In anesthesiology, the use of single-use devices has sky-rocketed over the last two decades. Single-use devices used in anesthesia practice, including laryngoscopes, video laryngoscopes, blood pressure cuffs, medical gowns, operating room hats and attire, and pulse oximetry probes are often discarded immediately after one-time use. In 2019, the U.S. disposable medical device market was already a $66.9 billion industry, and the industry continues to grow, currently increasing at a compound annual growth rate of 16.7%.1
Manufacturers tout the ease and safety of single-use products. Advocates suggest easier infection control with their use. Ambiguity and changes in processing requirements for medical equipment have led many health care organizations to default to disposable devices over fear of citations by accrediting bodies.
Recent pandemic-related supply chain disruptions starkly exposed the dangers of heavy reliance on disposable devices. Health care systems often keep relatively low supplies of these single-use devices, reordering only short-term supplies reflecting a “just in time” mentality focused on keeping costs low. Anesthesia professionals have faced occasional medication and product shortages in the past, but the frequent and profound shortages of equipment, supplies, and medications over the last three years is an abrupt departure from modern clinical practice in the United States. As anesthesia practices scramble for alternative equipment and supplies, they may be forced to use second- or third-line devices or medications, potentially creating significant patient safety concerns related to the frequent introduction of new and unfamiliar supplies.
These product shortages, and the supply chain fragility they expose, should prompt an evaluation of disposable device usage. Further, an increasing awareness of anesthesiology’s outsized environmental footprint, and the consequent impact on public health, has prompted many health care systems to re-evaluate purchasing processes to decrease the massive waste and greenhouse gas (GHG) emissions produced from surgical practices.
In this review, we outline methods to increase both sustainability and supply chain resiliency in anesthesiology practices based on evidence-based analysis of product safety, infection risk, and greenhouse gas emissions (GHG) related to product manufacturing, use, and disposal. Ultimately, maximizing the utilization of reusable devices promotes patient safety by reducing the risk of shortages of essential products. Reusable products confer a reduced environmental footprint by creating less physical waste and offer the potential of enormous cost benefits for health care systems.
Building sustainable, resilient, and cost-effective anesthesia practices demands an understanding of product costs and resource utilization. Life cycle costing and life cycle assessments are important concepts that are helpful for practice leaders who are evaluating purchase decisions. These life cycle concepts are dependent on the number of times a product is used and are determined by product-related energy costs, GHG emissions, and economic costs over its lifespan (Table 1).2 Additionally, it is imperative for health care organizations to understand waste management in the context of environmental impact, patient and community safety, and cost benefit. The Waste Hierarchy (Figure 1) is one tool practice leaders can utilize when evaluating potential device purchases.3 For example, the waste hierarchy suggests that a properly cleaned, re-used pulse oximetry probe offers less environmental impact (and is cheaper to hospital systems) than a recycled or discarded probe. Successful waste management improves the health of communities by reducing landfilled and incinerated waste, both of which may produce soil, water, and air toxins and other hazardous by-products. A waste management program that prioritizes properly cleaned, reusable devices improves planetary health by decreasing fossil fuel usage, carbon dioxide emissions, and energy required for the manufacture, transport, and disposal of these single-use items.
Table 1: Terms Related to Sustainable Device Purchasing.2,6

Figure 1: Recommended order of actions to manage waste, from most to least environmentally preferred. Goals of this process are to save resources and energy, reduce waste and its toxicity, and save money for health care systems and patients.
