 Postpartum peripheral nerve injuries occur in approximately 0.3–2% of all deliveries. The majority of nerve injuries are attributed to intrinsic obstetric palsies secondary to compression or stretch of the nerve during delivery; however, the possibility that neuraxial anesthesia/analgesia contributes to the injury exists. It is important that anesthesia professionals create systems to identify women who have experienced postpartum lower extremity nerve injuries and connect patients with resources.
Postpartum peripheral nerve injuries occur in approximately 0.3–2% of all deliveries. The majority of nerve injuries are attributed to intrinsic obstetric palsies secondary to compression or stretch of the nerve during delivery; however, the possibility that neuraxial anesthesia/analgesia contributes to the injury exists. It is important that anesthesia professionals create systems to identify women who have experienced postpartum lower extremity nerve injuries and connect patients with resources.
Childbirth is the most common reason for admission to the hospital within the United States.1 While neurologic complications during pregnancy and delivery are still fortunately a relatively rare event, when they do occur, they can have a significant impact. Nerve injuries during childbirth are traditionally attributed to intrinsic obstetric palsies, either due to compression or stretch of the nerve. Although this is still true in a majority of cases, neuraxial procedures may contribute to a small proportion of these injuries. Given the rarity of these injuries, there are not accurate risk-prediction models. Therefore, anesthesia professionals should work with obstetricians and nurses to develop systems to identify women who do develop postpartum lower extremity nerve injuries and also provide these women with resources regarding symptomatology and mobility safety, especially if there is a motor component to the injury.
Common Peripheral Nerve Injuries
The incidence of postpartum peripheral nerve injuries varies in the literature from 0.3 to 2% of all deliveries.2-4 In a study of over 6,000 parturients, the most common peripheral nerve injuries found postpartum were to the lateral femoral cutaneous nerve and the femoral nerve. Less common nerves affected include common peroneal, lumbosacral plexus, sciatic, obturator, and radicular nerves (table 1).4
Table 1. Common Postpartum Peripheral Nerve Injuries and Proposed Mechanisms of Injury
| Nerve | Observed deficit | Proposed mechanism and location of injury and risk factors |
| Lateral femoral cutaneous nerve | Sensory: decreased on anterolateral thigh, “meralgia paresthetica” | Compression under the inguinal ligament with prolonged hip flexion, obesity (secondary to increased pressure at the inguinal ligament) |
| Femoral nerve | Sensory: decreased on anterior thigh and medial calf Motor: weak thigh flexion (if involvement of the iliacus nerve), weak knee extension |
Compression under the inguinal ligament secondary to prolonged hip flexion, abduction, and external rotation; retraction during cesarean delivery; possibly decreased perineural flow to the iliacus nerve |
| Lumbosacral plexus and sciatic nerve | Sensory: decreased on posterior thigh Motor: weak quadriceps, weak hip adduction, foot drop, involves multiple levels |
Compression due to fetal position, compression against pelvic rim, forceps assisted vaginal delivery |
| Obturator nerve | Sensory: decreased on medial thigh Motor: weak hip adduction, wide gait |
Compression due to fetal position, improper positioning, forceps assisted vaginal delivery |
| Common peroneal nerve | Sensory: decreased on lateral calf Motor: foot drop |
Lower extremity positioning, compression at fibular head either by hand or stirrups while anesthetized, compression while pushing |
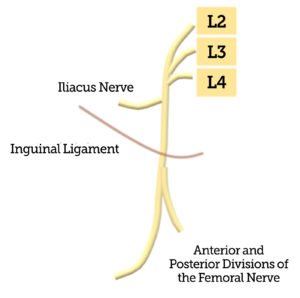
Figure 1. Illustration of the femoral nerve coursing underneath the inguinal ligament, and the iliacus nerve branching off more proximal to the inguinal ligament.
Lateral femoral cutaneous nerve injury occurs in approximately four out of 1000 parturients.4 The nerve, which supplies sensation to the anterolateral thigh, courses under the inguinal ligament, which makes it susceptible to compression while in lithotomy position. This purely sensory dysfunction, also known as meralgia paresthetica, is typically self-limited with a short recovery period, and can often be treated with nonsteroidal anti-inflammatory drugs or lidocaine patches.5
Femoral nerve injury is slightly less common, but involvement causes weakness in thigh flexion, knee extension, loss of patellar reflex, and sensory loss to the medial thigh and calf. The femoral nerve also courses under the inguinal ligament (figure 1) and compression at this point is traditionally believed to be the mechanism of injury.
Risk Factors
A variety of risk factors have been identified that contribute to peripheral nerve injuries. Some of these risk factors, such as duration of labor and mode of delivery are not modifiable. The attributable risk of any individual risk factor to the development of nerve injuries is not known. In this section, we will discuss several of the known risk factors.
