Our department was recently contacted to investigate a possible issue in the Datascope AS3000 Anesthesia System (Mindray North America, Mahwah, NJ), which is one of the manufacturers of anesthesia machines that measure breathing circuit pressure only in the expiratory limb. During testing of the system by engineers at GE Healthcare (Madison, WI), it was found that producing a complete occlusion of the expiratory limb at the junction of the disposable breathing circuit and the connection to the absorber during operation of the machine was not detected. Indeed, during the experimental occlusion of the expiratory limb of a circuit, the system indicated zero airway pressure and alarmed for low pressure, when in fact the circuit pressure was independently measured and found to be 63 mmHg.
In order to determine whether or not this was an event isolated to one particular anesthesia machine we asked 2 other users of Datascope AS 3000 systems to perform an experiment in which they placed a piece of plastic over the expiratory portion of the absorber and connected a disposable breathing circuit. In all instances, when the expiratory limb was occluded, the Datascope AS 3000 system’s airway pressure gauge showed zero airway pressure while the reservoir bag, used as a test lung, continued to grow larger.
We understand that other manufacturers’ machines respond in a similar fashion, and we would suggest that, in general, future anesthesia machine designs incorporate airway pressure measurements within the breathing circuit rather than in the absorber behind the one-way valves where the pressure transducer is isolated from the breathing circuit. It is not difficult to imagine a situation where the expiratory limb of a circuit may be either externally or internally occluded, and subsequent build up of fresh gas flow and ventilation with undetected high pressure could result in an untoward event.
George Mychaskiw II, DO, FAAP, FACOP
Professor and Chair, Department of Anesthesiology
Drexel University College of Medicine
245 North 15th Street
Philadelphia, PA 19102
Disclaimer: Dr. Mychaskiw is a consultant to and has received research support from GE Healthcare, Inc.
Reply:
Mindray appreciates the opportunity to respond to Dr Mychaskiw’s concerns regarding the Datascope AS3000 anesthesia machine. We appreciate receiving all comments and concerns regarding our products and take these very seriously. Mindray has patient safety at the forefront of product development and recognizes the value and responsibility of providing education concerning the safe use of anesthesia delivery systems and all medical equipment.
The issue in this case involves the fact that the Datascope AS3000 measures airway pressure in the expiratory limb of the circle breathing system (see Figure 1). When the exhalation limb becomes completely occluded, as in the case of a defective breathing circuit, the pressure in the lungs is not monitored correctly either by the electronic sensor or the circle pressure gauge (see Figure 2). Many manufacturers choose to sense airway pressure only in the expiratory limb. Ironically, Dorsch and Dorsch1 state that placing the sensor on the expiratory side of the circuit has an advantage over placement in the inspiratory side of the circuit, if there is an obstruction in the inspiratory limb.
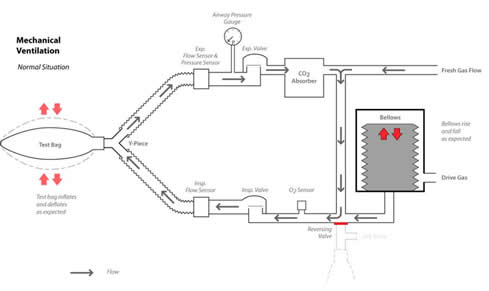
Figure 1. The Datascope AS3000 circle breathing system with inspiratory and exhalation sensors during mechanical ventilation.
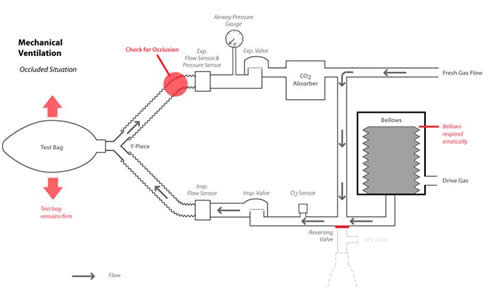
Figure 2. The Datascope AS3000 circle breathing system with expiratory limb occlusion during mechanical ventilation.
We have thoroughly investigated the issue of measuring breathing circuit pressure only in the expiratory limb during complete occlusion of the breathing circuit at the connection to the exhalation port on the absorber. It is pertinent to note that Dr. Mychaskiw’s finding came from bench laboratory studies and that no such no related adverse clinical events have ever been reported on the AS3000.