SURGICAL SITE INFECTION AND REUSABLE PRODUCTS
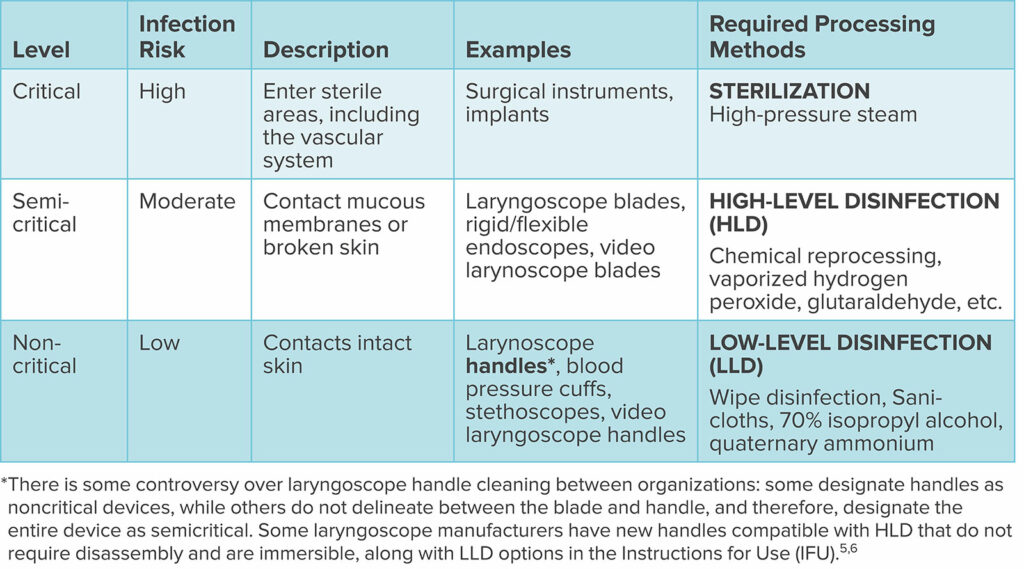
Prevention of surgical site infections (SSIs) is a priority for any health care system. SSIs are associated with an increased hospital length of stay, increased risk of readmission, and increased morbidity and mortality.4 While easy infection control is touted as a benefit to disposable device use, there is no evidence that reusable equipment leads to increased SSI when appropriate cleaning protocols are performed. In fact, the Center for Disease Control and Prevention has requirements for device disinfection and sterilization based on the Spaulding Device Cleaning Classification. This system classifies cleaning techniques and reprocessing methods for specific devices according to level of patient contact and infection risk during use (Table 2).5 In addition, all medical equipment should be cleaned in accordance with the manufacturer’s instructions for use, which provide additional guidance to maintain device safety and longevity, based on tested cleaning protocols.
Table 2: Spaulding Device Cleaning Classification.
COMMONLY USED DISPOSABLE AND REUSABLE ANESTHESIA PRODUCTS
Laryngoscope Handles and Blades
Evaluations of the life cycle assessment and total life cycle costs of reusable and disposable laryngoscope handles and blades show significant environmental and cost savings with reusable equipment,6 without compromising patient safety when cleaned according to established guidelines.
According to the Spaulding Classification, laryngoscope handles may be considered either low or moderate infection risk as consensus varies between professional organizations, requiring either “low-level” disinfection, using chemical wipes or 70% alcohol, or “high-level” disinfection, using chemical reprocessing. Either protocol still confers environmental benefit over single-use laryngoscope handles. For example, a disposable metal handle produces 20 times more GHG emissions per use than a low-level disinfected handle and nearly 27 times more GHG emissions than a high-level disinfected reusable steel handle, assuming a life span of 4000 uses. Reusable laryngoscope blades, which require high-level disinfection at minimum, are still environmentally preferable over single-use metal blades. These reusable blades produce between 2–7 times less GHG emissions per use, depending on sterilization or high-level disinfection, respectively.6
Safety data showing a clear benefit of disposable laryngoscopes over adequately cleaned reusable laryngoscope handles and blades are lacking. Further, there is no evidence to suggest infection transmission in the US from reusable handles or blades, appropriately cleaned according to Spaulding Classification criteria and the manufacturer’s instructions for use.7
Case reports of infection transmission in neonatal intensive care units describe inadequately disinfected laryngoscopes where current cleaning protocols were not followed.8 Older data show contamination of reusable blades and handles, but the majority of studies were judged to be very low or low quality, with inconsistent cleaning protocols.9 A study examining laryngoscope handles cleaned with low-level techniques demonstrated no pathogenic bacterial or viral colonies and only rare to few nonpathogenic bacterial colony growth, which decreased in samples as the study continued, perhaps reflecting increased attention to handle cleaning during the study period.10 Further, this bacterial contamination is of unclear significance, given that 50% of sterile fields are contaminated within a few hours, even in empty operating rooms,11 and bacteria have been cultured from sterile trays immediately after opening.12 In addition, anesthesia personnel routinely use laryngoscope handles without sterile gloves and even single-use devices are opened, touched, and contaminated during OR set-up. These studies highlight the importance of high-quality, careful protocolized cleaning and reprocessing.
