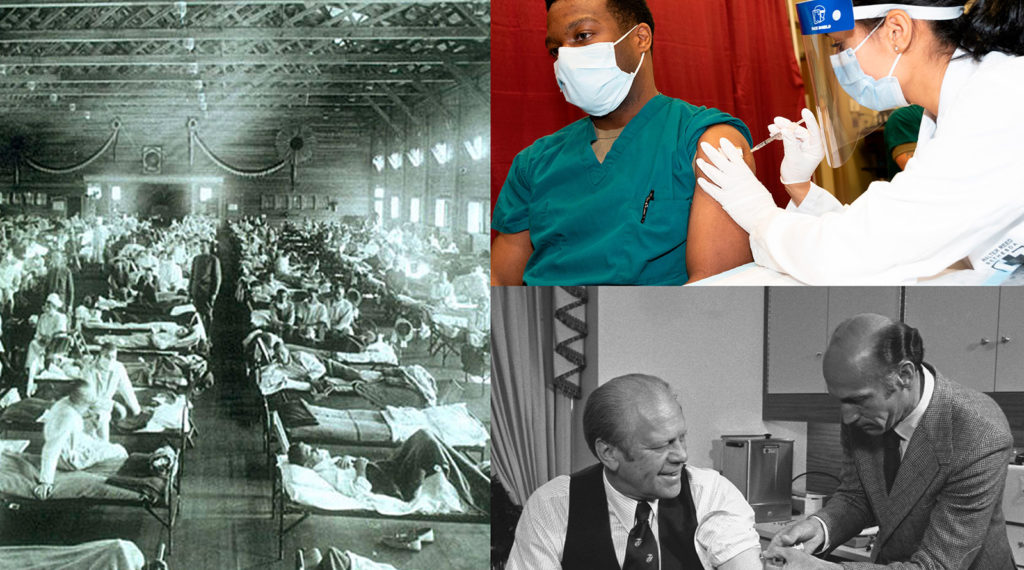
Left panel: 1918 influenza pandemic, National Museum of Health and Medicine, Armed Forces Institute of Pathology, Washington, DC, United States; Right upper panel: Army Cpt. Dr. Isaiah Horton receives COVID-19 vaccine, US Secretary of Defense; Right lower panel: US President Gerald Ford receiving swine flu vaccine, Gerald R. Ford Presidential Library;
Operation Warp Speed
Unlike the 1976 swine flu fiasco, when no subsequent cases of H1N1 were seen after the initial outbreak at Fort Dix, as of April 5, 2021, there have been over 130 million cases and 2.8 million deaths worldwide (figure 1) secondary to SARS-CoV-2. In the United States, 1 in 11 have been infected by SARS-CoV-2, and 1 in roughly 600 Americans have died from COVID-19 (more than 540,000 as of March, 2021).
Worldwide Projection as of April 5, 2021
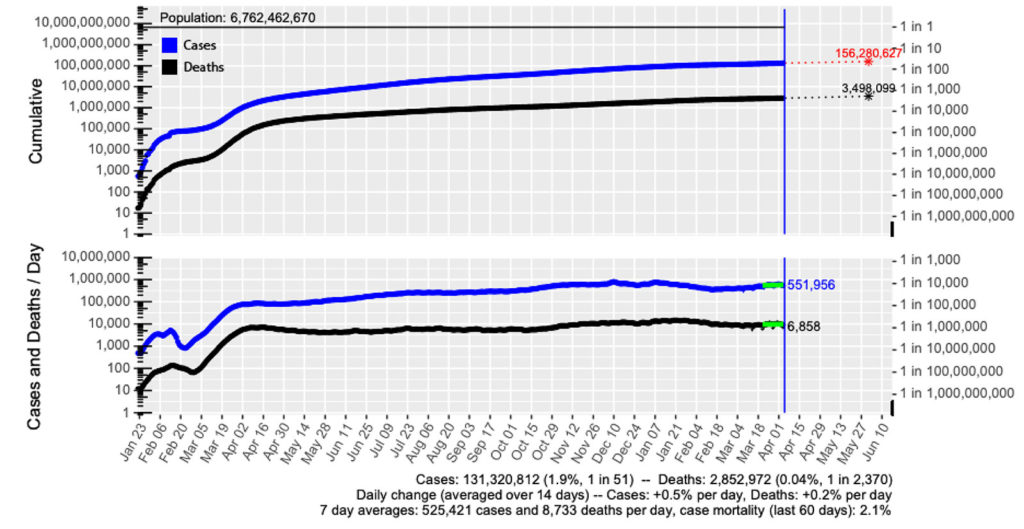
Figure 1: Worldwide projection as of April 5, 2021.32
Source: Daily COVID-19 update, used with permission from author.
