 For the past 2 years, the COVID-19 pandemic has bequeathed a plethora of barriers and unleashed its desolation on nearly every person on the planet. Infections have surpassed 445 million globally along with an excess of 5.9 million deaths;1,2 and these numbers continue to climb as the globalized vaccine efforts lag. Health care systems worldwide confronted unique challenges as hospital beds became saturated with infected patients during various surges throughout the pandemic. The challenge for health care systems to mitigate risks to patients while providing standard services remains, as immunization for SARS-CoV-2 continues to propagate and the in-hospital census of infected patients fluctuates.
For the past 2 years, the COVID-19 pandemic has bequeathed a plethora of barriers and unleashed its desolation on nearly every person on the planet. Infections have surpassed 445 million globally along with an excess of 5.9 million deaths;1,2 and these numbers continue to climb as the globalized vaccine efforts lag. Health care systems worldwide confronted unique challenges as hospital beds became saturated with infected patients during various surges throughout the pandemic. The challenge for health care systems to mitigate risks to patients while providing standard services remains, as immunization for SARS-CoV-2 continues to propagate and the in-hospital census of infected patients fluctuates.
Health care systems need to return to normalcy and provide surgical and procedural services safely, while mitigating the risk to SARS-CoV-2 positive patients. Determining the optimal timing of procedures for patients who have recovered from COVID-19 infection and the appropriate level of preoperative evaluation are challenging given the current lack of evidence or precedent for this disease. According to the Joint Statement Created by the APSF and ASA, “Elective surgeries should be performed for patients who have recovered from COVID-19 infection only when the anesthesia professional and surgeon or proceduralist agree jointly to proceed.”3 This should be done in conjunction with shared decision-making with the patient.
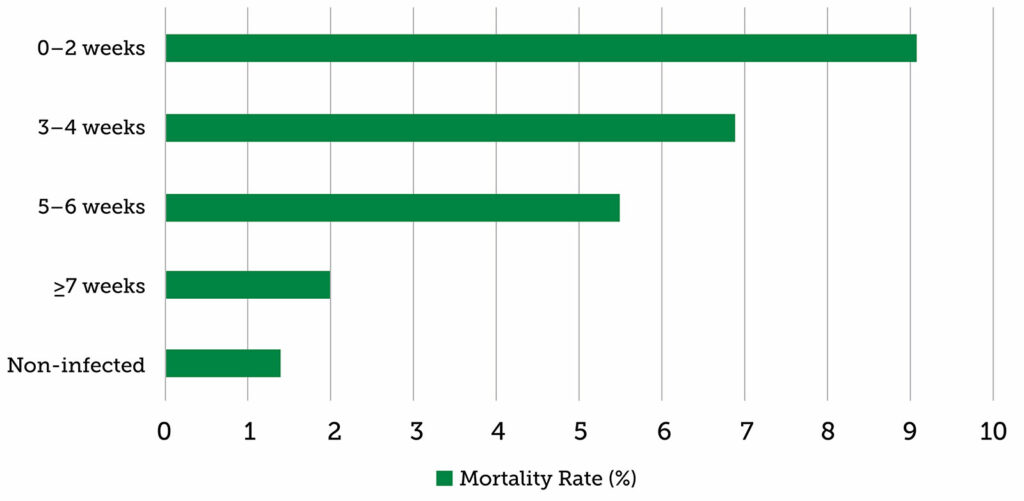
The most robust data analyzing perioperative outcomes of patients with SARS-CoV-2 infection emerged from the COVIDSurg Collaborative and GlobalSurg Collaborative study.4 This was an international, prospective, cohort study that included a total of 140,231 patients in 1, 674 hospitals throughout 116 countries. They assessed the postoperative complications in patients with a diagnosis of SARS-CoV-2 infection with a primary outcome measure of 30-day postoperative mortality and a secondary outcome measure of 30-day postoperative complications (defined as pneumonia, acute respiratory distress syndrome, and unexpected postoperative ventilation). For patients with a SARS-CoV-2 diagnosis prior to surgical intervention, the mortality rates were as follows: 9.1% 0–2 weeks prior, 6.9% 3–4 weeks prior, 5.5% 5–6 weeks prior and 2.0% at ≥7 weeks (Figure 1). The mortality rates in the ≥7 weeks group demonstrated no significant difference when compared to the noninfected control group. Amongst the SARS-CoV-2 positive subjects, symptomatic patients demonstrated a significantly higher 30-day mortality rate across all time interval subgroups, when compared to those who were asymptomatic or had resolution of symptoms at the time of surgery. SARS-CoV-2 infected patients 0–2 weeks, 3–4 weeks, and 5–6 weeks prior to surgery demonstrated higher rates of postoperative pulmonary complications as well, and those with ongoing symptoms demonstrated the greatest risk. The ≥7 weeks subgroup complication rates mirrored that of the noninfected subjects.4
The beginning of the COVID-19 pandemic resulted in an overwhelming amount of admissions. Attempts to minimize in-hospital transmissions coupled with the lack of initial data on elective surgical risk for COVID-19 positive patients discussed in the previous section caused millions of cancellations and delays of elective surgeries worldwide. The National Institute for Health Research (NIHR) unit on Global Surgery collected data from 1,674 hospitals to address the perioperative risk of SARS-CoV-2 infected patients. This dataset delivers valuable information to providers responsible for mitigating risk to surgical candidates. Nonetheless, the large-scale study is not without its limitations. The diagnosis of a SARS-CoV-2 infection was not standardized and relies on the unique preoperative testing strategies of each institution, leaving the true time from infection-to-surgery in question. Given the reliance on a single routine preoperative test, it is likely that a subgroup of subjects who recently recovered from a previous SARS-CoV-2 infection were misclassified as not infected. The pragmatic difficulties associated with conducting a cohort study of this scale rendered limitations in important subanalysis; assessing outcomes amongst groups controlling for type of surgery, type of anesthesia, airway device used, and chronic comorbidities would have been ideal with this dataset. The secondary outcomes measured in the study assessed the rates of pulmonary complications; however, it is well established that SARS-CoV-2 can cause an array of systemic complications including: thromboembolic events, myocarditis, overt stroke, cognitive deficiencies, and kidney injury.5 Lastly, data for this study were collected in October of 2020, prior to widespread vaccine distribution efforts and the emergence of a number of variants. As immunization propagates, through inoculation and infection, these outcomes will likely change.