When the lifetime costs of reusable laryngoscopes, including those related to reprocessing and device attrition, are evaluated against disposables, a reusable handle needs to be used only 4–5 times for cost benefit compared to a disposable handle, and reusable blades only 5–7 times compared to single-use blades. In one year of clinical practice, reusable handles and blades confer significant savings to health systems, regardless of cleaning protocol, despite initial higher upfront costs.6
Reusable products do not just confer cost benefit, they also may improve patient safety by safeguarding against critical shortages. The SARS-CoV-2 pandemic led to widespread shortages in single-use plastic video laryngo-scope blades. Many institutions adapted by reprocessing blades internally or through third-party reprocessing companies, demonstrating the supply chain vulnerability of single-use products versus reusable products, particularly during periods of high demand. The pandemic experience highlighted that reprocessing of single-use devices can be performed safely. External third-party reprocessing is highly regulated by the FDA, designed to restore products to their original quality, function, and sterility, while maintaining safety warranties. Even with stringent protocols, reprocessed device costs are still half the price of new equipment.13
BLOOD PRESSURE (BP) CUFFS
Life cycle data suggest reusable BP cuffs have far less environmental impact over disposable cuffs. Reusable BP cuffs are environmentally better in all clinical use settings, with a wide variety of cleaning protocols, generating close to 40 times fewer GHG emissions than disposable cuffs over their lifetime. Life cycle cost analysis demonstrates that reusable BP cuffs are far cheaper than disposable cuffs over their lifetime in both outpatient and procedural areas.14
From a patient safety perspective, there are no data that reusable blood pressure cuffs are responsible for increased infection versus disposable cuffs. However, inadequately disinfected reusable cuffs may be contaminated by bacteria.15 Single use cuffs may also be contaminated by the hands of health care workers if they are not frequently sanitized. Both scenarios emphasize the importance of protocolized cleaning techniques and handwashing. As noncritical devices, defined by the Spaulding classification, BP cuffs require low-level disinfection between patients.
Gowns (surgical and isolation)
Reusable surgical gowns and isolation gowns confer significant patient safety benefits because they are less vulnerable to critical shortages. Dramatic supply chain advantages emerged during the SARS-CoV-2 pandemic. In fact, institutions with reusable isolation gowns during the pandemic had a protective advantage compared to those institutions using disposables, when many resorted to garbage bags to provide personal protective equipment in the face of global shortages.
Further, reusable gowns are more durable, offering improved infection protection and substantial cost savings due to their durability and sustainability. A comparison of disposable vs. reusable medical gowns (laundered up to 75 times, according to CDC guidelines) showed that lower-level disposable gowns did not meet the industry standard, the Association of Advancement Instrumentation PB70 Performance Specifications, for impact penetration water resistance. In addition, all the tested disposable gowns (Level 1, 2, and 3) failed to meet the standard American Society of Testing and Materials performance requirements for breaking strength. The reusable gowns performed much better, meeting both performance requirements throughout 75 washings.16
The environmental footprint of reusable gowns is far smaller than disposables: one life cycle assessment showed that the use of reusable surgical gowns decreased natural resource energy consumption by 64%, GHG emissions by 66%, blue water use by 83%, and solid waste generation by 84%. Blue water consumption is water removed from the water supply and not returned.17
Similar analyses confirm the environmental benefit from reusable isolation gowns, which confer a 28% reduction in energy consumption, a 30% decrease in GHG emissions, a 41% reduction in blue water consumption, and a 93% reduction in solid waste generation.18
OR Hats and Arm Coverings
Over the last decade, guidelines regarding head coverings for operating room personnel have shifted, with current recommendations favoring clean, but not necessarily disposable head wear. Further, from a patient safety perspective, most published evidence suggests that reusable hats confer at least equivalent, if not better, infection protection with a far smaller environmental footprint.
