
From Left to Right: Dr. Mohammed Naguib (Professor of Anesthesiology, Cleveland Clinic Lerner College of Medicine of Case Western Reserve University, Cleveland, Ohio) and Dr. Sorin Brull (Professor of Anesthesiology at Mayo Clinic, Jacksonville, FL) are convening after their 2017 ASA Annual meeting lecture entitled “Assessing and Analyzing the Perceptions of Perioperative Professionals on Neuromuscular Blockade Monitoring and Residual Neuromuscular Blockade.”
At the 2017 ASA Annual meeting in Boston, MA, the APSF Collaborative Panel on Neuromuscular Blockade and Patient Safety presented the results of a survey conducted to assess practitioners’ attitudes towards dosing, monitoring, and reversal of neuromuscular blockade. In addition, the results of four Expert Discussion Groups, which were established to determine the key issues related to perioperative neuromuscular management, were reviewed in the session.
The primary objectives of the project were to identify the key risk factors in anesthetic-related morbidity and mortality associated with use of neuromuscular blocking agents; describe current practices of intraoperative neuromuscular monitoring; assess the incidence of postoperative residual neuromuscular blockade (PRNB); determine factors potentially responsible for the variations in practice with regard to neuromuscular management and monitoring; and describe changes in practice that will decrease residual neuromuscular block and improve patient safety.
Dr. Sorin Brull presented the results of the survey that was distributed to 50,690 anesthesiologists, nurse anesthetists, anesthesiologist assistants, and PACU nurses. The response rate to the survey was 5.7% (2,897 respondents). Sixty-four to 72% of respondents noted that they perceived the incidence of PRNB to be 1–10%. In contrast, numerous studies from medical centers around the world have demonstrated that 30–50% of patients are admitted to the PACU with PRNB.1 Although many of the respondents believed that PRNB was a rare event, 31–43% stated that residual paralysis can have a significant negative effect on patient outcomes. Forty-five percent of respondents reported assessing recovery of neuromuscular function using clinical tests (e.g., 5-second head lift) or a peripheral nerve stimulator. In addition, 8–51% of respondents believed that clinical tests were very or moderately reliable in excluding incomplete neuromuscular recovery. However, significant muscle weakness may still be present (TOF ratios as low as 0.4) when these methods are used.2 While the responses varied between individual providers, 88% of responding anesthesiologists had at least 1 peripheral nerve stimulator per operating room. Only half of the departments had any quantitative monitors (devices which measure and display a train-of-four (TOF) ratio from 0–1.0 or 0–100% in real-time—Figure 1). The primary reason stated for not using quantitative monitoring was the lack of availability of the devices.
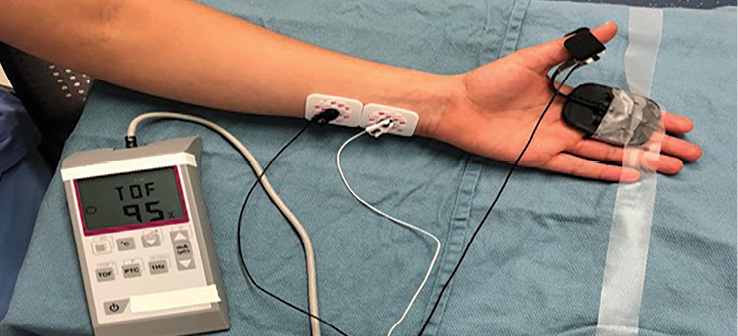
Figure 1: Depicts a quantitative neuromuscular blockade monitor being applied to the subject’s ulnar nerve.
Previous surveys have revealed that routine pharmacologic reversal is used in only 18–32% of practices in the European Union and United States.3 In the APSF survey, the primary reason noted for omitting reversal agents was the timing since last dose. However, clinical investigations have described significant incidence of PRNB nearly 3 hours after receiving even a small dose (25 mg) of rocuronium.4 Furthermore, most respondents stated that the minimal degree of neuromuscular recovery prior to neostigmine reversal was a TOF count of 1–2. Though somewhat controversial, some studies have suggested that it may not be possible to achieve adequate recovery within 1 hour at a TOF count of 1 to 2, and that neostigmine should not be administered until a TOF count of 4 is present.5 Thirty-five percent of respondents noted that an alternative, sugammadex, was either unavailable or its use was restricted by the pharmacy to specific clinical situations or patient populations.
At PACU admission, most clinicians provided information about the muscle relaxant and reversal agent given intraoperatively, but little other data were provided. One-half of the PACU nurses stated that reversal drugs were given to 1–5% of patients after admission. Although clinical tests of muscle strength were performed by 57% of PACU nurses, few (10%) used quantitative, objective monitors (and most nurses receive no training in using these devices). Overall, 75% of responding anesthesia professionals agreed that the American Society of Anesthesiologists (ASA), the American Association of Nurse Anesthetists (AANA) and the American Academy of Anesthesiologist Assistants (AAAA) should collaboratively develop clinical practice guidelines for perioperative monitoring of neuromuscular function.
