INTRODUCTION
 While patient safety is an established field of study, little attention has been directed to clinician safety. According to the Occupational Health Safety Network, 1 in 5 nonfatal occupational injuries occurs in the health care and social assistance industry, and health care workers experience seven times the national rate of musculoskeletal disorders.1 The most frequently documented causes of injury were patient transport and “slips, trips, and falls.”1
While patient safety is an established field of study, little attention has been directed to clinician safety. According to the Occupational Health Safety Network, 1 in 5 nonfatal occupational injuries occurs in the health care and social assistance industry, and health care workers experience seven times the national rate of musculoskeletal disorders.1 The most frequently documented causes of injury were patient transport and “slips, trips, and falls.”1
Occupational hazards of working in non-operating room anesthesia (NORA) locations, such as interventional radiology suites, electrophysiology and catheterization labs, endoscopy suites, and magnetic resonance imaging suites, have not been studied. Musculoskeletal pain is more common in health care workers working in interventional laboratories and is highest among nonphysician employees.2 This article highlights key occupational hazards anesthesia professionals face in NORA locations and provides suggestions to create a safer working environment.
ROOM SET-UP
Many NORA suites are retrofitted and have insufficient floor area to comfortably accommodate anesthesia machines, automated medication dispensers, and other equipment. Cramped and nonstandard positioning of anesthesia equipment may make clinician movements to access the patient, airway, and injection ports awkward and nonergonomic. The dimmed ambient lighting necessary for fluoroscopic imaging and lack of pathway lighting increases the risk of tripping or sustaining a concussion from a head strike on radiology screens or booms.