There are 2 rare but potential clinical occurrences that would fully occlude the expiratory port:
- A manufacturing defect in the breathing circuit, where the occlusion was in place before the pre-use checkout and the beginning of the case.
- A situation where the expiratory limb is placed over an already occluded expiratory port (i.e., the expiratory limb is forced over a piece of rubber or plastic that is large enough to entirely occlude the expiratory port.)
Figure 1 illustrates the normal breathing circuit operation with breathing circuit sensors during mechanical ventilation, while Figure 2 illustrates the effects of occluding the expiratory limb in this mode. Note that the pressure sensor and the pressure gauge are located in the expiratory limb before the expiratory valve. Other anesthesia machine manufacturers have taken this approach to monitoring breathing circuit pressure. Figure 3 illustrates the same breathing circuit in the manual mode with an occlusion in the expiratory limb from a defective breathing circuit. The breathing bag is filled from the fresh gas inflow from the anesthesia machine. As the breathing bag is squeezed, gas is displaced from the bag and travels through the inspiratory limb to the test lung made from another breathing bag. The pressure created in the breathing circuit, and registered on the pressure gauge as the bag is squeezed, is a function of breathing circuit compliance and fresh gas inflow. Because the expiratory limb is occluded, gas that enters the test lung when the bag is squeezed will be trapped in the test lung. The maximum pressure will be determined by the compliance of the “test lung” (breathing bag) and the APL valve in the manual mode, or the pressure limit set on the ventilator while in the mechanical ventilation mode. The pressure on the bag side of the inspiratory valve will be atmospheric until the fresh gas inflow fills the bag again. Meanwhile, the gas volume and pressure in the test lung remains due to the occlusion in the expiratory limb and a closed inspiratory valve disk. Additional gas volume can be added to the test lung by squeezing the breathing bag. If the fresh gas inflow is low or zero, squeezing the bag translocates the gas to the test lung and the breathing bag remains empty. In other words the breathing circuit appears to have a large leak, only the gas is accumulating in the test lung.
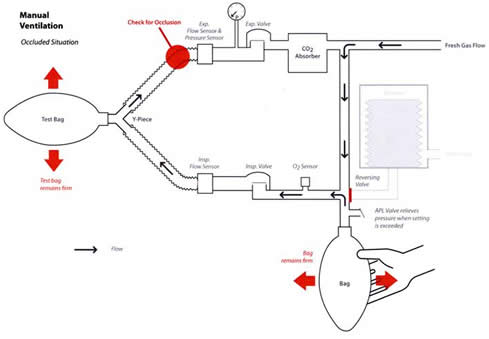
Figure 3. The Datascope AS3000 circle breathing system with expiratory limb occlusion during manual ventilation.
The purpose of the pre-use checkout is to detect potential problems before they impact the patient.
The manufacturers of anesthesia delivery devices all have effective safety concepts and alarm algorithms, but each manufacturer implements these differently. In this regard, the AS3000 is similar to many other anesthesia delivery systems in the United States. A simple, but effective method for detecting events like occlusion of the exhalation valve can be found in the recommended machine checkout procedures and the recommended machine checks between cases in the same room.2,3 For example, it is recommended that the breathing circuit be tested for the ability to effectively deliver positive pressure ventilation before the beginning of each case. A simple leak test by itself is of minimum value, but testing the ability to properly ventilate a test lung is essential and can quickly identify an occluded expiratory limb and other breathing circuit problems. At the start of each case, or before the start of each case to follow in that room, simply removing the breathing bag from the bag arm and connecting it to the patient connection (elbow or wye piece on the disposable circuit) will permit evaluation of the machine’s ability to ventilate the patient. The ventilator can be set to deliver a specific tidal volume to the test lung and verified by the exhaled tidal volume monitor. Observing that the test lung (breathing bag) inflates as the bellows descend, and that the test lung volume decreases during the exhalation phase, along with observing that the measured exhaled volume closely matches the tidal volume set on the ventilator, will provide a level of confidence necessary to proceed with the induction of anesthesia. With the fresh gas inflow set to minimum, or zero, if the bellows fail to rise to the same height during each exhalation then a leak should be suspected, identified, and repaired.
