Mounting evidence supports an association between intraoperative hypotension (IOH) and negative postoperative outcomes, most significantly acute kidney injury (AKI), myocardial injury after non-cardiac surgery, and mortality. Recent studies also show associations between IOH and delirium, stroke, and readmissions. The complications associated with IOH have far-reaching consequences for patients and the healthcare system. IOH is a modifiable risk and the approach to it is associated with significant practice variation that we cannot continue to tolerate.
A common request from patients to anesthesia professionals is to “take good care of me.” While we may confidently reply, “I will,” many anesthesia professionals may not be aware of the growing body of data relating intraoperative hypotension (IOH) with patient morbidity and mortality. Mounting evidence supports an association between IOH and negative postoperative outcomes, most significantly acute kidney injury (AKI), myocardial injury after non-cardiac surgery, and mortality.1-9 Recent studies also show associations between IOH and delirium, stroke, and readmissions.1,4,9-11 The complications associated with IOH have far-reaching consequences for patients and the health care system. Acute kidney injury alone is associated with the development of further complications including stroke, myocardial injury, chronic kidney disease, and both in-hospital and one-year mortality, as well as increased length of stay, health care resource utilization, and health care costs.4,12,13 Anesthesia professionals may be unaware of these alarming outcomes especially if they do not receive postoperative outcomes data. Evidence suggests that we must reduce IOH to avoid patient harm.
Intraoperative hypotension can be defined as the blood pressure (BP) below a “safe threshold” leading to hypoperfused organs.1-9 The incidence of IOH varies depending on the definition used, which can involve both a reduction in blood pressure as well as the duration of the reduction. One study assessed the relationship between IOH and the outcomes of AKI and myocardial injury.2 They evaluated both a relative reduction from baseline (i.e., 20% below preoperative BP) and an absolute threshold to define IOH. They found that absolute and relative thresholds had comparable ability to discriminate patients with myocardial or kidney injury from those without, thus suggesting an absolute threshold can be used. Mean arterial pressures (MAP) < 65 mm Hg lasting one minute were associated with an elevated risk of AKI and myocardial injury.2 The risk of developing AKI and myocardial injury increased with a longer duration of IOH.2 This discovery led to the definition of IOH as a MAP < 65 mm Hg for at least one minute.2
A review of available literature on IOH published from 2017 until late 2022, combined with relevant recent studies suggests that the most common definitions of IOH are any MAP < 65 mm Hg1,4,6,10,14-18 or a MAP < 65 mm Hg for at least one minute.8,10,19-26 Using this absolute MAP threshold, IOH is surprisingly common. A recent retrospective observational multicenter study of over 22,000 patients reported 88% of noncardiac surgery patients experienced at least one episode of IOH with a mean duration of 28.2 minutes (Table 1).27 The authors noted significant practice variation in IOH management across the eleven medical centers studied, suggesting differences in provider tolerance of IOH.27
Table 1. Comparison of IOH incidence between four studies
The Centers for Medicare and Medicaid Services (CMS) recognizes a new IOH quality measure, defined as a MAP < 65 mm Hg for greater than 15 minutes, as a criterion in the Merit-Based Incentive Payment System (MIPS).28 A lower overall IOH measure score indicates less time spent under the defined MAP.28 The MIPS score is totaled based on performance measurements of quality, improvement activities, promoting interoperability, and cost. The ePreop31 measure is one of six anesthesia measures that can be submitted for the quality portion of the MIPS score.28,29 The final MIPS score determines the payment adjustment applied to Medicare Part B claims.29 A recent study using the CMS MIPS definition of IOH found the incidence of IOH in community anesthesia practice was 29% in noncardiac procedures.30 This study found varying IOH incidence among clinicians, adding to the body of evidence supporting the reduction of practice variation in IOH management.14,30,31 The authors considered IOH a modifiable risk and suggested pursuing quality improvement initiatives to reduce IOH tolerance.30
A common theme, regardless of the IOH definition, is that more severe degrees of hypotension and a longer cumulative hypotension duration are associated with increased risk of patient morbidity and mortality. Numerous studies show a MAP < 65 mm Hg for extended periods of time, or any period of a MAP ≤ 55 mm Hg, is associated with a greater risk of negative outcomes.1-3,9,16,17,32 Consequently, we simply should minimize the occurrence, severity, and duration of IOH.