The COVID epidemic has focused the world’s scientific firepower as never before. In the United States, “Operation Warp Speed” was a public/private partnership to provide nearly unlimited government support to companies pursuing vaccines and other therapies to address COVID-19. Similar programs were established in Europe, India, and China, with unprecedented success.
According to the World Health Organization, there are currently 82 vaccines in clinical development (table 1).4 Of these, 13 are presently approved in at least 1 country.5 All of the vaccines have demonstrated efficacy. The only serious safety concern that has emerged is the exceedingly small possibility of thrombosis with the AstraZeneca vaccine. I will repeat that for emphasis: the only serious safety concerns that has emerged is the very low possibility of thrombosis with the AstraZeneca vaccine.
Table 1: Vaccine Platforms in Development.33
| Platform | Candidate Vaccines |
| Protein subunit | 28 |
| Viral Vector (non-replicating) | 12 |
| DNA | 10 |
| Inactivated Virus | 11 |
| RNA | 11 |
| Viral Vector (replicating) | 4 |
| Virus Like Particle | 4 |
| Live Attenuated Virus | 2 |
| Replicating Viral Vector + Antigen Presenting Cell | 2 |
| Non-replicating Viral Vector + Antigen Presenting Cell | 1 |
Vaccines stimulate the immune system. Very obviously, that is the entire point! You know what this feels like: fatigue, headache, myalgias, lethargy, and generalized “flu-like” symptoms. These responses aren’t caused by the virus per se. This is simply what it feels like when your immune system gets activated. Since the vaccines activate the immune system, any of the vaccines can make you feel ill for a couple of hours to perhaps a day or two. Just as you should expect the shot to hurt a little, because they are sticking a needle in your arm, you should expect to feel somewhat viral, because the shot activates your immune system.
The safety question is: what other unwelcome effects might the vaccine have, other than making you feel like you have a virus?
Vaccine Safety and Efficacy
1. BNT162b2 is an mRNA vaccine developed by Pfizer and BioNTech. It is the first vaccine approved via an Emergency Use Authorization (EUA)* in the United States. In a study of 43,548 subjects, the vaccine demonstrated an outstanding 95% efficacy and nearly 100% efficacy against severe disease.6 This is similar to the efficacy of the MMRV vaccine.7 Only 1 patient who received the vaccine (out of > 20,000) developed severe COVID-19. Common adverse events were limited to injection site pain and flu-like symptoms. The safety and efficacy demonstrated in the Phase 3 study was subsequently reproduced when the vaccine was deployed on a large scale in Israel.8
Shortly after the BNT162b2 vaccination program was launched several cases of anaphylaxis were observed.9 The most recent assessment is that the risk of anaphylaxis is approximately 1 in 100,000.10 The mRNA vaccines incorporate a lipid nanoparticle to facilitate mRNA entry into the cell. It is currently thought that the lipid nanoparticle is responsible for the rare allergic reactions.11 The risk of anaphylaxis was mitigated through introduction of immediate postvaccination monitoring of individuals for up to 30 minutes, reducing the risk of injury from anaphylaxis to nearly 0. The CDC estimates that there have no deaths associated with the BNT162b2 vaccine.12
2. mRNA-1273 is an mRNA vaccine developed by Moderna. It is the second vaccine with EUA approval in the United States. In the phase 3 study of 30,420 individuals, the mRNA-1273 vaccine also demonstrated outstanding efficacy of 94%. To place this into perspective, the FDA set a bar of 50% efficacy for vaccine approval.13
The risks of the Moderna mRNA-1273 are identical to that of the Pfizer/BioNTech.14 This is expected, because both vaccines use the same lipid nanoparticles to facilitate entry into the cell.15 The risk of anaphylaxis is about 1 in 200,000. There have been no deaths or serious injuries. Otherwise, recipients of mRNA-1273 should expect to feel mildly ill while their immune system ramps up.