Procedures may require the proceduralist and anesthesia professional to change locations often, depending on imaging modality and anatomic site being treated. The anesthesia machine and the drug dispensing system may need to be moved frequently from one side of the room to the other, which poses two specific hazards to anesthesia professionals.
First, anesthesia machines can weigh between 100 and 165 kilograms. While the machines are on wheels, persons moving them need to pay attention to proper body mechanics as well as the presence of cords or other floor obstacles obstructing the wheels. The undue physical strain may be multiplied when different physical layouts increase the frequency of anesthesia machine movement.
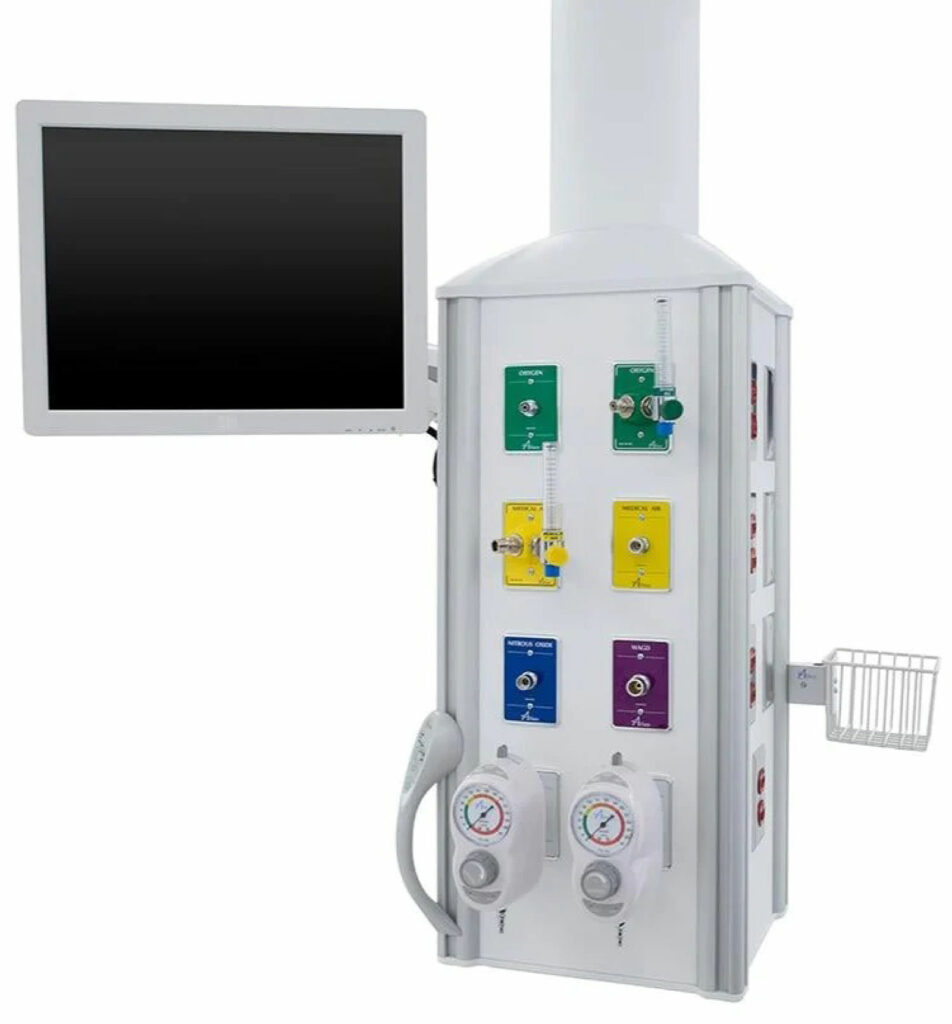
The second hazard that arises from variable placement of the anesthesia machine is from the associated cables, hoses, and lines. Anesthesia machines will have at least three hoses (oxygen, air, and scavenger waste gas) and a power cord. Additional connections can include a vacuum hose for suction, nitrous oxide, computer cords, and data cables (Figure 1). At MD Anderson, at least two anesthesia professionals in the last ten years reported falling after tripping over such cables (Figure 2). There are various solutions to mitigate the risk of falls, including commercially available cable “sleeves” and specially designed mats (Figure 3). These have their own challenges, however, such as the potential for increased bacterial contamination and the mat itself slipping on procedure room floors. One strategy is to place “anti-fatigue” mats over the cables, but this solution relies on having conscientious team members place them at the beginning of every case. Another mitigation strategy is to braid the gas lines (Figure 4). The best solution would be to design procedure rooms with gas and electrical outlets arising from mobile overhead booms so that gas lines can be stored behind the anesthesia machine (Figure 5).

Figure 1. Gas lines, data cable, power cord, suction tubing from anesthesia machine causing trip hazard.
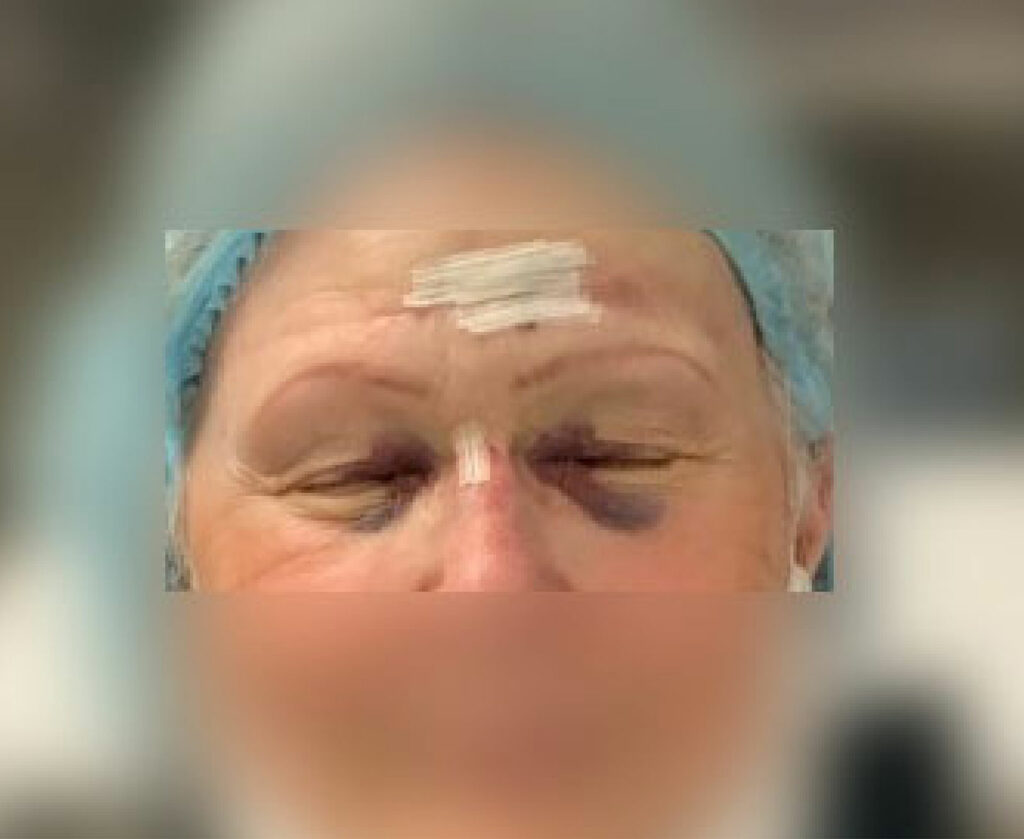
Figure 2: Anesthesia provider injuries to face after tripping over exposed cables. Permission to use from provider granted.