Reducing IOH may be challenging when using traditional intermittent oscillometric BP (IOBP) monitoring with an arm cuff. Concerns with IOBP monitoring include delayed or missed detection of BP changes or hypotensive episodes, inaccuracy during hemodynamic extremes, and overestimation of BP during hypotension resulting in more severe episodes than realized.19,33 The potential for missed hypotensive events with IOBP monitoring varies depending on the frequency of measurements chosen or the default setting on the physiologic monitor. One recent study found the most common frequencies of IOBP measurements chosen were every two to five minutes.25 These infrequencies allow for the undetected accumulation of hypotensive minutes between measurements, which may increase the patient’s risk of experiencing harm and may have been minimized with continuous monitoring.15,19,25
New studies support using continuous BP monitoring, touting several benefits over IOBP, including less BP variability,16 improved hemodynamic stability,33 detection of hypotensive episodes missed by IOBP,19 earlier recognition and treatment of IOH,15,25 and overall reductions in IOH.15,19,25,33 Continuous BP monitoring may be accomplished invasively with intra-arterial BP monitoring, but comes with risks, such as infection, nerve damage, thrombus, and pseudoaneurysm.25 Continuous noninvasive BP monitoring with a finger cuff avoids the risks associated with invasive arterial lines while providing comparable MAP values.25,33 Limitations of this technology include additional costs compared to intermittent oscillimetry and the potential for less accuracy in elderly patients or those with atherosclerosis.25,34,35 The noninvasive finger cuff described in recent studies employs volume clamp technology using varying cuff pressure over the finger arteries to maintain a constant volume.25,33 The finger arterial BP is reconstructed to an arterial waveform, allowing for pulse wave analysis, which provides advanced hemodynamic variables (e.g., stroke volume, cardiac output, stroke volume variation) useful for determining the cause of IOH.25 A noninvasive finger cuff can be an appropriate option for continuous BP monitoring when arterial blood samples are not needed during the surgical procedure.33
Clinicians Should Minimize the Occurrence, Severity, and Duration of IOH
Inappropriate fluid and vasopressor management may cause organ hypoperfusion and lead to end organ injury, emphasizing the importance of intentional strategies to avoid these problems.1,3,6,7,12,32 Ariyarathna et al. described a relationship between high vasopressor use and postoperative AKI, independent of IOH.12 Another study suggested that implementation of fluid restriction with their ERAS protocol was associated with significant increases in postoperative hypotension.31 In this study, those with postoperative hypotension also experienced significant IOH and received less total intraoperative fluids. In a recent five-year multicenter retrospective study among Multicenter Perioperative Outcomes Group (MPOG) institutions of over 32,000 abdominal surgery patients, increased AKI rates were observed despite an overall IOH reduction.6 Additionally, they discovered a decline in intraoperative fluid administration and increased vasopressor use, both of which were associated with increased AKI incidence. When crystalloid administration increased from one to ten milliliters per kilogram per hour (mL/kg/hr), they observed a 58% decrease in AKI risk. These poignant findings support the physiologic concept that relying on vasopressors to maintain BP while minimizing fluid administration may diminish already compromised splanchnic and renal perfusion, and potentially cause iatrogenic harm in the forms of ileus, postoperative nausea and vomiting, surgical site infections, and AKI.6,7,12,36
The causes of IOH are multifactorial and include reduced myocardial contractility, vasodilation, hypovolemia, bradycardia, extrinsic compression of heart chambers (e.g., pericardial effusion or pneumothorax), or a “mixed type” explained by multiple hemodynamic alterations.8,18Using monitors that provide advanced hemodynamic variables (e.g., stroke volume, cardiac output, stroke volume variation) may be beneficial for preventing, diagnosing, and treating hypotension.8 Interventions may then be targeted at the root cause of IOH using a goal-directed therapy (GDT) strategy, rather than simply improving the MAP number displayed on the physiologic monitor.