3. AD26.COV2.S is not an mRNA type vaccine but rather a non-replicating viral vector vaccine developed by Johnson & Johnson. It is the third vaccine with EUA approval in the United States. The phase 3 trial of 44,325 adults found an efficacy of 72% in the United States, 66% in Latin American countries, and 57% in South Africa.16 No vaccinated patients died of COVID-19. The safety data has not appeared in the peer-reviewed literature. However, the safety profile is well described in the FDA briefing document17: “Safety analysis through the January 22, 2021 data cutoff included 43,783 randomized (1:1) participants ≥18 years of age with 2-month median follow-up. The analysis supported a favorable safety profile with no specific safety concerns identified that would preclude issuance of an EUA.” There were no instances of anaphylaxis in the study, but one individual had a hypersensitive reaction two days after vaccination that was not classified as anaphylaxis.*
4. AZD1222, also known as ChAdOx1 nCoV-19, is an adenovirus vectored vaccine developed by a partnership between Oxford University and AstraZeneca. It is approved throughout Europe, Asia, and South America. AstraZeneca recently completed a phase 3 trial in the United States and has announced plans to seek EUA approval in the US.
AZD1222 would seem to have been cursed since the outset. In the pivotal phase 3 trial, there was a dosing error resulting in a subpopulation of patients having a lower dose than intended. Amazingly, these patients had a better immune response, but it is unclear exactly why that was the case! In an interim analysis, the vaccine was 62% effective in patients who received the higher dose as specified in the protocol, and 90% effective in patients who received the lower dose.18 In the final analysis, vaccine efficacy was 76% after a single standard dose.19 No patients in the vaccinated group required hospitalization after 21 days, and there were no COVID deaths in the vaccinated group.
The safety analysis identified two concerning adverse events: one case of transverse myelitis, and one instance of a fever following vaccination of 40°C without explanation. Both cases resolved. One subsequent case of transverse myelitis was reported, but subsequently was determined by the site investigator to be unrelated.
A paper from South Africa published in the New England Journal of Medicine after approval of AZD1222 showed that it didn’t work against the B.1.351 variant that has become the predominant strain in South Africa.20
In March 2021, three patients in Norway suffered thrombotic events after receiving the AZD1222 vaccine, and one patient died. Norway suspended use of the vaccine pending investigation. Several additional thrombotic events were reported in Europe, including 22 in the UK.21 A case has also been reported in Australia.22 What is unusual about these cases is that they are associated with low platelet counts, suggesting a mechanistic link to heparin-induced thrombocytopenia.23 In response, Denmark, Norway, Iceland, Bulgaria, Ireland, the Netherlands, Germany, Italy, France, Spain, Portugal, Sweden, Luxembourg, Cyprus, and Latvia all suspended use of the vaccine. Subsequently the European Medicines Agency, the World Health Organization,24 and AstraZeneca25 determined that the cases of thrombosis were not related to the vaccine, and recommended continued use.
