
![]() Dear Q&A,
Dear Q&A,
We are writing to inquire about the Anesthesia Patient Safety Foundation’s position on peripheral intravenous (IV) catheter placement or noninvasive blood pressure readings taken on the ipsilateral arm in patients who have undergone prior breast surgery. Historically, these patients have been taught to never allow the arm on the affected side to be used for any of these procedures due to concerns of causing lymphedema.
In our institution there are two policies in place reinforcing this practice of no blood draws, IV, or blood pressure cuff use on the affected arm, irrespective of whether the patient has any pre-existing lymphedema. These policies are directed towards our lab technicians who draw blood samples and our allied health care staff who manage the patients with peripheral intravenous catheters. Our institutional process includes placement of laminated warning cards on the front of the patient’s chart and text in their electronic medical record stating no blood pressure cuff readings or IVs on the affected arm.
This issue can cause significant distress and confusion among this patient group and the medical support staff caring for them. Frequently, our anesthesia professionals have to spend a significant amount of time discussing the issue with the patients. This discussion often takes place at a time when the patients are already under duress from their impending surgery. Furthermore, our breast cancer surgeons at our institution do not feel there is any evidence to preclude using the IV or blood pressure cuff on the affected side in the absence of pre-existing lymphedema.
A recent prospective study done at Harvard by Ferguson et al. looked at 632 mastectomy patients with invasive breast cancer over a period of 5 years and did not show any link between IV placement or blood pressure cuff placement and lymphedema.1 The accompanying editorial in the same journal issue also questions the current practice and suggests it is time to abandon old practices.
Our local perioperative quality committee is in the process of revisiting our current policies regarding this issue. It will require significant education and change management amongst many levels of health care providers within our institution. Perhaps a position statement or guideline from our anesthesia governing bodies (using this high-grade evidence) would help support implementing these changes. In light of this recent evidence in the literature, does the APSF know of or have any such policies or guidelines?
Andrew D. Milne, MD, MSc, PEng, FRCPC, Associate Director of Quality and Patient Safety, Dept of Anesthesia, Dalhousie University, Halifax, NS, Canada.
Gregory R. Dobson, MD, FRCPC. Director of Quality and Patient Safety, Dept of Anesthesia, Dalhousie University, Halifax, NS, Canada.
Reference:
- Ferguson CM, Swaroop MN, Horick N, Skolny MN, et al. Impact of ipsilateral blood draws, injections, blood pressure measurements, and air travel on the risk of lymphedema for patients treated for breast cancer. J Clin Oncol 2016;34:691–698.
![]() Dear Dr. Milne,
Dear Dr. Milne,
(From the Editors: The APSF does not have a formal position on this issue. We have recruited Drs. Feldman and Nudelman who are experts on the topic of lymphedema to provide a response to these concerns.)
The risk of developing Breast Cancer Related Lymphedema (BCRL) is a major concern to breast cancer patients. The necessity of adhering to lymphedema risk reduction practices has been questioned and rebutted.1,2 Drs. Milne and Dobson cite the Ferguson Massachusetts General Hospital observational study where patients undergoing treatment for breast cancer between 2005 and 2014 had bilateral arm volume measurements pre and postoperatively using a Perometer. The authors implied that a 10% increase in volume was synonymous with BCRL. Perometry criterion will not diagnose lymphedema in patients with subclinical or mild lymphedema, variable changes, or lymphedema in the hand, breast, or trunk. The conclusion was that venipuncture, injections, blood pressure readings, and air travel “may not” be associated with arm volume increases, a narrow finding and not a definitive conclusion. Debunking the long-standing practices of lymphedema risk reduction practices is not warranted based on the available evidence.