The term GDT may be conceptualized as an umbrella term describing the optimal administration, at the most appropriate time, of fluids, inotropes, and vasopressors using an advanced hemodynamic monitor. Intraoperative interventions within a GDT strategy are aimed at specific endpoints or goals to optimize tissue oxygen delivery and prevent organ hypoperfusion (Figure 1).5,17 The hemodynamic monitor is used to assess patient responsiveness to these interventions. Goal-directed fluid therapy (GDFT) was the first and the most commonly known iteration of the modern GDT concept. The advanced hemodynamic monitor is used to identify preload dependence, aid in the decision to treat with fluid bolus(es) to optimize a patient’s position on the Frank-Starling curve, and then assess for fluid responsiveness.7,17 The 2020 Perioperative Quality Initiative (POQI) consensus statement on fluid management affirmed the safest and most effective method for guiding fluid therapy is to assess for fluid responsiveness.7 Further, optimizing SV with fluids leads to better gastrointestinal perfusion and fewer complications, suggesting the importance of adequate circulating volume and gut perfusion.36 Goal-directed hemodynamic therapy (GDHT), another component of GDT, improved the original GDFT strategy by incorporating the maintenance of MAP to avoid IOH into the protocol.17 GDT strategies incorporating hemodynamic optimization are associated with significant reductions in morbidity and mortality.5,11,32 Figure 1 depicts the modern GDT concept encompassing the components of its first iteration of GDFT, then GDHT, which incorporates the entire picture of perfusion.
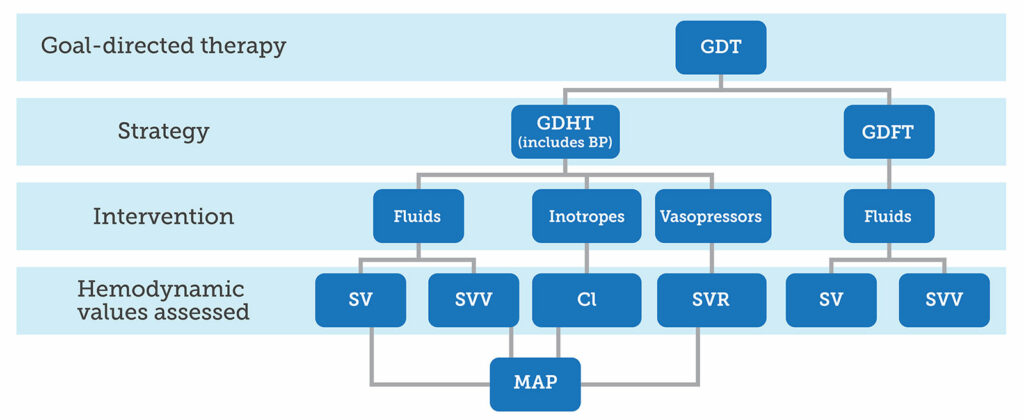
Figure 1. Depiction of the components of a GDT strategy, resulting from a conceptual merging of GDHT and GDFT strategies, along with hemodynamic values guiding specific targeted interventions.
GDT: Goal-directed therapy; GDHT: Goal-directed hemodynamic therapy; GDFT: Goal-directed fluid therapy; SV: Stroke volume;
SVV: Stroke volume variation; CI: Cardiac index; SVR: Systemic vascular resistance; MAP: Mean arterial pressure
Multiple studies using advanced hemodynamic monitoring with a GDT protocol aimed at determining appropriate treatments specific to the cause(s) of IOH and optimizing hemodynamics have shown significant reductions in postoperative complications.5,11,24,32 One study found using a GDHT protocol reduced complications and hospital length of stay (LOS) in low to moderate-risk surgery patients in the landmark FEDORA trial.5 Another study included high-risk patients undergoing major surgery also used a GDHT protocol. This trial resulted in a reduced risk of postoperative organ dysfunction.32 One strength of this later study was the protocolized hemodynamic algorithm used for guiding fluid administration to optimize stroke volume, thereby maintaining intravascular volume and organ perfusion pressure, and protecting against hypoperfusion. In a study of elderly patients undergoing spine surgery, the use of a GDT protocol resulted in less IOH, postoperative nausea and vomiting, and delirium in the GDT group compared to the control group.11 Additionally, other studies using a GDT protocol reported reductions in IOH, further supporting the use of protocols to guide care aimed at targeting IOH causes.21,23,24 GDT has been shown to benefit a range of patients, including low, moderate, and high-risk patients.