ROOM DESIGN
Anesthesia professionals need to be involved in the design, planning, and construction of new procedure suites. A well-designed room for patient care decreases both floor trip hazards and hanging obstacles like gas lines or electrical cords. This reduces physical strain on clinicians as they reach for needed equipment for patient care.
The proper placement of anesthesia equipment should be prioritized, with related placement of gas lines, suction, electrical outlets, and internet ports. Adequate space needs to be allotted for anesthesia equipment in the correct configuration to the right of the patient’s head (at least for induction and emergence) and for the clinician to have unimpeded access to the patient.3 The ASA Statement on NORA Locations states, “There should be in each location, sufficient space to accommodate necessary equipment and personnel and to allow expeditious access to the patient, anesthesia machine (when present), and monitoring equipment.”3 Weill Cornell Medicine’s Department of Anesthesiology has designated that the minimum space for anesthesia services planned in all new procedure or operating room designs should be 12 feet by 7 feet. This 84-square-foot area should be reserved for the anesthesia machine, medication and equipment cart, IV pole, and chair as a minimum to ensure the anesthesia professional’s ability to safely move around the workspace.4 Many imaging suites are designed solely to accommodate the large imaging equipment and moving patients in and out of the room, with little attention paid to workflow for the technologists, nurses, advanced practice providers, and physicians who care for the patient. Space for patient beds and accessibility for easy transfer of both mobile and immobile patients should be ensured.
Based on the authors’ experience, whenever possible, patients should enter on the side of the room opposite from the anesthesia machine, gas lines, and cords. Rooms should have two doors to allow for easy access of equipment and personnel for regular workflow and in case of emergency. In rooms with only one door, the anesthesia professional and patient’s head should be closest to the door so that personnel arriving to help in an emergency can immediately assist. Gas lines should be piped in close to and behind the anesthesia machine with a dedicated line for waste anesthesia gas disposal (WAGD). The National Fire Protection Agency (NFPA) states that any location where nitrous oxide or halogenated anesthetic gas is intended to be administered should have a WAGD inlet.5 While NFPA guidelines are not legally binding, this is a consensus standard referenced by the Joint Commission.6
In addition to the procedure room, anesthesia professionals should advocate for appropriate preprocedure and postprocedure recovery locations close to the procedure room, and have an expeditious pathway for patient transfer to the intensive care unit. Too often not enough space or thought is given to these important aspects of patient care that can significantly impact safety and efficiency.
PATIENT TRANSFERS
Patient transfers can be particularly awkward in NORA locations due to cramped workspace and lack of equipment to assist in patient transfers, since these areas may have been designed for nonanesthetized patients to move themselves. There are several patient repositioning systems, such as AirTap (Prevalon AirTap, Sage Stryker, Cary, IL) or HoverMatt (HoverTech International, Allentown, PA), designed to help transfer patients who cannot move themselves. While they were designed to enhance patient safety, they also improve clinician safety by limiting musculoskeletal strain.7
INVISIBLE HAZARDS
Chemical hazards such as solvents, adhesives, paints, toxic dust, or, more commonly, waste anesthetic gas are potential dangers to clinicians. However, identifying such exposures might not always be easy since some of the hazards might not be visible (gases) or have an odor. Techniques such as infrared spectrophotometry can be used to identify and quantify a gas leak. Long-term exposure to waste anesthetic gas may affect the antioxidant defense system and likely vital organ function.8 Preventative measures such as daily machine checks, effective scavenging and ventilation systems, proper vaporizer filling, and prompt cleanup of spills should be employed routinely.
In addition, the use of intraprocedural fluoroscopy has increased in both operating rooms and NORA sites. Radiation safety training for anesthesia professionals may be limited. The primary tenet of radiation safety is that exposure dose varies proportionally to the unprotected area of the person and inversely with the square of the distance.9 The small size of many NORA procedure rooms prohibits the anesthesia professional from positioning themselves at sufficient distance from the X-ray tube. Small room size also makes it challenging to add a rolling shield between the anesthesia professional and the radiation source.
Shielding with skirts fixed to the procedural table, rolling lead shields, or protective aprons worn by individual clinicians are essential to reduce exposure area for each person in the room. Eye protection decreases the incidence of cataracts.10 If lead glasses are not available, glass or plastic lenses do provide some reduction of exposure. A circumferential lead apron with thyroid shield protection is essential for anesthesia professionals as they frequently need to turn their backs to the X-ray tube.10 These aprons and thyroid shields should be provided by the institution for the use of clinicians working in a particular procedure suite.
