![]() Dear Q&A,
Dear Q&A,
In our institution we provide anesthesia for major spinal surgery including ALIF. We have always placed a pulse oximeter on the foot for detection of blood flow to the lower extremity. SSEP monitoring is also utilized. Our vascular surgeon questions the validity of the pulse oximeter when there is a loss of signal. Surveying 2 surrounding institutions that also provide anesthesia for ALIF procedures indicated that one requires pulse oximetry, the other relies on SSEP monitoring alone.
As we have provided anesthesia for our patients for ALIF procedures for many years, I can no longer find the original protocol on which we based our monitoring requirements. I sincerely appreciate any information you may provide in this area.
Name Withheld By Request
![]() Dear Reader,
Dear Reader,
The issue here is to define what information we are expecting to receive from each monitoring modality, and how each modality may improve patient safety and outcomes. Pulse oximetry (SpO2) and somatosensory evoked potentials (SSEPs) monitor very different aspects of anterior lumbar interbody fusion (ALIF).
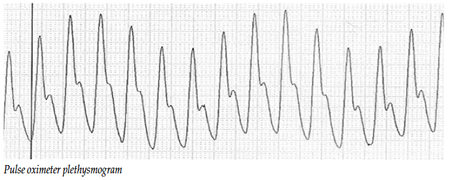
Pulse Oximetry
The waveform displayed on the patient monitor that is associated with the pulse oximeter is called the plethysmogram, and is an indicator of changes in blood volume within the skin that is between apposing parts of the probe. Physiologically and mathematically the plethysmogram is related to the change in blood flow to the skin. Each stroke volume produces a measurable change in the plethysmogram waveform according to the flow delivered to that specific segment of skin. A loss of plethysmographic waveform is indicative of diminished blood flow to the tissues within the probe. There are many factors that play a role in blood flow to the skin of the extremities, including surgical occlusion of vessels, sympathetic nervous activity that increases vascular resistance, patient’s temperature, and the patient’s blood volume.
Surgical issues related to arterial vasculature complications during ALIF are well recognized. According to Fantini et al,1 there is a 2.9% incidence of major vascular complications during anterior lumbar spinal surgery. Vascular injuries occurred during initial spinal exposure or during maintenance of exposure during discectomy, corpectomy, osteotomy, or spinal reconstruction. They described 9 injuries of the common iliac vein and a single aortic injury. If the aorta or common iliac arteries were displaced and mechanically occluded during surgical exposure, the plethysmogram would depict diminished amplitude or no amplitude if the occlusion was complete. Unfortunately, if the surgical injury occurred slightly more distal in the common iliac arteries, the pulse oximeter probes would have to be placed on both right and left toes in order to identify injury to either vessel. In this report the aortic injury resulted in a tear in the terminal aorta at the level of L3-L4. If the tear was significant, the blood flow would exit the aorta as the path of least resistance and no flow would be available to the common iliac arteries, an injury that could have been identified with careful attention to the plethysmogram. A small hole in the aorta might go undetected by the amplitude of the plethysmogram, and tears or other complications of the veins could not be detected by the plethysmogram from pulse oximetry. Faciszewski et al.2 reported earlier studies indicated that injury to the iliac vein or vena cava occurred in 15.6% of cases. Their current study included a single aortic laceration with no common iliac vein lacerations. The literature suggests that venous injury is much more likely than an arterial injury, making the use of the pulse oximeter placed on the toes of questionable value in routine monitoring of ALIF.
Summary of the use of pulse oximeter plethysmogram measured on the toe in ALIF:
- Abrupt changes in the plethysmogram can signal arterial vascular compromise either from bleeding from a tear or from occlusion of an arterial vessel.
- A slow progressive decrease in plethysmogram amplitude may signal growing hypovolemia or hypothermia.
- Peripheral vascular disease in the leg
Somatosensory Evoked Potential
SSEP monitoring for spine procedures is useful for detecting surgical maneuvers that block sensory signals from traveling from the posterior tibial nerve to the cortex via the dorsal column. This may be caused by loss of blood supply to the spinal cord from stretching vasculature or nerve fibers in the cord while straightening the spine. However, they cannot detect an interrupted motor pathway in the anterior spinal cord. Patients with intact SSEPs may awaken in the recovery room unable to move their legs. The ventral and dorsal spinal cord have separate blood supplies with very limited collateral flow; an anterior cord syndrome (paralysis or paresis with some preserved sensory function) is a possible surgical sequela.
Transcranial Motor Evoked Potentials
TcMEP, either electrical (TceMEP) or magnetic (TcmMEP), stimulation of the cerebral cortex, can theoretically monitor the descending motor pathway in the anterior cord. TcMEP alone has been touted as being more sensitive to spinal cord injury intraoperatively than SSEP.
3,4 The combination of the 2 forms of evoked potential monitoring should provide a very powerful tool for intraoperative spinal cord monitoring.3,4 However, there have been reports of patients waking with paraplegia after having intact MEPs intraoperatively.5
Summary
There is probably little justification for monitoring the plethysmogram on one toe during ALIF. If this is the only site used for SpO2 monitoring, the extreme time delay for measuring changes in oxygen saturation in the body, as measured from the toe, probably does not justify its use. SSEPs most likely measure the ability of the spinal cord to conduct sensory information in the dorsal column of the cord, while TcMEP has the potential to measure the function of descending motor tracts in the anterior spinal cord.
References
- Fantini GA, Pappou IP, Girardi FP, Sandhu HS, Cammisa FP. Major vascular injury during anterior lumbar spinal surgery. Spine 2007;32:2751-8.
- Faciszewski T, Winter RB, Lonstein JE, Denis F, Johnson, L. The surgical and medical perioperative complications of anterior spinal fusion surgery in the thoracic and lumbar spine in adults. Spine 1995;20:1592-9.
- MacDonald DB, Al Zayed Z, Khoudeir I, Stigsby B. Monitoring scoliosis surgery with combined multiple pulse transcranial electric motor and cortical somatosensory evoked potentials from the lower and upper extremities. Spine 2003:28;194-203.
- Pelosi I, Lamb J, Grevitt M, Webb JK. Blumhardt LD. Combined monitoring of motor and somatosensory evoked potentials in orthopedic spinal surgery. Clin Neurophysiol 2002;113:1082-91.
- Modi HN, Suh SW, Yang JH, Yoon Ji. False-negative transcranial motor-evoked potentials during scoliosis surgery causing paralysis: a case report with literature review. Spine 2009;34:E896-E900.
The APSF Committee on Technology
The information provided is for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of the APSF. It is not the intention of the APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall the APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information.


 Issue PDF
Issue PDF