Lymphedema is an inflammatory edema that can occur due to disruption of lymph flow secondary to axillary node dissection or axillary radiation and results in the development of interstitial fibrosis and subcutaneous adipose deposition.3 The lymphedema not only involves the arm but can involve the adjacent trunk and, if conserved, the breast. Other risk factors include cellulitis, BMI >25, and possibly a genomic risk.4 One of the lymphatic system’s main functions is host defense. There is an increased risk of cellulitis in at-risk individuals and individuals with lymphedema. Erysipelas may be the first sign of subclinical lymphedema.5
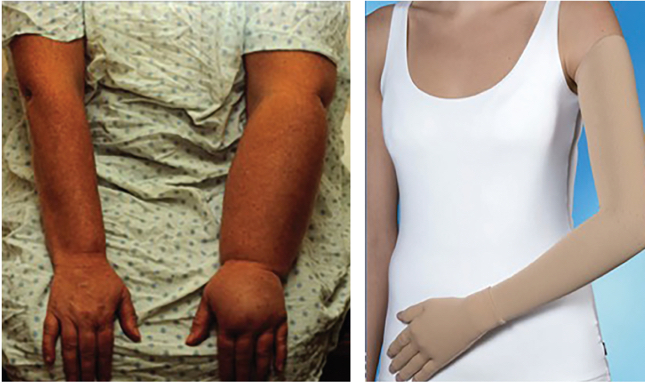
Figures 1A and 1B: Fig. 1A figure depicts a patient with significant lymphedema. Fig 1B depicts a patient with lymphedema wearing a compression garment.
The protective measures are based on the physiological principles of taking care not to do anything that puts the reduced transport capacity at risk or that may increase the lymphatic load.5 Risk reduction practices include avoiding taking blood pressure readings and avoiding invasive procedures in the at-risk extremity. They are also based on years of experience treating at-risk individuals and lymphedema patients.6 The risk reduction practices have been called “burdensome” to patients and clinicians but certainly less of a burden than living with lymphedema.
Although BCRL usually occurs within 3 years postoperatively, there is a lifelong risk of developing lymphedema.7 Lymphedema can be latent or obvious. Lymphedema can cause physical symptoms such as limb heaviness and discomfort and possibly reduce arm function. Diagnostic sensitivity is increased with validated symptom surveys and clinical assessment. Breast conservation and radiation therapy (RT) can be associated with breast lymphedema. Lymphedema creates an economic and emotional burden.8 There is no cure for lymphedema although the condition can be managed by a certified lymphedema therapist through an individualized program of complete decongestive therapy (CDT), wearing compression garments (see Figure 1B, above), and a lymphedema exercise program.
The risk of developing lymphedema is less for SLNB (Sentinel Lymph Node Biopsy) patients than for ALND (Axillary Lymph Node Dissection) patients, but the risk is not zero. A recent publication stated that the overall two-year cumulative incidence of lymphedema was 10% for SNLB + RT compared with 19.3% for ALND-no RT and 30.1% ALND + RT.9
The methodology for detecting lymphedema is far from perfect and there are no universal diagnostic criteria. Lymphedema is a clinical diagnosis, but in an effort to introduce objective criteria, various volume calculations and tissue fluid measurements h
ave been adopted. Lymphedema can be present without volume increases and no objective volume increase has been proven to accurately diagnose lymphedema. There is no agreement on what deviation from baseline measurements represents a significant increase. Various investigators have considered increases ranging from 3% to 10% to be significant.10 Recent direct studies of the lymphatic pump demonstrate that at-risk women have high pump function in both arms, but this objective measurement is not widely available.
Arm volumes can be calculated from the arm measurements, by direct volumetry using the water displacement method or perometry. Perometry is not widely available due to equipment cost and does not measure hand or chest volume. Bioimpedance measurements can also help detect early fluid accumulation, but there is limited access to this technology due to cost.
The National Lymphedema Network (NLN) position paper on Lymphedema Risk Reduction Practices offers a rational approach to risk reduction.11 Ideally, a member of the cancer treatment team or a lymphedema professional should discuss the lymphedema risk factors and the risk reduction practices with the at-risk individual. Recognizing that medical necessity may override the precautions, the NLN position paper summary states that “If possible, avoid punctures such as injections and blood draws,” and “If possible, avoid having blood pressure taken on the at-risk extremity, especially repetitive pumping.” The decision to override the risk reduction practices has to be based on weighing the multiple factors unique to the at-risk individual. Földi and Földi state, “In an emergency, the anesthesiologist’s attention should not be diverted for a single second.”12
Cellulitis is a known lymphedema risk factor.13 The most common bacteria associated with cellulitis is ß-Streptococcus. In patients with lymphedema or at risk, especially patients with a history of cellulitis, perioperative antibacterial treatment will decrease the risk of cellulitis.