In 2015, the Association of periOperative Registered Nurses released guidelines on OR attire directed at decreasing surgical site infection (SSI) risk. The guidelines, requiring disposable bouffant hats and long sleeves among all nonscrubbed personnel, were accepted by accrediting bodies, including the Centers for Medicare and Medicaid Services, despite no definitive evidence to support the recommendation.19
These guidelines were followed by a series of published studies demonstrating no infection benefit with disposable versus reusable hats. One study of 70 surgeons performing over 6000 ventral hernia repairs showed no significant difference in surgical site infection with respect to surgeon head wear.20 Another study showed potential safety benefit for nondisposable hats, showing that airborne particle contamination was significant lower with cloth “skull” hats versus disposable bouffants. Passive microbial shedding was also significantly higher with disposable bouffants compared to disposable skull caps and other cloth hats. In fact, disposable bouffant hats were the most permeable and had largest pore size.21
Current guidelines from multiple organizations, including the American Society of Anesthesiology, American College of Surgeons, and Association of periOperative Registered Nurses, now confirm the lack of scientific evidence showing any association between head covering type, extent of hair coverage, and SSIs, with new recommendations simply favoring clean surgical coverings during procedures.22
It is unclear why the use of disposable hats and gowns remains so entrenched, despite the lack of evidence for improved infection control. While disposable products may seem cheaper, cost analyses demonstrate that these items confer high costs to health care systems. In a recent study of over 12,000 matched pairs of surgical patients, more strict attire of disposable bouffants, disposable beard covers, and disposable long-sleeved jackets among nonscrubbed operating room personnel drove total attire costs up 10 to 20 times per person entering the OR without improving surgical site infection risk.23
Pulse oximetry probes
The use of disposable pulse oximetry probes is widespread and reflexive in anesthesia practices. From an individual patient safety standpoint, there is a paucity of data showing any difference in the safety profile and accuracy between reusable versus disposable pulse oximetry probes, nor any data showing increased infection risk with appropriately cleaned reusable probes versus disposable probes. Further, increased availability of reusable pulse oximetry probes may improve safety in resource-poor countries. Pulse oximetry has been included on the World Health Organization’s Surgical Safety Checklist since 2007, but is still missing from 15% of operating rooms in resource-poor settings.24 Further, decreased waste generation and resource utilization with the use of reusable pulse oximetry probes can lead to improved community and planetary health.
The potential clinical benefits of reusable probes are matched by cost savings to clinical practices, as well. Data from emergency medicine literature suggest that reusable pulse oximeters may provide equivalent monitoring without safety concerns and with less costs. One quality improvement project performed by an emergency department with roughly 70,000 annual patient visits showed a 56% reduction in cost with reusable pulse oximeters. Likewise, monthly pulse oximeter acquisition costs dropped by $30,000.25 Another analysis of an emergency medicine department with 55,000 annual visitors demonstrated annual savings of $129,000 with reusables. To generate cost-savings, a reusable monitor needed to be used 22 times.26
CONCLUSIONS
As highlighted, no evidence suggests that single-use devices in anesthesiology with low or intermediate infection risks provide better or safer care for our patients compared to appropriately cleaned reusable devices. Rather, patient safety is put at risk when heavy reliance on disposable clothing, equipment, and devices renders hospital systems vulnerable to severe supply chain shortages, prompting scrambles for products which may be inferior, unfamiliar, and more costly. Further, massive amounts of disposable medical equipment are either incinerated or go to landfills, with obvious environmental and public health consequences. As such, sustainability standards, greenhouse gas emissions, lifetime costs, and supply chain resiliency should be emphasized during purchasing decisions in hospital systems, along with evaluation of device quality, safety, and ease of use.
Molly Herr, MD, is an instructor of anesthesiology and perioperative medicine at the Mayo Clinic, Rochester, MN.
Leal Segura, MD, is an assistant professor of anesthesiology and perioperative medicine at the Mayo Clinic, Rochester, MN
The authors have no conflicts of interest.
REFERENCES
- Medical disposables market size, share & trends analysis report by product (wound management products, drug delivery products), by raw material (plastic resin, nonwoven material), by end-use, by region, and segment forecasts, 2021–2028. Grand View Research. Report ID: GVR-1-68038-258-7; number of pages 117; electronic PDF. https://www.grandviewresearch.com/industry-analysis/medical-disposables-market Accessed March 7, 2023.
- Rebitzer G. Integrating life cycle costing and life cycle assessment for managing costs and environmental impacts in supply chains. 2002. In: Seuring, S., Goldbach, M. (eds) Cost Management in Supply Chains. Physica, Heidelberg, 127–149.
- Health care without harm. US and Canada. Issue: Waste Management. Five principles for sustainable health care waste management. https://noharm-uscanada.org/issues/us-canada/waste-management. Accessed March 7, 2023.
- Seidelman JL, Mantyh CR, Anderson DJ. Surgical site infection prevention: a review. JAMA. 2023;329:244–252. PMID: 36648463
- Centers for Disease Control and Prevention. Spaulding EH. A rational approach to disinfection and sterilization. Guideline for disinfection and sterilization in healthcare facilities (2008). https://www.cdc.gov/infectioncontrol/guidelines/disinfection/rational-approach.html Accessed March 7, 2023.
