Downloaded from http://journals.lww.com/anesthesia-analgesia. Accessed on 6/28/2023.
| Reprinted from Anesthesia & Analgesia 137(2):p e8-e11, August 2023. | DOI: 10.1213/ANE.0000000000006539 with permission from the International Research Society. Professional titles and nomenclature were standardized and modified within the text consistent with APSF policy. |
GLOSSARY: APSF = Anesthesia Patient Safety Foundation; ASA = American Society of Anesthesiologists; NORA = nonoperating room anesthesia; OR = operating room
 Nonoperating room anesthesia (NORA) cases are projected to exceed 50% of total anesthesia cases in the near future.1 Although one large-scale study failed to show a difference in mortality between NORA and operating room (OR) settings,2 multiple analyses of data from the American Society of Anesthesiologists (ASA) Closed Claims database have revealed that adverse events occur nearly twice as often in NORA locations as they do in the OR.2-4
Nonoperating room anesthesia (NORA) cases are projected to exceed 50% of total anesthesia cases in the near future.1 Although one large-scale study failed to show a difference in mortality between NORA and operating room (OR) settings,2 multiple analyses of data from the American Society of Anesthesiologists (ASA) Closed Claims database have revealed that adverse events occur nearly twice as often in NORA locations as they do in the OR.2-4
Patient safety in NORA locations may be compromised by problems with ergonomics, location, staffing, teamwork and communication, access to equipment, lack of adequate preoperative optimization, and much more. Other than the ASA Statement on Nonoperating Room Anesthetizing Locations, there have been no widely available recommendations on how to establish, maintain, and standardize safe workflows in NORA.5
In 2022, the Anesthesia Patient Safety Foundation (APSF) convened a multidisciplinary group of experts to organize the annual Stoelting Consensus Conference on “Crucial Patient Safety Issues in Office-Based and Non‑operating Room Anesthesia.” The objective of the conference was to determine consensus recommendations for best practices in NORA around areas of facility and location, equipment and supplies, staffing and teamwork, patient selection, periprocedural care, and quality improvement. A brief summary of our process and results follows.
METHODS
The conference planning committee (the authors) created a conference program to address the unique challenges of NORA (Table 1). Simultaneously, they created the first draft of NORA recommendations, which was then revised and sent to conference speakers and attendees. The recommendations were revised consistent with the feedback provided and presented to breakout groups during the conference. Additional feedback and revisions were then presented to all conference attendees on the last day for discussion and voting. After the meeting, there were further revisions from the conference planning committee, speakers, and participants, which led to consensus-derived recommendations (Supplemental Digital Content 1, Table 1, http://links.lww.com/AA/E369). Ethical considerations, inclusion and exclusion criteria, a list of speakers, and further details of the consensus development process can be found in Supplemental Digital Content 2, Appendix A, http://links.lww.com/AA/E370.
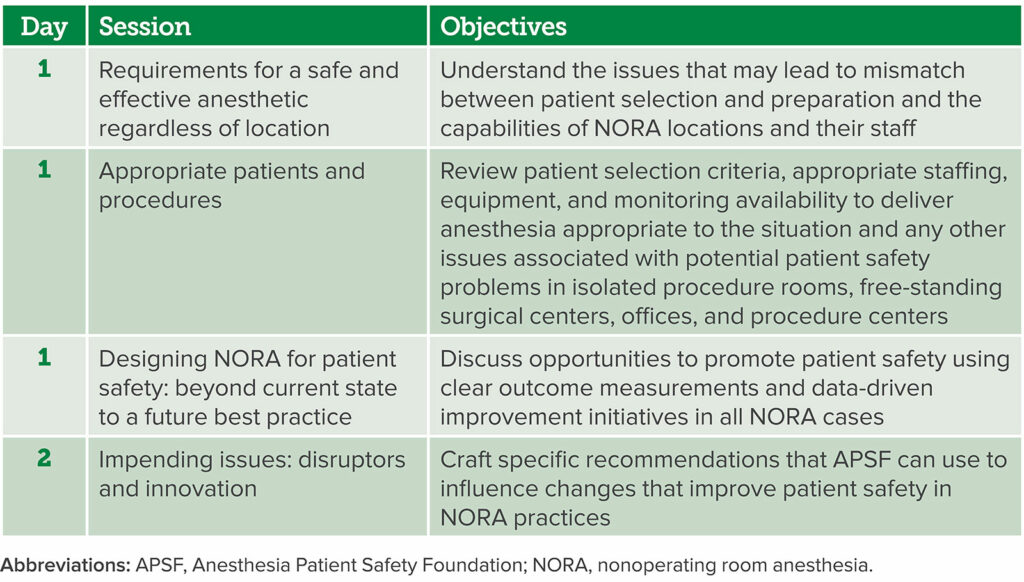
Table 1: 2022 Stoelting Conference Session Description.