The recent article by Ferguson et al. drew a narrow conclusion that in the minority of women who experienced at-risk behaviors, cellulitis was significantly associated with swelling in both the univariate and multivariate analyses, while trauma approached significance in the univariate analysis (p = 0.08). Another prospective study showed venipuncture associated with swelling.13 There are no objective criteria to document immediate damage or triggering of lymphedema by iatrogenic trauma to the at-risk arm/quadrant, but clinical experience and the literature show that infection, venipuncture, and trauma can provoke swelling. Therefore, the prudent medical approach should be to limit unnecessary trauma to the at-risk arm/quadrant. Patients and providers should be thoroughly educated on the risks, the limitations of knowledge, the limitations of diagnosis of the condition, the possible delay in developing lymphedema, and, with this full knowledge, engage in shared decision-making. The current state of knowledge is that some risk is involved with traumatizing an at-risk limb and that no objective criteria exists for diagnosis. If an incurable disease can be prevented by limiting iatrogenic trauma, medical providers should strive to avoid these actions. Traumatizing an at-risk limb has not been proven to never provoke swelling and/or lymphedema, and clinical guidelines should reflect the evidence and clinical experience that indicates risk.
Joseph L. Feldman, MD, is Co-Director of the American Lymphedema Framework Project and founding board member and current President of the Lymphology Association of North America (LANA). He is also the Director of the NorthShore University HealthSystem Lymphedema Treatment Program, Evanston, IL, and Senior Clinician Educator, University of Chicago Pritzker School of Medicine., Chicago, IL
Judith Nudelman, MD, is affiliated with the Providence Community Health Centers and is Clinical Associate Professor of Family Medicine, Brown University, Providence, RI.
The authors report no conflicts of interest for this article.
References:
- Ferguson CM, Swaroop MN, Horick N, et al. Impact of ipsilateral blood draws, injections, blood pressure measurements, and air travel on the risk of lymphedema for patients treated for breast cancer. J of Clinical Oncology 2016;34;691–698.
- Nudelman J. Editorial: Debunking lymphedema risk-reduction behaviors: risky conclusions. Lymphatic Research & Biology 2016;14:124–126.
- Mallick A, Bodenham AR. Disorders of lymph circulation: their relevance to anesthesia and intensive care. Brit J Anaesth 2003;91:265–272.
- Nitti M, Hespe GE, Cuzzone D, et al. In Cheng, M, et al. Principles and practices of lymphedema surgery. Amsterdam: Elsevier, 2016:45–48.
- Damstra RJ, van Steensel MAM, Boomsma JHB, et al. Erysipelas as a first sign of subclinical primary lymphedema: a prospective quantitative scintigraphic study of 40 patients with unilateral erysipelas of the leg. British J of Dermatology 2008;158:1210–1215.
- Földi M, Földi E (eds). Földi’s textbook of lymphology (3rd ed). Amsterdam: Elsevier; 2012. Chapter 3.2.8;212–218.
- Petrek JA, Senie RT, Peters, M, et al. Lymphedema in a cohort of breast carcinoma survivors 29 years after diagnosis. Cancer 2001; 92:1368–1392.
- Fu MR, Ridner SH, Hu SH, et al. Psychosocial impact of lymphedema: a systematic review of the literature 2004 to 2011. Psycho-Oncology 2013;22:1466–1484.
- Miller CL, Specht MC, Skolny M, et al. Risk of lymphedema after mastectomy: potential benefit of applying ACOSOG Z0011 protocol to mastectomy patients. Breast Cancer Res Treat 2014;144:71–77.
- Stout Gergich NL, Pfalzer LA, et al. Preoperative assessment enables the early diagnosis and successful treatment of lymphedema. Cancer 2008;112: 809–2819.
- National Lymphedema Network Position Paper on Risk Reduction Practices. May, 2012. www.lymphnet.org
- Földi M, Földi E (eds): Földi’s textbook of lymphology (3rd ed). Amsterdam: Elsevier; 2012. Chapter 3.2.8;217.
- Clark B, Sitza J, Harlow W. Incidence and risk of arm oedema following treatment for breast cancer: a three-year follow-up study. QJM 2005;98:343–348.
The APSF sometimes receives questions that are not suitable for the Dear SIRS column. This Q and A column allows the APSF to forward these questions to knowledgeable committee members or designated consultants. The information provided is for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of the APSF. It is not the intention of the APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall the APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information.