Subsequent studies aimed at assessing similar findings have replicated the outcomes found in the COVIDSurg Collaborative study. Deng et al., recently published a retrospective study suggesting worse postoperative outcomes in those patients who had surgery within 8 weeks of SARS-CoV-2 diagnosis.6 In addition, the authors reported that a shorter time interval between diagnosis and intervention correlated with a higher rate of postoperative respiratory complications.6
The American Society of Anesthesiologists (ASA) and the American Patient Safety Foundation (APSF) released a joint statement in March of 2021 (recently updated in February 2022) with recommendations advising clinicians of the appropriate timing of elective surgeries in SARS-CoV-2 infected patients. Based on the accumulated data at that time, the statement issued the following recommended waiting times for infected patients:
- 4 weeks for asymptomatic patients or recovered from mild nonrespiratory symptoms
- 6 weeks for symptomatic patients who did not require hospitalization
- 8–10 weeks for a symptomatic patient who is diabetic, immunocompromised, or hospitalized
- 12 weeks for patients admitted to the Intensive Care Unit due to COVID-19 infection.3
These recommendations are not comprehensive and may not reflect patient demographics that providers will inevitably encounter. They are fluid recommendations that offer providers a framework to help estimate a risk/benefit ratio to decipher the appropriate timing of intervention patients need. The United Kingdom released similar guidelines, in February 2022, from a conglomerate of multidisciplinary perioperative specialists with analogous recommendations, discouraging operative intervention within 7 weeks of SARS-CoV-2 diagnosis.7
Although there is increasing information to address the timing of surgery after COVID-19 infection, the data and science unfortunately continues to lag behind the emerging variants, and data that supports the premise that vaccinated patients have a lower a risk of postoperative complications as compared to unvaccinated patients.8 According to the CDC, the Omicron variant causes less severe disease, and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.9 Perioperative providers used these observations to prematurely conclude that the vaccinated patients recovering from Omicron should be at less risk for adverse events. However plausible, such a conclusion remains unproven. While there continues to be emerging data that can provide clinicians and patients with improved management strategies, health care providers will continue to require more data to fill in the knowledge gaps of the nuanced subanalyses.
Jeremy Laney, MD, is an assistant clinical professor in the Department of Anesthesiology, Cardiothoracic Division, Keck School of Medicine of USC.
Joseph W. Szokol, MD, FASA, is chief health policy officer of the American Society of Anesthesiologists and clinical professor in the Department of Anesthesiology at the Keck School of Medicine of USC. He is also the executive director of Mentorship and Physician Development in the Department of Anesthesiology at the Keck School of Medicine of USC.
The authors have no conflicts of interest.
References
- Centers for Disease Control. CDC COVID Data tracker. https://covid.cdc.gov/covid-data-tracker Accessed on March 25, 2022.
- World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard. https://covid19.who.int Accessed on March 25, 2022.
- ASA and APSF Joint Statement on Elective Surgery/Procedures and Anesthesia for Patients after COVID-19 Infection. Updated Feb. 22, 2022. https://www.asahq.org/about-asa/newsroom/news-releases/2022/02/asa-and-apsf-joint-statement-on-elective-surgery-procedures-and-anesthesia-for-patients-after-covid-19-infection. Accessed on April 12, 2022.
- COVIDSurg Collaborative, GlobalSurg Collaborative, Nepogodiev D, et al. Timing of surgery following SARS-CoV-2 infection: an international prospective cohort study. Anaesthesia. 2021;76:748–758.
- Wijeysundera DN, Khadaroo RG. Surgery after a previous SARS-CoV-2 infection: data, answers and questions. Anaesthesia. 2021;76:731–735.
- Deng JZ, Chan JS, Potter AL, et al. The risk of postoperative complications after major elective surgery in active or resolved COVID-19 in the United States. Ann Surg. 2022;275:242–246.
- El-Boghdadly K, Cook TM, Goodacre T, et al. Timing of elective surgery and risk assessment after SARS-CoV-2 infection: an update: a multidisciplinary consensus statement on behalf of the Association of Anaesthetists, Centre for Perioperative Care, Federation of Surgical Specialty Associations, Royal College of Anaesthetists, Royal College of Surgeons of England. Anaesthesia. 2022 Feb 22: 1–8. 35194788. Accessed on April 19, 2022.
- Prasad NK, Lake R, Englum BR, et al. COVID-19 vaccination associated with reduced postoperative SARS-CoV-2 infection and morbidity. Ann Surg. 2022;275:31–36.
- Omicron variant: what you need to know. Updated Mar. 29, 2022. https://www.cdc.gov/coronavirus/2019-ncov/variants/omicron-variant.html. Accessed on April 19, 2022.