On March 22, 2021, AstraZeneca announced the results of the 32,449-subject phase 3 US trial.26 The vaccine was 79% effective, and 100% effective at preventing severe disease. The data safety monitoring board reviewed thrombotic events, including cerebral venous sinus thrombosis, and found no evidence of increased risk. No cases of cerebral venous sinus thrombosis occurred in the trial. The following day, the Data and Safety Monitoring Board (DSMB) issued through the National Institute of Allergy and Infectious Disease, a statement disputing the AstraZeneca press announcement, stating that the DSMB expressed concern that AstraZeneca may have included outdated information from that trial, which may have provided an incomplete view of the efficacy data.”27
As mentioned, AZD1222 seems to have been cursed, starting with a dosing error in the clinical trial. Controversy continued with the findings of lower doses producing greater efficacy, concerns over very rare transverse myelitis cases, thrombosis, and now with concerns over the cherry picking of data. There is an excellent review of the odd twists and turns of ADZ1222 in Nature News.28 The Medicines and Healthcare products Regulatory Agency in the United Kingdom has published guidelines for diagnosing and treating thrombosis and cytopenia following vaccination.29
Would I get the AZD1222 vaccine? Absolutely! The vaccine has been given to more than 17 million people. There have been about 50 embolic events, a rate of about 3 per million. Case mortality for COVID-19 is currently running about 2.4%, and more than 20% of all patients who get COVID-19 have some level of persistent symptoms. Some simple math: 17 million × 75% herd immunity threshold × 2% case rate mortality × 90% efficacy in preventing death = ~230,000 lives saved already through vaccination with AZC1222.
5. Sputnik V is a combination of two adenovirus vectored vaccines, developed by Gamaleya Research Institute in Russia and currently approved in Russia and multiple countries in Asia, Africa, The Middle East, and South America. The phase 3 trial of 21,977 subjects found an efficacy of 92%.30 The vaccine was 100% effective against serious disease and death after 21 days. No serious adverse events have been reported related to vaccination.
6. Coronavac is an inactivated SARS-CoV-2 vaccine developed by Sinovac, and currently approved in China and multiple countries in Asia and South America. The data from large phase 3 trials is currently only available in press releases, but these suggest “the efficacy rate against diseases caused by COVID-19 was 51% for all cases, 84% for cases requiring medical treatment, and 100% for hospitalized, severe, and fatal cases.31
7. BBIBP-CorV is an inactivated SARS-CoV-2 vaccine developed by Sinopharm, and currently approved in China and multiple countries in Asia, South America, and the Middle East. There seems to be even less safety and efficacy data than for CoronaVac. A summary in Wikipedia suggested 86% efficacy in a study in Bahrain, with 100% efficacy in preventing severe disease. These data have not been published.
In summary, the currently approved vaccines appear to be highly effective at preventing infection and almost 100% effective in preventing severe disease and death. There are some exceptionally rare events, such as anaphylaxis with the mRNA vaccines, and possibly very rare cases of thrombosis from the AstraZeneca AZD1222 vaccine. Given this profile, and the profound health, social, and economic costs of an unmitigated pandemic, the ratio of benefit to risk is asymptotically 1:0.
Steven Shafer, MD, is professor of Anesthesiology, Perioperative and Pain Medicine at Stanford University.
The author has no conflicts of interest.
References
- Neustadt RE, Fineberg HV. The swine flu affair: decision-making on a slippery disease. Washington (DC): National Academies Press (US); 1978.
- Sivadon-Tardy V, Orlikowski D, Porcher R, et al. Guillain-Barré syndrome and influenza virus infection. Clin Infect Dis. 2009;48:48–56.
- Xie H, Li X, Gao J, Lin Z, et al. Revisiting the 1976 “swine flu” vaccine clinical trials: cross-reactive hemagglutinin and neuraminidase antibodies and their role in protection against the 2009 H1N1 pandemic virus in mice. Clin Infect Dis. 2011;53:1179–87.