IOH is a Serious Public Health Issue
In the 2021 article on the association of IOH and adverse outcomes, IOH was described as a “serious public health issue” that is not permissible for any age group and for any time.1 Due to the size of the population at risk of IOH exposure, the authors urgently recommended future research focusing on IOH prevention. Traditional hemodynamic management relies on reactively treating IOH after it occurs, which is too late as it is already causing organ damage.20 In a 2021 APSF Newsletter article, Sessler alluded to the benefits of predicting IOH with recent technological advancements based on artificial intelligence and machine learning.37 Numerous studies have since been published validating the use of new technology for accurately predicting and reducing IOH.8,20-23,26 One such available technology for predicting the likelihood of impending IOH, along with its root cause, utilizes a parameter called the Hypotension Prediction Index (HPI). HPI provides a unitless number on a scale from zero to 100, indicating the probability that a hypotensive event will occur.38 Using the information provided by the monitor regarding the underlying cause of the impending IOH allows the clinician to intervene appropriately with targeted treatments, thereby avoiding IOH. In a systematic review of randomized controlled trials evaluating the ability of HPI to reduce IOH, authors stated HPI has the potential to reduce the occurrence, duration, and severity of IOH during noncardiac surgery, but emphasized the importance of protocolized adherence to management when using the technology.22 This finding further supports the role of reducing practice variation in reducing the incidence of IOH.
Anesthesia professionals strive to provide excellent anesthetic care for their patients but may be unaware of the potentially detrimental consequences of their hemodynamic management. As Gregory and colleagues powerfully stated, IOH is a “public safety issue,” which must be minimized.1 A step by step approach may help to achieve this goal. Figure 2 lists suggested next steps from the authors of this article incorporating recently published consensus recommendations and best practices from the APSF.39
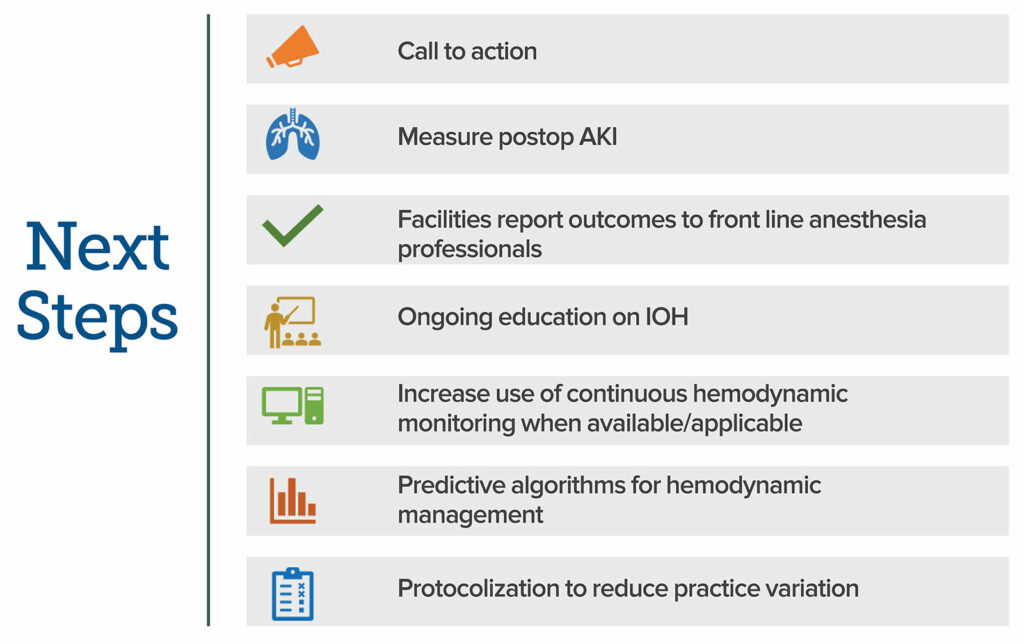
Figure 2. Suggested next steps for anesthesia professionals. The figure incorporates recently published consensus recommendations and best practices from the APSF to help achieve the goal of minimizing IOH and associated patient harm.39
AKI: acute kidney injury; IOH: intraoperative hypotension.