Annual limits of radiation exposure are delineated by the National and/or International Council on Radiation Protection (NRCP, IRCP). Radiation dosimeters should be worn by all clinicians who have occupational exposure. Any individual who is pregnant or has greater than 10% of the recommended annual exposure should have monthly dosimeter assessments.11 Institutions need to distribute dosimeters and monitor results, as well as assess the integrity of all lead shields annually.11 Regular dosimeter assessment can be challenging given the large size of anesthesia departments and the multiple sites that are covered.9
Fortunately, multiple studies have confirmed that radiation exposure for anesthesia providers is generally well below established limits.9 However, when the X-ray tube is adjacent to the anesthesia professional, the clinician’s exposure can be three times greater than the operator due to the anesthesia professional needing to move beyond any rolling shield to administer medications or attend to the patient.12 In addition, the use of novel equipment or techniques in newer NORA procedures may result in inadvertently high radiation exposure, for example those that utilize continuous high-resolution imaging in the neurointerventional and cardiac interventional suites.12
CONCLUSION
As the number of nonoperating room procedures requiring anesthesia increases, anesthesia professionals are exposed to more hazards than in typical operating rooms. While many improvise ad hoc safety measures to avoid injury, there is a role for an organized, multidisciplinary effort to improve clinician safety. This includes being involved in room design, arranging equipment and associated cords/lines in the least obtrusive way, and following a safety checklist that accounts for appropriate equipment and removal or mitigation of physical hazards. Each anesthesia leader can work within their institution’s system to reduce such risks.
Candace Chang, MD, MPH, is an associate professor of anesthesiology at the University of Utah, Salt Lake City, Utah, USA.
Jens Tan, MD, is a professor of anesthesiology at University of Texas MD Anderson Cancer Center, Houston, Texas, USA.
Patricia Fogarty Mack, MD, is a professor of clinical anesthesiology at Weill Cornell Medicine/New York Presbyterian Hospital, New York City, New York, USA.
Diana Anca, MD, is an associate professor of clinical anesthesiology at Weill Cornell Medicine/New York Presbyterian Hospital, New York City, New York, USA.
The authors have no conflicts of interest.
REFERENCES
- Gomaa AE, Tapp LC, Luckhaupt SE, et al. Occupational traumatic injuries among workers in healthcare facilities – United States, 2012–2014; Centers for Disease Control and Prevention; Morbidity and Mortality Weekly Report. 2015;64:405–410. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6415a2.htm Accessed August 3, 2023.
- Orme NM, Rihal CS, Gulati R, et al. Occupational health hazards of working in the interventional laboratory; a multisite case control study of physicians and allied staff. J Am Coll Cardiol. 2015;65:820–826. PMID: 25720626
- Weill Cornell Medicine Department of Anesthesiology Internal Strategic Operating Room and Procedure Suite Development Requirements: 2010–2021.
- Statement on Nonoperating Room Anesthetizing Locations. American Society of Anesthesiologists. 2018. https://www.asahq.org/standards-and-guidelines/statement-on-nonoperating-room-anesthetizing-locations Accessed August 3, 2023.
- National Fire Protection Agency 99 Health Care Facilities Code 2021. 2020;99-49 (page 52). https://www.nfpa.org/codes-and-standards/all-codes-and-standards/list-of-codes-and-standards/detail?code=99 Accessed August 4, 2023.
- https://www.jointcommission.org/standards/standard-faqs/home-care/life-safety-ls/000001288/ Accessed August 5, 2023.
- Wiggermann N, Zhou J, McGann N. Effect of repositioning aids and patient weight on biomechanical stresses when repositioning patients in bed. Hum Factors. 2021 Jun;63:565–577. PMID: 31999485
- Hua HX, Deng HB, Huang XL, et al. Effects of occupational exposure to waste anesthetic gases on oxidative stress and DNA damage. Oxid Med Cell Longev. 2021. PMID: 33542785
- Wang RR, Kumar AH, Tanaka P, Macario A. Occupational radiation exposure of anesthesia providers: a summary of key learning points and resident-led radiation safety projects. Semin Cardiothorac Vasc Anesth. 2017;21:165–171. PMID: 28190371
- Dagal A. Radiation safety for anesthesiologists. Curr Opin Anaesthesiol. 2011;24:445–450. PMID: 21577097
- López PO, Dauer LT, Loose R , et al. Occupational radiological protection in interventional procedures. Ann ICRP. 2018;47:1–118. PMID: 29532669
- Anastasian ZH, Strozyk D, Meyers PM, et al. Radiation exposure of the anesthesiologist in the neurointerventional suite. Anesthesiology. 2011;114:512–20. PMID: 21285864