- World Health Organization. Draft landscape and tracker of COVID-19 candidate vaccines. https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines. Accessed March 25, 2021.
- Regulatory Affairs Professionals Society. COVID-19 vaccine tracker. https://www.raps.org/news-and-articles/news-articles/2020/3/covid-19-vaccine-tracker. Accessed March 25, 2021.
- Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383:2603–2615.
- Henry O, Brzostek J, Czajka H, et al. One or two doses of live varicella virus-containing vaccines: efficacy, persistence of immune responses, and safety six years after administration in healthy children during their second year of life. Vaccine. 2018;36:381–387.
- Dagan N, Barda N, Kepten E, et al. BNT162b2 mRNA Covid-19 vaccine in a nationwide mass vaccination setting. N Engl J Med. 2021:NEJMoa2101765. doi: 10.1056/NEJMoa2101765. Epub ahead of print.
- CDC COVID-19 Response Team; Food and Drug Administration. Allergic reactions including anaphylaxis after receipt of the first dose of Pfizer-BioNTech COVID-19 vaccine – United States, December 14–23, 2020. MMWR Morb Mortal Wkly Rep. 2021;70:46–51. doi: 10.15585/mmwr.mm7002e1.
- Turner PJ, Ansotegui IJ, Campbell DE, et al. COVID-19 vaccine-associated anaphylaxis: a statement of the World Allergy Organization Anaphylaxis Committee. World Allergy Organ J. 2021;14:100517.
- Moghimi SM. Allergic reactions and anaphylaxis to LNP-based COVID-19 vaccines. Mol Ther. 2021;29:898–900.
- Gee J, Marquez P, Su J, Calvert GM, et al. First month of COVID-19 vaccine safety monitoring – United States, December 14, 2020–January 13, 2021. MMWR Morb Mortal Wkly Rep. 2021;70:283-288.
- Development and Licensure of Vaccines to Prevent COVID-19 – Department of Health and Human Services, FDA Center for Biologics Evaluation and Research. June 2020
- CDC COVID-19 Response Team; Food and Drug Administration. Allergic reactions including anaphylaxis after receipt of the first dose of Moderna COVID-19 vaccine – United States, December 21, 2020–January 10, 2021. MMWR Morb Mortal Wkly Rep. 2021;70:125–129.
- Cross R. Without these lipid shells, there would be no mRNA vaccines for COVID-19. Chemical & Engineering News. March 2021 (https://cen.acs.org/pharmaceuticals/drug-delivery/Without-lipid-shells-mRNA-vaccines/99/i8). Accessed March 25, 2021.
- Janssen Investigational COVID-19 Vaccine: interim analysis of Phase 3 clinical data released. National Institutes of Health News Release, January 29, 2021 (https://www.nih.gov/news-events/news-releases/janssen-investigational-covid-19-vaccine-interim-analysis-phase-3-clinical-data-released). Accessed March 25, 2021
- Janssen Ad26.COV2.S vaccine for the prevention of COVID-19. FDA Briefing Document. https://www.fda.gov/media/146217/download. Accessed March 25, 2021.
- Voysey M, Clemens SAC, Madhi SA, et al. Oxford COVID Vaccine Trial Group. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021 Jan 9;397(10269):99–111. doi: 10.1016/S0140-6736(20)32661-1. Epub 2020 Dec 8. Erratum in: Lancet. 2021 Jan 9;397(10269):98. PMID: 33306989; PMCID: PMC7723445.
- Voysey M, Clemens SAC, Madhi SA, et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials. Lancet. 2021;397:881–891.
- Madhi SA, Baillie V, Cutland CL, et al. Efficacy of the ChAdOx1 nCoV-19 Covid-19 vaccine against the B.1.351 variant. N Engl J Med. 2021. doi: 10.1056/NEJMoa2102214. Epub ahead of print.