We must acknowledge that IOH is a common problem and raise awareness among colleagues through education and by monitoring and tracking postoperative outcomes such as AKI and myocardial injury after noncardiac surgery. We should monitor continuously with advanced hemodynamic technologies where pertinent to avoid undetected IOH. We must appropriately manage hemodynamics by balancing the circulation to correct the problem (e.g., GDT protocol), rather than treating the number on the monitor with vasopressors. IOH is a modifiable risk that we simply should not continue to tolerate.
Amy Yerdon, DNP, MNA, CRNA, CNE, CHSE, is an assistant professor at the University of Alabama at Birmingham School of Nursing and assistant program director of the Nurse Anesthesia Program, Birmingham, AL.
Matthew Sherrer, MD, MBA, FASA, FAACD, is an associate professor at the University of Alabama at Birmingham, Department of Anesthesiology and Perioperative Medicine, Birmingham, AL.
Desiree Chappell, MSNA, CRNA, FAANA, is the vice president of clinical quality for Northstar Anesthesia, Louisville, KY.
Amy Yerdon, DNP, MNA, CRNA, is a member of the speaker’s bureau for Edwards Lifesciences. D. Matthew Sherrer, MD, MBA, FASA, FAACD, has no conflicts of interest. Desiree Chappell, MSNA, CRNA, FAANA, is the editor in chief and lead anchor, TopMedTalk, a member of the speakers’ bureau for Edwards Lifesciences and Medtronic, and a member of the Provation Advisory Board.
REFERENCES
- Gregory A, Stapelfeldt W, Khanna A, et al. Intraoperative hypotension is associated with adverse clinical outcomes after noncardiac surgery. Anesth Analg. 2021 ;132:1654–1665. PMID: 33177322.
- Salmasi V, Maheshwari K, Yang D, et al. Relationship between intraoperative hypotension, defined by either reduction from baseline or absolute thresholds, and acute kidney and myocardial injury after noncardiac surgery: a retrospective cohort analysis. Anesthesiology. 2017;126:47–65. PMID: 27792044.
- Wesselink E, Kappen T, Torn H, et al. Intraoperative hypotension and the risk of postoperative adverse outcomes: a systematic review. Br J Anaesth. 2018;121:706–721. PMID: 30236233.
- Stapelfeldt WH, Khanna AK, Shaw AD, et al. Association of perioperative hypotension with subsequent greater healthcare resource utilization. J Clin Anesth. 2021;75. PMID: 34536719.
- Calvo-Vecino JM, Ripollés-Melchor J, Mythen MG, et al. Effect of goal-directed haemodynamic therapy on postoperative complications in low–moderate risk surgical patients: a multicentre randomized controlled trial (FEDORA trial). Br J Anaesth. 2018;120:734–744.
- Chiu C, Fong N, Lazzaresch, D, et al. Fluids, vasopressors, and acute kidney injury after major abdominal surgery between 2015 and 2019: A multicentre retrospective analysis. Br J Anaesth. 2022;12:317–326. PMID: 29576114.
- Martin GS, Kaufman DA, Marik PE, et al. Perioperative Quality Initiative (POQI) consensus statement on fundamental concepts in perioperative fluid management: fluid responsiveness and venous capacitance. Perioper Med (Lond). 2020;9:1–12. PMID: 32337020.