This simple and quick evaluation is the minimum checkout that should be performed between cases. Other published checkout procedures are somewhat more involved, and may evaluate other aspects of the machine, but making sure that a test lung (breathing bag) can be effectively manually or mechanically ventilated indicates that the patient has a high probability of being ventilated.
Specifically in the case of the Datascope AS3000:
As explained above, in the case of an occluded expiratory limb caused by a defective breathing circuit, using the checkout procedure that evaluates the ability of the machine to mechanically ventilate a test lung (the breathing bag) while the fresh gas inflow is set to zero, the occlusion would be easily recognized:
- Visually the breathing bag will inflate further with each delivered breath, because it cannot exhale.
- There will not be an appropriate indication of breathing circuit pressure on the pressure gauge or the monitor as the breath is being delivered.
- The exhaled volume monitor will indicate zero exhaled volume, and it will activate an apnea alarm because the delivered volume measured by the inspiratory flow sensor will not be detected by the expiratory limb flow sensor
- A low pressure alarm will be activated because the expiratory sensor does not detect a rise in pressure associated with an expiratory breath.
- The ventilator bellows will not rise during the exhalation phase and will collapse after delivering a few breaths.
These not so subtle signs of breathing circuit problems should alert the anesthesia provider to troubleshoot the breathing circuit or call for help.
If the semi-automated checkout procedure for the AS3000 is performed while the expiratory limb is occluded, the following abnormal results will be observed:
- Under the Leak Test heading, depressing the oxygen flush button until the PAW is between 25 and 35 cmH2O is between 25 and 35 cmH2O will cause the pressure measured at the circuit to increase abnormally and extraordinarily slowly, possibly never reaching 25 cmH2O as required by the test, providing the first indication of an abnormal condition.
- Under the Safety Valve test, pushing the oxygen flush button to fill the bellows results in an abnormal sound associated with excess pressure venting through an internal relief valve.
- Under the Leak Test, the machine indicates a leak greater than 1 liter/minute because flow is not detected by the expiratory flow sensor.
During this condition the machine disables the ventilator and will not permit the clinician to use mechanical ventilation, until the issue is resolved and the leak test is re-run and passed.
Clearly, the most useful pre-use checkout procedure that works for any anesthesia machine is to set up the breathing circuit, relocate the breathing bag from the bag arm to the breathing circuit patient connection, set all flowmeter to off or minimum, depress the oxygen flush button and fill the bellows, then turn on the ventilator and observe the ventilation of the breathing bag connected to the disposable breathing circuit. Additionally, note the exhaled tidal volume on the monitor and verify that it is close to your set tidal volume. Look for the bellows to re-inflate without loss of volume, indicating that there is not a leak in the breathing circuit.
Mindray is driven to the highest product quality and performance standards. Based on our research and testing of the expiratory limb occlusion, we feel this scenario, which was identified and duplicated in the laboratory, is a rare clinical event that would be readily detected during both the startup testing and during a normal pre-use machine check out. An understanding of specific equipment functionality and conducting the recommended pre-use pressure, leak, and flow tests will assist the anesthesia provider in identifying rare untoward events such as complete occlusion of an expiratory limb of a disposable breathing circuit. We sincerely appreciate the opportunity to interact with the anesthesia community regarding the performance of our products. Our goal is to continually enhance new product performance based upon input from clinicians.
Sincerely,
Scot C. Carriker, MBA
Strategic Marketing Manager
Anesthesia and Ventilation
Richard G. Cipolli
Director
Anesthesia Systems/Hardware Engineering
Mindray North America
Mahwah, NJ 07430
References
- Dorsch JA, Dorsch SE. Understanding anesthesia equipment, 5th ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins, 2008: 246.
- Olympio MA. Dear SIRS: Reader seeks standards for equipment check. APSF Newsletter 2006-07; 21(4):78-9.
- Sub-Committee of ASA Committee of Equipment and Facilities. Recommendations for pre-anesthesia checkout procedures (2008). Available at: http://www.asahq.org/clinical/FINALCheckoutDesignguidelines02-08-2008.pdf. Accessed July 29, 2010.
| The information in this column is provided for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of APSF. It is not the intention of APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information. |


 Issue PDF
Issue PDF