Parturients who suffer a nerve injury are more likely to be nulliparous and spend longer time in the second stage of labor while in the lithotomy position than those without injury.4 Patients who have an assisted vaginal delivery (either with forceps or a vacuum device) are also more likely to have a postpartum peripheral nerve injury.4 Patients with neuraxial catheters are typically less mobile and maintain the same position for longer periods of time, which may make compression injury more likely. Anatomical variations in the epidural space could cause a high concentration of local anesthetic surrounding individual nerve roots (detected as an unequal distribution of a block) which could be neurotoxic at a high enough dose.6 In addition, a low concentration of local anesthetic through the epidural catheter should be considered. Although this has not been explicitly studied, it is reasonable to assume that patients with a dense analgesic block may be more likely to have compressive nerve injuries, as the local anesthetic may inhibit nociceptive warning signs of neuropathic pain.
A large retrospective study evaluating 20,000 laboring parturients who received neuraxial anesthesia identified a nerve injury incidence of 0.96%, with a higher incidence of lumbosacral plexus injuries.7 Risk factors identified included a forceps assisted vaginal deliveries, newborn birth weight >3.5 kg, late gestational age (≥41 weeks), and late initiation of the neuraxial procedure.7 They did not find any significant difference when looking at time of day of neuraxial placement or provider level of training. Out of the 19 injuries identified, four were attributed to direct trauma from either the Touhy needle or catheter to the nerve root, based on either electromyography, magnetic resonance Imaging, or a computerized tomography scan within 48 hours of delivery. Of those four injuries, three of the patients experienced a paresthesia during placement at the same level.7 In addition, in three of the four patients, the neuraxial procedure was performed with a cervical dilation greater than five centimeters, and all four of the patients had a documented difficult neuraxial placement with either severe pain or several attempts.7 Given this significance, it is especially important to include nerve injury in anesthetic consent for neuraxial procedures, and appropriately counsel patients if a traumatic placement occurs. Further evaluation needs to be conducted regarding appropriate troubleshooting when a paresthesia occurs during neuraxial placement, as this limited study indicated that these patients may be at higher risk of postpartum neuralgia. Our institutional practice is as follows: If a patient complains of a transient paresthesia with either the spinal or epidural needle, and it resolves without further intervention, injection may proceed. If the patient has a persistent paresthesia, the needle is moved away from the direction of the paresthesia. If the paresthesia occurs with spinal injection of local anesthetic, the injection is aborted and the intrathecal space is re-identified prior to injecting local. Finally, if the patient experiences a persistent paresthesia when the epidural catheter is threaded, the catheter is typically removed. At this point saline can be infused prior to re-attempting to thread the catheter to help expand the epidural space or the Touhy needle can be directed away from the direction of the paresthesia and the epidural space located again.
In a prospective observational study of new-onset postpartum lower-extremity nerve injuries, there were some injuries which did not fit the classic mechanism of nerve compression or stretch.4 Twenty-four patients had lateral femoral cutaneous nerve injuries, which are believed to be due to compression under the inguinal ligament in lithotomy position; however, four of these patients had a scheduled cesarean section. In addition, all 22 of the femoral nerve injuries had iliopsoas weakness, which is anatomically more cranial than the inguinal ligament, and also supporting the theory that nerve hypoperfusion may contribute to postpartum nerve injuries.4,8 Further work is needed to elucidate the role of blood pressure management on nerve injuries, and understand if treatment of blood pressure can prevent or mitigate certain nerve injuries. Our group is currently investigating risk factors for new onset postpartum lower-extremity nerve injuries in an Agency for Healthcare Research and Quality (AHRQ)-funded study. The study will evaluate the contribution of patient-related, as well as obstetric, neonatal, and anesthetic risk factors. We hope to further our understanding of these nerve injuries and identify potentially-modifiable factors.
Our Role as Anesthesia Professionals
Anesthesia professionals in collaboration with obstetrics play an important role in the identification of nerve injuries and connecting patients to resources for management of these injuries. An important consideration after a nerve injury is that patients are at a significant fall risk. If there is significant motor dysfunction, as seen with femoral neuropathies and lumbosacral plexus injuries, patients should be thoroughly assessed and counseled prior to discharge. Thankfully, prognosis on nerve injury during labor is favorable as recovery typically occurs on the order of weeks.2 In one study, the median duration of symptoms was two months.4 In another prospective study, the median time to recover from nerve injury was 18 days, but three women continued to have a neurologic deficit after a year.9
Anesthesia professionals should work with the obstetricians and nurses at their institution to ensure that all patients are evaluated after delivery and asked about symptoms consistent with postpartum lower extremity nerve injuries. If the post-anesthetic evaluation occurs immediately after delivery, the residual effects of the neuraxial block may mask any new-onset lower extremity nerve injuries. Ideally, on postpartum day one, either anesthesia professionals, obstetricians, or postpartum nurses should ask patients, are you having any difficulty walking or do you have any new numbness or weakness in your legs? If the patient endorses a new sensory deficit or weakness, these patients should have a more thorough evaluation by the anesthesia team (if the patient had a neuraxial anesthetic), or by a physiatrist or physical therapist if the patient did not have an anesthetic for delivery. If the pattern of injury is unclear, a neurology consult may be indicated as electromyography could assist in revealing individual nerve and muscle dysfunction.10 It is critically important for patients with any weakness to be evaluated for safe ambulating because there is the potential that the new mother could injure herself, or her infant, if she is unable to bear weight due to the nerve injury. A physical therapy evaluation will identify if any assistive devices such as a knee brace, orthotic shoe, or walker are needed prior to leaving the hospital. While typically no medical treatment is needed for new onset lower extremity nerve injuries, gabapentin could be considered if the patient complains of neuropathic pain. Studies in this patient population have been small, but gabapentin has not been shown to have an effect on the neonate through breast milk exposure.11 The more significant risk is that gabapentin has a wide side-effect profile, including increased fatigue, which may be undesirable. Lastly, emotional support is crucial, as a debilitating injury could further exacerbate any postpartum depression or anxiety; therefore, close follow-up with their obstetrician after delivery is vital. Typically, follow-up with a neurologist or physical medicine and rehabilitation is not needed, as long as symptoms continue to resolve and are not worsening in nature.