Supplemental Table 1: Consensus summary for the safe conduct of anesthetic care in NORA locations.
| FACILITY |
|
| EQUIPMENT, MEDICATIONS, AND SUPPLIES |
|
| STAFF AND TEAMWORK |
|
| PREPROCEDURAL CARE AND PATIENT SELECTION |
|
| INTRAPROCEDURE CARE |
|
| POSTPROCEDURE CARE |
|
| CONTINUOUS QUALITY IMPROVEMENT |
|
| NORA, non-operating room anesthesia; OR, operating room; PACU, post-anesthesia care unit; MH, malignant hyperthermia; MRI, magnetic resonance imaging; ACLS, advanced cardiovascular life support; BLS, basic life support; PALS, pediatric advanced life support; BMI, body mass index; OSA, obstructive sleep apnea; ASA, American Society of Anesthesiologists |
RESULTS
A summary of the 42 recommendations is presented in Supplemental Digital Content 1, Table 1, http://links.lww.com/AA/E369. These recommendations apply to the provision of anesthesia or sedation in NORA locations, which include, but are not limited to, non-OR procedural areas in the inpatient and outpatient settings, including office-based areas like dentistry. These recommendations related to the following domains: facility (9 statements), equipment, medications, and supplies (16 statements), staff and teamwork (4 statements), preprocedure care and patient selection (6 statements), intraprocedure care (2 statements), postprocedure care (3 statements), and continuous quality improvement (2 statements).
DISCUSSION
NORA locations are known to be fraught with patient safety concerns and high stress.6,7 The ASA’s Statement on Nonoperating Room Anesthesia provides guidance on safety considerations for NORA related primarily to facility and equipment issues. The APSF recommendations build on these considerations and provide a template for clinicians to improve teamwork, personnel, and preoperative optimization, which are key patient safety issues in NORA.1,5
The recommendations address many areas that are cited as contributory to safety problems in NORA: facility and location, access to equipment and supplies, teamwork issues, periprocedural care, and quality improvement. While the need for anesthesia services outside of the OR has expanded exponentially in the past decade,8,9 few hospitals are constructed with NORA as a priority. Accordingly, anesthesiology departments have had to retrofit what they need for safe anesthetic care into spaces designed for other purposes. NORA locations may be on different floors than the main OR, or even in different buildings, impeding rapid access to additional personnel and equipment in case of emergency. These consensus recommendations establish clear expectations for the facility, including grouping of procedural areas close to one another and the main OR when possible, establishment of scavenging capabilities and adequate oxygen supply, and need for sufficient electrical outlets and lighting to facilitate safe care.
Many NORA locations do not have sufficient equipment to provide safe anesthetic care, which may contribute to patient safety events.2,10 These recommendations provide standards for facilities to provide emergency airway equipment and capability for rescuing malignant hyperthermia and local anesthetic toxicity, if applicable. The consensus recommendations also provide guidelines for clinician safety; many areas lead clinicians to perform procedures under fluoroscopy. In fact, the anesthesia provider may have radiation exposure equivalent to the proceduralist, and thus, sufficient protection from radiation is required.11
In many procedural suites, the proceduralist and nursing team may not be as familiar with working with anesthesia teams. This lack of familiarity may lead to unfavorable team dynamics and the lack of “belonging,” which can impede patient safety.7 Lack of familiarity—both among team members, and with anesthetic procedures and concerns—as well as poor communication, can lead to adverse events in NORA.1,2,12-14 While the physical space, ergonomics, and location of NORA areas may be more difficult to alter, human factors-related interventions may be easier to implement. Improvements in teamwork and communication are imperative to improving patient safety in these areas and can be facilitated by team training, smaller, more dedicated teams, and shared knowledge about complex cases.
There can be significant production pressure in NORA that can lead to shortcuts. The consensus recommendations advocate for thorough preoperative workup as well as standardized communication before the procedure begins (e.g., formal timeout). Periprocedural monitoring should occur according to standards established by the ASA.15,16 The recommendations also acknowledge the need for both anesthesia and procedural services to review cases for quality of care, with focus on continuous quality improvement.
