Anesthesia equipment malfunction may contribute to increased patient morbidity and mortality. We present a complication resulting from an accidental break in the Laryngotracheal Analgesia Device (International Medical Systems (IMS) Lidocaine Laryng-O-Jet Kit® (LOJ)), Figure 1.
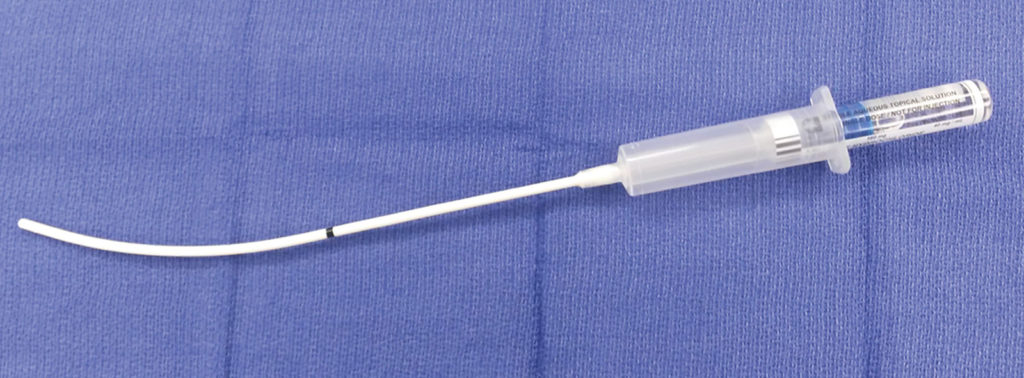
Figure 1. Depicts an intact Laryngotracheal Analgesia Kit at University of Texas, Southwestern Medical Center.
A 59 year-old, morbidly obese male with symptomatic atrial fibrillation and rapid ventricular response required anesthesia for a transesophageal echocardiogram with subsequent cardioversion. The decision was made to perform general endotracheal anesthesia by providing topical airway anesthesia (TAA) via a Lidocaine Laryng-O-Jet Kit®. This device was used to decrease airway reactivity without the use of opioids in order to potentially reduce the risk of opioid-induced ventilatory impairment.
Using video laryngoscopy, the LTA was passed through the vocal cords and upon withdrawal, the tip broke off and remained in between the patient’s vocal cords, Figure 2. Multiple attempts at retrieval via Yankauer suction were unsuccessful. An endotracheal tube was advanced through the vocal cords over the retained portion of the LTA in order to provide oxygen to the patient during this time. Pulmonology was consulted, and the foreign body was retrieved successfully via bronchoscopy with a snare device. The planned procedure was completed and patient was successfully extubated.

Figure 2. Depicts the horizontal image of a broken Laryngotracheal Analgesia Kit discussed in the reported case.
Author’s Recommendations
This event did not result in any long-term sequelae. The February 2018 APSF Newsletter reported a similar incident with a mucosal atomizer.1 The present article provides potential management strategies in case an LTA fracture occurs: 1) Avoid repeated attempts at retrieval via suction or forceps because the broken portion may cause tracheal rupture or vocal cord injury. 2) Secure the airway and subsequently retrieve the device via bronchoscopy with a snare with a specialist’s assistance, if available.
Osamudiamen Obanor, MD, is a resident anesthesiologist in the Department of Anesthesiology and Pain Medicine at the University of Texas Southwestern Medical Program, Dallas, TX.
Omaira Azizad, MD, is a staff anesthesiologist in the Department of Anesthesiology and Pain Medicine at the University of Texas Southwestern Medical Program, Dallas, TX.
Irina Gasanova, MD, PhD, is a staff anesthesiologist in the Department of Anesthesiology and Pain Medicine at the University of Texas Southwestern Medical Program, Dallas, TX.
None of the authors have any conflicts of interest/disclosures as they relate to this article.
Reference
- Wang M, Mathur P, Abdelmalak B. Airway topicalization atomizer parts break off in patient’s airway. APSF Newsletter 2018;32:74. https://www.apsf.org/article/airway-topicalization-atomizer-parts-break-off-in-patients-airway/
Reply:
This is in response to Dr. Obanor’s report regarding International Medical Systems (IMS) Lidocaine Laryng-O-Jet Kit (LOJ), Lot No. DL067G7, Stock No. 6300, in which it was reported that upon removal of the device, the tip of the cannula broke and was lodged between the patient’s vocal cords. Upon receipt of this report, an investigation was conducted to determine the root cause that may have contributed to the reported incident.
The batch records for this lot were reviewed and no anomalies were noted at the time of manufacturing, testing, or release. We inspected the reserve samples from Lot DJ067G7 and all other product lots that were manufactured with the same Laryng-O-Jet injector lots used in Lot DJ067G7. All of the reserve samples inspected passed visual inspection including the inspection of the injector’s tubes for cracks or breakage. The reserve samples were also inspected for the alignment of the holes on the cannula and all units met specification. Random sample units from each lot were also inspected for functionality, and all units inspected met specification.
The subject unit was not available to be returned for investigation. However, a photo was provided showing a unit with a broken tip. It was reported that at the time of the incident, the device was inserted using the assistance of a video laryngoscope and the product did not get stuck on anything and was not bent during the insertion. Upon attempted removal of the device, the end of the cannula was found to have broken off. The patient underwent bronchoscopy for removal of the broken piece and was subsequently discharged with no further complications.
The Laryng-O-Jet applicator tubes are manufactured with Makrolon® Polycarbonate. During the injector assembly process, the assembled LOJ Injector is subjected to a glue curing tunnel and to a bending fixture, neither one of which degrades the Makrolon material. The bend in the LOJ tube is created with a pre-determined amount of heat and is anatomically curved for administration of lidocaine in the larynx and trachea. The LOJ tube is 100% inspected under magnification at the conclusion of the assembly process; if any crack or breakage is found, the tube is culled out at the time. Additionally, the finished product is inspected twice under magnification for any damage before final packaging. Per the product labeling, the LOJ tube should not be further bent or manipulated prior to use. Additionally, per the product labeling, caution is needed with laryngoscope use to avoid cannula breakage. We have received no other similar reports from other customers for Lot DJ067G7. Please note that the report of the incident was submitted to the FDA via the Safety Reporting Portal as an expedited report.
We value our customers’ concerns. The feedback we receive provides us with valuable information for evaluating our processes and maintaining our quality standards.
Sincerely,
Alan Go
Manager, Quality Systems
The information provided is for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of APSF. It is not the intention of APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information.

