Dear Rapid Response:
I would like to present a case of lower lip injury in a patient during intubation associated with the design of a Macintosh laryngoscope blade (BritePro Solo™, Flexicare Inc., Irvine, CA). The patient was a 75-year-old male who was scheduled for a urologic procedure. He was intubated smoothly with a 7.5 endotracheal tube (ETT) using a disposable Macintosh 3 laryngoscope. The Cormack-Lehane view was Grade 2b on laryngoscopy. While the nurse anesthetist was securing the ETT, I picked up the disposable laryngoscope to discard it and noticed blood on the blade and handle (figure 1).
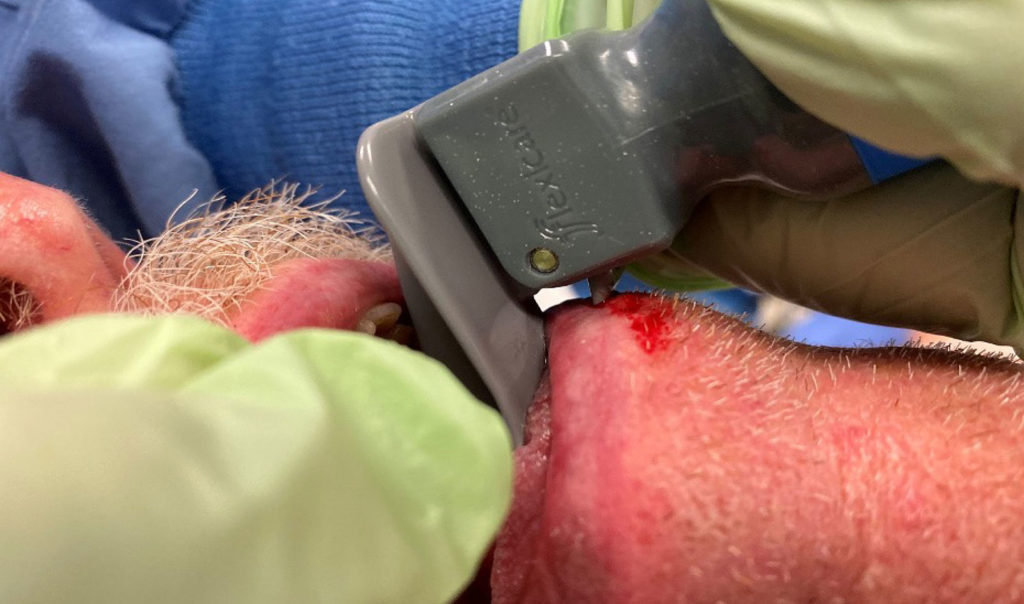
On examination of the patient’s face, we noticed a laceration on the lower lip with fresh bleeding (figure 2). We applied pressure, antibiotic ointment, and sterile gauze on the wound after which the bleeding stopped. By the end of the procedure, the laceration had sealed off with no significant injury or consequences. The patient was on a daily low dose of aspirin and was not worried about the incident.
Discussion

Figure 3: The blue arrow points to the hook on the laryngoscope blade (BritePro Solo from Flexicare, Inc.).
Soft tissue injuries during laryngoscopy and tracheal intubation are common especially in a difficult airway. However, we had no problem intubating this gentleman. Tongue injuries were found to have the highest incidence in a study on the electrophysiology population in a large academic center.1 In another review study, soft tissue trauma was observed in 52% of patients after direct laryngoscopy with Mac 3 or 4 blade with maximum injuries to the tongue (36.3%), followed by lower lip injuries (22.3%).2 Old age was not found to be a significant risk factor in that study except in injuries to the oral mucosa. However, injuries with significant bleeding are possible in patients on anticoagulation.
The reason for presenting this incident is to bring to your attention the design of this particular laryngoscope blade, BritePro Solo™ from Flexicare, Inc. The blade has an overhang or hook at the fulcrum where it attaches and engages at the handle (figure 3).
This hook helps in securing the blade to the handle, but has the potential to dig into the soft tissue of the lower lip area. This is a patient safety issue that warrants attention. There are other Macintosh blades available that do not have this design.
Thank you for your attention to this matter.
Tazeen Beg, MD
Assistant Professor of Anesthesiology
Division Chief, Non-Operating Room Anesthesia
Stony Brook University Medical Center
Tong J Gan, MD, MBA, MHS, FRCA
Professor and Chairman
Dept of Anesthesiology,
Stony Brook University Renaissance School of Medicine,
Stony Brook, NY
The authors have no conflicts of interest.