In addition to the findings of the survey, the conclusions of the four Expert Discussion Groups were presented. The groups were composed of anesthesiologists, nurse anesthetists, PACU nurses, pharmacists, and anesthesiologist assistants. The first group was assigned to address the question of what the most important patient safety issues are related to PRNB. The group noted that there were a number of provider knowledge deficits, including reliance on clinical (head-lift) and subjective (peripheral nerve stimulator-PNS) tests, use of facial muscles instead of the hand for monitoring, the misconception that monitoring is not required if sugammadex is used, and the perception that residual block is rare, and if it occurs, it is not clinically significant. Furthermore, many clinicians do not recognize that PRNB may result in postoperative adverse respiratory events, pneumonia, prolonged PACU length of stay, and unpleasant symptoms of muscle weakness. The second group addressed barriers to the use of subjective and objective monitoring devices. The belief that PNS provide data that indicate adequate recovery and that improper application of devices may fail to detect significant PRNB was discussed. Barriers to the use of objective quantitative monitors include lack of user-friendly devices, cost, unfamiliarity with quantitative technology, lack of appropriate training, and the fact that objective monitors are not considered standard of care. Group three recommended that details about dosing, monitoring, and reversal of neuromuscular blockade should be provided to PACU nurses during transfer of care. Group four addressed education and training requirements which included limitations of clinical tests and subjective (qualitative) evaluation; advantages of quantitative monitoring; proper application (site) of stimulating electrodes; responses of various muscle groups being monitored; importance of documenting baseline TOF ratios; and limitations of neostigmine reversal (depth of block, time to peak onset, ceiling effect). In addition, the group emphasized the importance of documenting competency validation in the use of monitoring at the institutional level.
Dr. Mohamed Naguib concluded by providing recommendations from the Collaborative Panel on perioperative neuromuscular management. These included
- Quantitative (objective) monitoring should be used whenever a neuromuscular blocking drug (NMBD) is administered. These devices should be available at all anesthetizing sites, and information recorded should be incorporated into electronic medical records. Electromyography technology may provide advantages over other categories of monitors.
- During the period of transition to quantitative monitoring , the use of a peripheral nerve stimulator (PNS) in any patient receiving a NMBD is “mandatory.”
- Clinical signs do not guarantee complete resolution of postoperative residual neuromuscular blockade (PRNB), and no longer have a place as the sole determinant of adequate recovery of neuromuscular function.
- Professional organizations should develop practice standards and guidelines detailing how best to monitor and manage perioperative administration of NMBDs.
The session concluded with a summary of the findings of the Collaborative Panel. The Panel noted that a majority of all practitioners think that PRNB is a very significant or moderately significant safety issue that impacts patient outcomes; this validates the purpose of the panel and likely confirms the need for clinical practice guidelines. Equipment availability for anesthesia providers still appears to be an issue, especially with regards to quantitative monitoring. Of note, a significant proportion of practitioners think monitoring is not needed with sugammadex, although no reasoning was provided for this response. Very little information is communicated to PACU nurses regarding intraoperative neuromuscular management, and this information gap must be addressed. In addition to clinical guidelines, educational guidelines are needed for anesthesia providers and PACU nurses. Finally, the survey indicates that 75% of practitioners agree that clinical guidelines are needed, which validates the need for (and acceptance of) guidelines.
Dr. Glenn Murphy is Director of Anesthesiology Research at NorthShore University HealthSystem and is Clinical Professor in the Department of Anesthesia/Critical Care at the University of Chicago.
Dr. Murphy discloses that he is on the advisory board of Merck and has served as a consultant for Merck.
References
- Brull SJ, Kopman AF. Current status of neuromuscular reversal and monitoring: challenges and opportunities. Anesthesiology 2017;126:173–190.
- Murphy GS, Brull SJ. Residual neuromuscular block: lessons unlearned. Part I: definitions, incidence, and adverse physiologic effects of residual neuromuscular block. Anesth Analg 2010;111:120–8.
- Naguib M, Kopman AF, Lien CA, et al. A survey of current management of neuromuscular block in the United States and Europe. Anesth Analg 2010;111:110-9.
- Murphy GS, Szokol JW, Avram MJ, et al. Neostigmine administration after spontaneous recovery to a train-of-four ratio of 0.9 to 1.0: A randomized controlled trial of the effect on neuromuscular and clinical recovery. Anesthesiology2018;128:27-37.
- Kim KS, Cheong MA, Lee HJ, et al. Tactile assessment for the reversibility of rocuronium-induced neuromuscular blockade during propofol or sevoflurane anesthesia. Anesth Analg 2004;99:1080–5.


 Issue PDF
Issue PDF