- Sherman J, Raibley L, Eckelman M. Life cycle assessment and costing methods for device procurement: comparing reusable and single-use disposable laryngoscopes. Anesth Analg. 2018;127:434–443. PMID: 29324492
- Sherman J, Hopf H. Balancing infection control and environmental protection as a matter of patient safety: the case of laryngoscope handles. Anesth Analg. 2018;127:576–579. PMID: 29324490
- Neal TJ, Hughes CR, Rothburn MM, Shaw NJ. The neonatal laryngoscope as a potential source of cross-infection. J Hosp Infect. 1995;29:315–316. PMID: 7499813
- Negri de Sousa AC, Levy CE, Freitas MIP. Laryngoscope blades and handles as sources of cross-infection: an integrative review. J Hosp Infect. 2013;83:269–275. PMID: 23332194
- Call TR, Auerbach FJ, Riddell SW, et al. Nosocomial contamination of laryngoscope handles: challenging current guidelines. Anesth Analg. 2009;109:479–483. PMID: 19608822
- Wistrand C, Söderquist B, Sundqvist A-S. Time-dependent bacterial air contamination of sterile fields in a controlled operating room environment: an experimental intervention study. J Hosp Infect. 2021;110:97–102. PMID: 33516797
- Dalstrom DJ, Venkatarayappa I, Manternach AL, et al. Time-dependent contamination of opened sterile operating-room trays. J Bone Joint Surg AM. 2008;90:1022–1025. PMID: 18451394
- Practice Greenhealth. Greening the OR implementation module: medical device reprocessing. https://practicegreenhealth.org/sites/default/files/upload-files/gorimpmod-meddevicerepr_r5_web_0.pdf Accessed March 7, 2023.
- Sanchez SA, Eckelman MJ, Sherman JD. Environmental and economic comparison of reusable and disposable blood pressure cuffs in multiple clinical settings. Resour Conserv Recy. 2020;155:104643. https://doi.org/10.1016/j.resconrec.2019.104643
- Grewal H, Varshney K, Thomas L, et al. Blood pressure cuffs as a vector for transmission of multi-drug resistant organisms: colonisation rates and effects of disinfection. Emerg Med Australas. 2013;25:222–226. PMID: 23759042
- McQuerry M, Easter E, Cao A. Disposable versus reusable medical gowns: a performance comparison. Am J Infect Control. 2021;49:563–570. PMID: 33091509
- Vozzola E, Overcash M, Griffing E. An environmental analysis of reusable and disposable surgical gowns. AORN J. 2020 Mar;111:315–325. PMID: 32128776
- Vozzola E, Overcash M, Griffing E. Environmental considerations in the selection of isolation gowns: a life cycle assessment of reusable and disposable alternatives. Am J Infect Control. 2018;46:881–886. PMID: 29655666
- Guideline summary: surgical attire. AORN J. 2015;101:198–200. https://doi.org/10.1016/j.aorn.2014.11.007
- Haskins IN, Prabhu AS, Krpata DM, et al. Is there an association between surgeon hat type and 30-day wound events following ventral hernia repair? Hernia. 2017;21:495–503. PMID: 28631104
- Markel TA, Gormley T, Greeley D, et al. Hats off: a study of different operating room headgear assessed by environmental quality indicators. J Am Coll Surg. 2017;225:573–581. PMID: 29106842
- American College of Surgeons. Consensus statements: A statement from the meeting of ACS, AORN, ASA, APIC, AST, and TJC concerning recommendations for operating room attire. Feb. 27, 2018. https://www.facs.org/about-acs/statements/or-attire/ Accessed March 7, 2023.
- Elmously A, Gray KD, Michelassi F, et al. Operating room attire policy and healthcare cost: favoring evidence over action for prevention of surgical site infections. J Am Coll Surg. 2019;228:98–106. PMID: 30359824
- Starr N, Capo-Chichi N, Moore J, et al. Perioperative provider safety in low- and middle-income countries during the COVID-19 pandemic: a call for renewed investments in resources and training. Ann Surg. 2021 ;274:E525–627. PMID: 34225300
- Arciaga Z, Ackerman A, Justice P, et al. Reusable pulse oximetry sensors: a cost-saving quality improvement project. Qual Manag Health Care. 2020;29:35–39. PMID: 31855934
- Dale S, Owrey M, Tomlak A, et al. Reusable pulse oximeters could result in significant cost savings and waste reductions. 5/5/2022 Poster presentation; 2022 House Staff Quality Improvement and Patient Safety Conference; Jefferson’s Health Disparities Summit. https://jdc.jefferson.edu/patientsafetyconference/2022/Quality/9/ Accessed March 7, 2023.