References
- Yan Z, Tanner JW, Lin D, et al. Airway trauma in a high patient volume academic cardiac electrophysiology laboratory center. Anesth Analg. 2013;116:112–117.
- Mourao J, Moreira J, Barabosa J, et al. Soft tissue injuries after direct laryngoscopy. J Clin Anesth. 2015;27:668–671.
In Response:
Safety of Macintosh Blade Design
Thank you for sharing this report with us and the opportunity to provide the rationale for different Macintosh blade designs. Flexicare regards patient safety and clinician feedback as the highest priorities for the organization. As an industry leader in the disposable laryngoscope market, and with roughly 15 million laryngoscopes sold in the United States over the past decade, we could not have succeeded without direct input from clinicians such as yourself and value your feedback.
We pride ourselves as an innovator in this space and the design of BriteBlade™ Pro/BritePro™ Solo was created with the clinician’s input and patient safety in mind. This particular product is designed to offer a disposable blade, but a reusable handle. More specifically, the design is intended to address the concerns around cross contamination amongst reusable laryngoscope handles. Studies have shown that decontamination using impregnated germicidal wipes is ineffective—between 75%–86% of “patient ready” reusable handles tested positive for bacterial contamination remained positive for bacterial contamination.1,2 The “hook”, as noted in the attached report, was created not only as a mechanism to attach the blade to the handle, but more importantly designed to help prevent the laryngoscope blade from touching the handle when collapsed after use, therefore, reducing the risk for cross contamination. Although Flexicare is highlighted as the product in use in the attached report, we are not the only disposable laryngoscope manufacturer with this design.3,4 This has become a standard design for other manufacturers in this market as it provides superior performance if used properly.
The adverse event in the attached report is the first of its kind reported to Flexicare with over 15 million laryngoscopes of this blade/handle design sold in the past decade. We have investigated the event to the best of our ability with the information provided thus far and escalated it to the highest levels of the organization. This includes not only input from our engineering, technical, and quality teams, but also our consulting anesthesiologist.
As stated in the report, although the patient may have been “intubated smoothly,” an airway with a Cormack Lehane score of 2b may require a significant amount of force to elevate the soft tissue and expose the vocal cords for endotracheal tube placement during direct laryngoscopy. It is apparent, as depicted in the pictures of the report, the incident occurred with the patient’s lip coming into direct contact with the “hook” of the blade. While it is impossible to determine based on the information provided, we want to reassert that proper blade size selection is extremely important. As noted in the attached report, a Mac 3 blade was selected for this 75-year-old male patient and may not have provided a sufficient amount of clearance between the blade and the soft tissue of the lower lip. The Mac 4 size was created specifically to address anesthetists concerns that a Mac 3 is too small for many male patients. “The standard adult Macintosh blade (equivalent to today’s size 3) proved too short for many patients, simply because Macintosh had tested his prototypes on women presenting for gynecological surgery.” (Sir R.R. Macintosh, letter to Sir A. Jephcott, May 9, 1983). Following requests from anesthetists, a larger adult blade (the size 4) was added to the Longworth range in 1951.4 By 1952, Foregger was also producing 4 sizes of Macintosh blade.”5
In summary, the product used in this report is specifically designed to reduce handle contamination and shares that design with other products on the market. The patient injury is not a product safety issue as much as it is an issue with blade size selection and direct laryngoscopy technique.
Ian Ross
Product Manager, Anesthesia
Flexicare Inc.
References
- Williams D, Dingley J, Jones C, Berry N. Contamination of laryngoscope handles. J Hosp Infect. 2010;74:123–128.
- Call T, Auerbach F, Riddell S, et al. Nosocomial contamination of laryngoscope handles: challenging current guidelines. Anesth Analg. 2009;109:479–483.
- Callisto Macintosh Laryngoscope Blade. http://www.timesco.com/product/callisto-macintosh-laryngoscope-blades/. Accessed March 23, 2021.
- Disposable laryngoscope blades. https://www.medicalexpo.com/medical-manufacturer/disposable-laryngoscope-blade-39116.html. Accessed March 23, 2021.
- Scott, J. and Baker, P.A. How did the Macintosh laryngoscope become so popular? Pediatr Anesth. 2009;19:24–29.
The information provided is for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of APSF. It is not the intention of APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information.