Summary
Postpartum nerve injuries are very rare, but can be very worrisome to both the patient and the anesthesia professional. The majority of nerve injuries are attributed to intrinsic obstetric palsies secondary to compression or stretch of the nerve during delivery. However, it is important to be aware of our role as it relates to hypoperfusion of nerves, traumatic neuraxial placement, and decreased motor function during labor secondary to dense local anesthetic. Further research is needed to help understand which factors place patients at increased risk for these injuries. Anesthesia professionals can directly impact safety by educating other perinatal providers and ensuring that all patients, regardless of whether or not they had a neuraxial procedure, are assessed by a provider for new-onset postpartum nerve injuries. If a nerve injury is detected, the nerve(s) affected should be identified, and the injury described in the medical record (motor, sensory, or mixed). The patient should also be evaluated by physical therapy or physiatry to ensure that the patient is safe to ambulate with her infant prior to discharge from the hospital.
Emery McCrory, MD, is an assistant professor of Anesthesiology at Northwestern University Feinberg School of Medicine, Chicago, IL.
Jennifer Banayan, MD, is editor, APSF Newsletter, and an associate professor of Anesthesiology at Northwestern University Feinberg School of Medicine, Chicago, IL.
Paloma Toledo, MD, MPH, is an assistant professor of Anesthesiology at Northwestern University Feinberg School of Medicine, Chicago, IL.
Ermery McCrory, MD, and Jennifer Banayan, MD, do not have any conflicts of interest. Paloma Toledo, MD, is supported by grants from the Agency for Healthcare Research and Quality and National Institute on Minority Health and Health Disparities (R03MD011628, R03HS025267, R18HS026169). The content is solely the responsibility of the authors and does not necessarily represent the official views of the Agency for Healthcare Research and Quality or the National Institute on Minority Health and Health Disparities.
References
- HCUP fast stats – most common diagnoses for inpatient stays. https://www.hcup-us.ahrq.gov/faststats/NationalDiagnosesServlet. Accessed March 2, 2021.
- Richards A, McLaren T, Paech MJ, et al. Immediate postpartum neurological deficits in the lower extremity: a prospective observational study. Int J Obstet Anesth. 2017;31:5–12.
- Scott DB, Tunstall ME. Serious complications associated with epidural/spinal blockade in obstetrics: a two-year prospective study. Int J Obstet Anesth. 1995;4:133–139.
- Wong CA, Scavone BM, Dugan S, et al. Incidence of postpartum lumbosacral spine and lower extremity nerve injuries. Obstet Gynecol. 2003;101:279–288.
- Meier T, Wasner G, Faust M, et al. Efficacy of lidocaine patch 5% in the treatment of focal peripheral neuropathic pain syndromes: a randomized, double-blind, placebo-controlled study. Pain. 2003;106:151–158.
- Verlinde M, Hollmann MW, Stevens MF, et al. Local anesthetic-induced neurotoxicity. Int J Mol Sci. 2016;17:339.
- Haller G, Pichon I, Gay FO, Savoldelli G. Risk factors for peripheral nerve injuries following neuraxial labour analgesia: a nested case-control study. Acta Anaesthesiol Scand. 2017;61:1203–1214.
- Biedmond A. Femoral neuropathy. In: Vinken P, Bruyn, GW, ed. Handbook of clinical neurology. Vol 8. New York: John Wiley & Sons; 1977:303–310.
- Tournier A, Doremieux AC, Drumez E, et al. Lower-limb neurologic deficit after vaginal delivery: a prospective observational study. Int J Obstet Anesth. 2020;41:35–38.
- Richard A, Vellieux G, Abbou S, Benifla JL, et al. Good prognosis of postpartum lower limb sensorimotor deficit: a combined clinical, electrophysiological, and radiological follow-up. J Neurol. 2017;264:529–540.
- Ohman I, Vitols S, Tomson T. Pharmacokinetics of gabapentin during delivery, in the neonatal period, and lactation: does a fetal accumulation occur during pregnancy? Epilepsia. 2005;46:1621–1624.


 Issue PDF
Issue PDF