- Medicines & Healthcare products Regulatory Agency. Research and analysis: Coronavirus vaccine – weekly summary of Yellow Card reporting. https://www.gov.uk/government/publications/coronavirus-covid-19-vaccine-adverse-reactions/coronavirus-vaccine-summary-of-yellow-card-reporting. Accessed April 5, 2021.
- Reuters. Australia to continue AstraZeneca vaccination despite blood clotting case. https://www.reuters.com/article/us-health-coronavirus-australia/australia-to-continue-astrazeneca-vaccination-despite-blood-clotting-case-idUSKBN2BQ06B. Accessed April 5, 2021.
- Greinacher A, Thiele T, Warkentin TE, et al. A prothrombotic thrombocytopenic disorder resembling heparin-induced thrombocytopenia following Coronavirus-19 vaccination. Research Square Preprint Server. DOI: 10.21203/rs.3.rs-362354/v1.
- World Health Organization. Statement of the WHO Global Advisory Committee on Vaccine Safety (GACVS) COVID-19 subcommittee on safety signals related to the AstraZeneca COVID-19 vaccine. https://www.who.int/news/item/19-03-2021-statement-of-the-who-global-advisory-committee-on-vaccine-safety-(gacvs)-covid-19-subcommittee-on-safety-signals-related-to-the-astrazeneca-covid-19-vaccine.Accessed March 25, 2021.
- AstraZeneca. Update on the safety of COVID-19 Vaccine AstraZeneca. March 14, 2021. https://www.astrazeneca.com/media-centre/press-releases/2021/update-on-the-safety-of-covid-19-vaccine-astrazeneca.html. Accessed March 25, 2021.
- AstraZeneca. AZD1222 US Phase III trial met primary efficacy endpoint in preventing COVID-19 at interim analysis. March 22, 2021. https://www.astrazeneca.com/media-centre/press-releases/2021/astrazeneca-us-vaccine-trial-met-primary-endpoint.html. Accessed March 25, 2021.
- National Institues of Health. NIAID statement on AstraZeneca vaccine. https://www.nih.gov/news-events/news-releases/niaid-statement-astrazeneca-vaccine. Accessed March 25, 2021.
- Mallapaty S, Callaway E. What scientists do and don’t know about the Oxford-AstraZeneca COVID vaccine. March 24, 2021. https://www.nature.com/articles/d41586-021-00785-7. Accessed March 25, 2021.
- Guidance produced from the Expert Haematology Panel (EHP) focussed on syndrome of thrombosis and thrombocytopenia occurring after coronavirus vaccination. https://b-s-h.org.uk/media/19498/guidance-version-07-on-mngmt-of-thrombosis-with-thrombocytopenia-occurring-after-c-19-vaccine_20210330_.pdf. Accessed April 6, 2021.
- Logunov DY, Dolzhikova IV, Shcheblyakov DV, et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet. 2021;397:671–681.
- Mallapaty, S. China COVID vaccine reports mixed results — what does that mean for the pandemic? Nature. January 15, 2021. https://www.nature.com/articles/d41586-021-00094-z. Accessed March 25, 2021.
- Source: Steve’s Daily COVID-19 update, distributed by the author daily except Sunday. The update is freely available. To be added to the distribution list just send me an e-mail ([email protected]). Case and death data are from the repository maintained by Johns Hopkins.
- World Health Organization. Draft landscape and tracker of COVID-19 candidate vaccines. April 6, 2021. https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines. Accessed April 6, 2021.
*Since this article entered production, the Johnson & Johnson vaccine appears to be associated with the syndrome of thrombosis and low platelets (see https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/JJUpdate.html). The incidence appears to be approximately 1 case per million vaccine doses. The CDC had recommended pausing administration while the association was studied and risk factors were identified. On April 23, 2021, the CDC and FDA lifted the pause for administration of the Johnson & Johnson vaccine, citing that its potential benefits outweigh its risks.


 Issue PDF
Issue PDF