- Ranucci M, Barile L, Ambrogi F, Pistuddi V. Surgical and Clinical Outcome Research (SCORE) Group. Discrimination and calibration properties of the hypotension probability indicator during cardiac and vascular surgery. Minerva Anestesiol. 2019;85:724–730. PMID: 30481996.
- Putowski Z, Czajka S, Krzych ŁJ. Association between intraoperative BP drop and clinically significant hypoperfusion in abdominal surgery: a cohort study. J Clin Med. 2021;10(21):5010. PMID: 34768530.
- Maheshwari K, Ahuja S, Khanna AK, et al. Association between perioperative hypotension and delirium in postoperative critically ill patients: a retrospective cohort analysis. Anesth Analg. 2020;130:636–643. PMID: 31725024.
- Zhang N, Liang M, Zhang D, et al. Effect of goal-directed fluid therapy on early cognitive function in elderly patients with spinal stenosis: a case-control study. Int J Surg. 2018;54:201–205. PMID: 29678619.
- Ariyarathna D, Bhonsle A, Nim J, et al. Intraoperative vasopressor use and early postoperative acute kidney injury in elderly patients undergoing elective noncardiac surgery. Ren Fail. 2022;44:648–659. PMID: 35403562.
- French WB, Shah PR, Fatani YI, et al. Mortality and costs associated with acute kidney injury following major elective, non-cardiac surgery. J Clin Anesth. 2022;82:110933. PMID: 35933842.
- Christensen AL, Jacobs E, Maheshwari K, et al. Development and evaluation of a risk-adjusted measure of intraoperative hypotension in patients having nonemergent, noncardiac surgery. Anesth Analg. 2021;133:445–454. PMID: 33264120.
- Maheshwari K, Khanna S, Bajracharya GR, et al. A randomized trial of continuous noninvasive BP monitoring during noncardiac surgery. Anesth Analg. 2018;127:424–431. PMID: 29916861.
- Park S, Lee HC, Jung CW, et al. Intraoperative arterial pressure variability and postoperative acute kidney injury. Clin J Am Soc Nephrol. 2020;15:35–46. PMID: 31888922.
- French WB, Scott M. Fluid and hemodynamics. Anesthesiol Clin. 2022;40:59–71. PMID: 35236583.
- Kouz K, Brockmann L, Timmermann LM, et al. Endotypes of intraoperative hypotension during major abdominal surgery: a retrospective machine learning analysis of an observational cohort study. Br J Anaesth. 2023;130:253–261. PMID: 36526483.
- Kouz K, Weidemann F, Naebian A, et al. Continuous finger-cuff versus intermittent oscillometric arterial pressure monitoring and hypotension during induction of anesthesia and noncardiac surgery: the DETECT Randomized Trial. Anesthesiology. 2023;139:298–308. PMID: 37265355.
- Frassanito L, Giuri PP, Vassalli F, et al. Hypotension Prediction Index with non-invasive continuous arterial pressure waveforms (ClearSight): clinical performance in gynaecologic oncologic surgery. J Clin Monit Comput. 2022;36:1325–1332. PMID: 34618291.
- Grundmann CD, Wischermann JM, Fassbender P, et al. Hemodynamic monitoring with Hypotension Prediction Index versus arterial waveform analysis alone and incidence of perioperative hypotension. Acta Anaesthesiol Scand. 2021;65:1404–1412. PMID: 34322869.
- Li W, Hu Z, Yuan Y, Liu J, Li K. Effect of hypotension prediction index in the prevention of intraoperative hypotension during noncardiac surgery: a systematic review. J Clin Anesth. 2022;83:110981. PMID: 36242978.
- Schneck E, Schulte D, Habig L, et al. Hypotension Prediction Index based protocolized haemodynamic management reduces the incidence and duration of intraoperative hypotension in primary total hip arthroplasty: a single centre feasibility randomised blinded prospective interventional trial. J Clin Monit Comput. 2020;34:1149–1158. PMID: 31784852.