There have been other recommendations for how to improve anesthetic care in NORA. Notably, Herman et al1 published a recent narrative review of safety issues in NORA and used an engineering framework to provide recommendations for improvement. The recommendations presented here differ as they originate from a multidisciplinary cohort of clinicians and health care representatives with extensive expertise in NORA, who, through an iterative process, have provided consensus statements on approaches for the safe conduct of anesthesia in NORA locations. Indeed, these consensus recommendations supplement existing literature and should be used in concert with previous work.
While most general principles were agreed on by the vast majority of conference attendees and experts, the scope of the recommendations generated the greatest amount of discussion and passion during the development process. There was extensive discussion regarding whether to narrow the scope of the recommendations to inpatient only, or if there should be separate recommendations for ambulatory and office-based anesthesia. This is likely a reflection of the diversity of NORA practice, including inpatient, ambulatory, and office practices. In particular, the example of patient harm in pediatric dental cases generated significant discussion.17 Indeed, patient morbidity and procedural complexity in inpatient locations differ significantly from complexity in outpatient and office-based locations, and there was extensive discussion about whether facility and personnel requirements for inpatient NORA should be required in outpatient or office-based NORA. Some requirements may not be possible—for example, having separate preanesthetic and postanesthetic care areas. The consensus recommendations are the “bare minimum” for safe patient care in NORA and are intended to apply to all NORA locations. Many common patient safety elements apply across the entire NORA population, and the final recommendations were endorsed by clinicians working in inpatient, ambulatory, and office-based NORA.
These recommendations provide a starting point for dedicated anesthesia teams in NORA to improve patient safety, but do not provide strategies for implementation, as these may be specific to both the individual facility and the hospital system. There were several other limitations in the process used to develop the recommendations. First, the content and focus of the conference itself may not fully capture all essential considerations during NORA practice. Second, the final draft of recommendations is dependent on the first draft, which was created by a small group of experts, each of whom may have biases regarding NORA best practices. Third, the planning committee members and speakers were predominantly from academic practices, which may bias the content of the recommendations themselves. Fourth, although nonanesthesiology specialties were represented, they were individual specialists and may not be representative of their entire specialties. Fifth, the conference attendees self-selected for the conference and may not be representative of the general medical community. Finally, while significant effort was put forth to create an inclusive and psychologically safe environment for all participants, it is possible that group discussions may have led to suppression of contrary viewpoints and unexpressed opposition or support. The multiround survey and recommendations review process enabled anonymity to other participants; however, the breakout, discussion, and voting sessions of the conference were likely influenced by the public nature of the discourse and understandable reluctance from participants to share opinions openly.
In summary, these recommendations represent another step toward improving patient safety for NORA patients. They are intended to facilitate the reengineering of health care systems in the best interests of the patient so that medical errors are designed out of the NORA component of the system. NORA cases will continue to comprise an ever-increasing portion of anesthetic practice, and clinicians must continue to remain advocates for patient safety.
| From the *Department of Medical Affairs, GE HealthCare, Patient Care Solutions, Chicago, Illinois; †Department of Anesthesiology and Perioperative Medicine, David Geffen School of Medicine; University of California, Los Angeles, California; ‡Department of Anesthesiology and Perioperative Medicine, Case Western Reserve University School of Medicine, Washington, DC; and §Department of Anesthesiology, The Ohio State University and Wexner Medical Center, Columbus, Ohio.
Accepted for publication April 6, 2023. Funding: This conference was funded by the Anesthesia Patient Safety Foundation. Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (www.anesthesia-analgesia.org). Address correspondence to Richard D. Urman, MD, MBA, Department of Anesthesiology, The Ohio State University and Wexner Medical Center, 410 West 10th Ave, Columbus, OH. Address e-mail to [email protected]. Copyright © 2023 International Anesthesia Research Society |
J.W. Beard, MD, is Chief Medical Officer at GE Healthcare-Patient Care Solutions.
Emily Methangkool, MD, MPH, is an associate professor of clinical anesthesiology at the David Geffen School of Medicine, UCLA, Los Angeles, CA.
Shane Angus, CAA, MSA, is assistant professor at Case Western Reserve University School of Medicine, Washington, D.C.
Daniel J. Cole, MD, is president of the APSF and professor of clinical anesthesiology, UCLA, Los Angeles, CA.