- Wijnberge M, Geerts BF, Hol L, et al. Effect of a machine learning-derived early warning system for intraoperative hypotension vs standard care on depth and duration of intraoperative hypotension during elective noncardiac surgery: the HYPE randomized clinical trial. JAMA. 2020;323:1052–1060. PMID: 32065827.
- Wijnberge M, van der Ster B, Vlaar APJ, et al. The effect of intermittent versus continuous non-invasive BP monitoring on the detection of intraoperative hypotension, a sub-study. J Clin Med. 2022;11:4083. PMID: 35887844.
- Kouz K, Monge García MI, Cercutti E, et al. Intraoperative hypotension when using hypotension prediction index software during major noncardiac surgery: a European multicentre prospective observational registry (EU HYPROTECT). BJA Open. 2023;6:100140. PMID: 37588176.
- Shah N, Mentz G, Kheterpal S. The incidence of intraoperative hypotension in moderate to high risk patients undergoing non-cardiac surgery: a retrospective multicenter observational analysis. J Clin Anesth. 2020;66:1–12. PMID: 32663738.
- Provation. ePreop 31: Intraoperative hypotension (IOH) among non-emergent noncardiac surgical cases. 2020. https://www.provationmedical.com/wp-content/uploads/2022/08/ePreop-Provation_IOH_Specifications.pdf . Accessed March 2024.
- U.S. Centers for Medicare & Medicaid Services (CMS). Traditional MIPS overview. CMS Quality Payment Program. https://qpp.cms.gov/mips/traditional-mips. Accessed January 3, 3024.
- Saasouh W, Christensen AL, Xing F, et al. Incidence of intraoperative hypotension during non-cardiac surgery in community anesthesia practice: a retrospective observational analysis. Perioper Med (Lond). 2023;12:29. Published 2023 Jun 24. PMID: 37355641.
- Anolik RA, Sharif-Askary B, Hompe E, et al. Occurrence of symptomatic hypotension in patients undergoing breast free flaps: is enhanced recovery after surgery to blame? Plast Reconstr Surg. 2020;145:606–616. PMID: 32097291.
- Futier E, Lefrant JY, Guinot PG, et al. Effect of individualized vs standard BP management strategies on postoperative organ dysfunction among high-risk patients undergoing major surgery. JAMA. 2017;318:1346. PMID: 28973220.
- Meidert AS, Nold JS, Hornung R, et al. The impact of continuous non-invasive arterial BP monitoring on BP stability during general anaesthesia in orthopaedic patients: a randomised trial. Eur J Anaesthesiol. 2017;34:716–722. PMID: 28922340.
- Klose P, Lorenzen U, Berndt R, et al. Continuous noninvasive monitoring of arterial pressure using the vascular unloading technique in comparison to the invasive gold standard in elderly comorbid patients: a prospective observational study. Health Sci Rep. 2020;3:e204. PMID: 33204849.
- Maheshwari K, Buddi S, Jian Z, et al. Performance of the Hypotension Prediction Index with non-invasive arterial pressure waveforms in non-cardiac surgical patients. J Clin Monit Comput. 2021;35:71–78. PMID: 31989416.
- Davies SJ, Mythen M. Hemodynamic and intestinal microcirculatory changes in a phenylephrine corrected porcine model of hemorrhage. Anesth Analg. 2021;133:1060–1069. PMID: 33755645.
- Sessler D. Perioperative hypotension. APSF Newsletter. 2021;36:89–94. https://www.apsf.org/article/perioperative-hypotension/ . Accessed March 2024.
- Edwards Lifesciences. Acumen IQ sensor: minimally-invasive predictive decision support. https://www.edwards.com/healthcare-professionals/products-services/predictive-monitoring/acumen-iq-sensor. Accessed March 27, 2023.
- Scott MJ, and the APSF Hemodynamic Instability Writing Group. Perioperative patients with hemodynamic instability: consensus recommendations of the Anesthesia Patient Safety Foundation. Anesth Analg. 2024;138:713–724. PMID: 38153876.