Richard D. Urman, MD, MBA, is the Jay J. Jacoby Professor and Chair of Anesthesiology at The Ohio State University, Columbus, OH.
J.W. Beard is an employee and shareholder of GE HealthCare. Emily Methangkool receives author royalties from UpToDate and honoraria from Edwards LifeSciences (Speakers Bureau and Trial Steering Committee). Daniel J. Cole is the president of the Anesthesia Patient Safety Foundation, which sponsored the conference. R. D. Urman reports fees/funding from AcelRx, Covidien, Pfizer, and Merck. Shane Angus has no conflicts of interest.
This manuscript was handled by Richard C. Prielipp, MD.
REFERENCES
- Herman AD, Jaruzel CB, Lawton S, et al. Morbidity, mortality, and systems safety in non-operating room anaesthesia: a narrative review. Br J Anaesth. 2021;127:729–744. PMID: 34452733
- Metzner J, Posner KL, Domino KB. The risk and safety of anesthesia at remote locations: the US closed claims analysis. Curr Opin Anaesthesiol. 2009;22:502–508. PMID: 19506473
- Robbertze R, Posner KL, Domino KB. Closed claims review of anesthesia for procedures outside the operating room. Curr Opin Anaesthesiol. 2006;19:436–442. PMID: 16829728
- Woodward ZG, Urman RD, Domino KB. Safety of non-operating room anesthesia: a closed claims update. Anesthesiol Clin. 2017;35:569–581. PMID: 29101947
- American Society of Anesthesiologists. Statement on nonoperating room anesthetizing locations. 2018. https://www.asahq.org/standardsand-guidelines/statement-on-nonoperating-
room-anesthetizing-locations. Accessed February 28, 2023. - Alfred MC, Herman AD, Wilson D, et al. Anaesthesia provider perceptions of system safety and critical incidents in non-operating theatre anaesthesia. Br J Anaesth. 2022;128:e262–e264. PMID: 35115155
- Schroeck H, Taenzer AH, Schifferdecker KE. Team factors influence emotions and stress in a non-operating room anaesthetising location. Br J Anaesth. 2021;127:e95–e98. PMID: 34253321
- Nagrebetsky A, Gabriel RA, Dutton RP, Urman RD. Growth of nonoperating room anesthesia care in the United States: a contemporary trends analysis. Anesth Analg. 2017;124:1261–1267. PMID: 27918331
- Louer R, Szeto M, Grasfield R, et al. Trends in pediatric non-operating room anesthesia: data from the National Anesthesia Clinical Outcomes Registry. Paediatr Anaesth. 2023;33:446–453. PMID: 36726283
- Chang B, Kaye AD, Diaz JH, et al. Interventional procedures outside of the operating room: results from the national anesthesia clinical outcomes registry. J Patient Saf. 2018;14:9–16. PMID: 29461406
- Anastasian ZH, Strozyk D, Meyers PM, et al. Radiation exposure of the anesthesiologist in the neurointerventional suite. Anesthesiology. 2011;114:512–520. PMID: 21285864
- Girshin M, Shapiro V, Rhee A, et al. Increased risk of general anesthesia for high-risk patients undergoing magnetic resonance imaging. J Comput Assist Tomogr. 2009;33:312–315. PMID: 19346867
- Webster CS, Mason KP, Shafer SL. Threats to safety during sedation outside of the operating room and the death of Michael Jackson. Curr Opin Anaesthesiol. 2016;29:S36–S47. PMID: 26926333
- Yeh T, Beutler SS, Urman RD. What we can learn from nonoperating room anesthesia registries: analysis of clinical outcomes and closed claims data. Curr Opin Anaesthesiol. 2020;33:527–532. PMID: 32324655
- American Society of Anesthesiologists. Standards for basic anesthetic monitoring. 2020. https://www.asahq.org/standards-and-guidelines/ standards-for-basic-anesthetic-monitoring. Accessed February 28, 2023.
- Apfelbaum JL, Silverstein JH, Chung FF, et al; American Society of Anesthesiologists Task Force on postanesthetic care. Practice guidelines for postanesthetic care: an updated report by the American Society of Anesthesiologists task force on postanesthetic care. Anesthesiology. 2013;118:291–307. PMID: 23364567
- Society for Pediatric Anesthesia. Joint statement on pediatric dental sedation. 2019. https://pedsanesthesia.org/joint-statement-on-pediatric-dental-sedation/ Accessed February 28, 2023.