Introduction
 Anemia is a frequently encountered condition on the labor floor. Coupled with the potential for massive blood loss in the peripartum period, management of maternal anemia is critical to keeping mothers and their babies safe. Antepartum anemia has been associated with adverse maternal and neonatal outcomes, including preterm labor, miscarriage, growth restriction, cesarean delivery, and intrauterine infection.1,2 Postpartum anemia has been associated with depression, fatigue, and impaired cognition.3
Anemia is a frequently encountered condition on the labor floor. Coupled with the potential for massive blood loss in the peripartum period, management of maternal anemia is critical to keeping mothers and their babies safe. Antepartum anemia has been associated with adverse maternal and neonatal outcomes, including preterm labor, miscarriage, growth restriction, cesarean delivery, and intrauterine infection.1,2 Postpartum anemia has been associated with depression, fatigue, and impaired cognition.3
Iron deficiency is the most common cause of anemia in the peripartum period. Iron replacement therapies have been extensively studied and have been regularly shown to improve hematologic indices,4 but to date, no studies have demonstrated consistent improvement in maternal or neonatal clinical outcomes. This requires us to ask the question: how can we make a meaningful difference in the treatment and management of maternal anemia so as to improve clinical outcomes? How can we as anesthesia professionals partner with our obstetric colleagues to decrease the impact of anemia on childbirth? Here, we highlight our current understanding of maternal anemia, its treatment, management strategies, and future areas for research.
Leveraging the Perioperative Home
Anesthesia professionals are well-equipped to prepare patients for elective, high-blood-loss surgery, and society-level guidelines establish recommendations for the treatment of anemia across the perioperative spectrum.5 Yet, few of these recommendations make specific reference to the pregnant population. Antepartum consultation with an anesthesia professional is often performed for hematologic conditions, such as thrombocytopenia or heritable coagulopathies, but rarely for anemia. By the time an anemic parturient reaches the care of an anesthesia professional, the window for treatment has often closed. Forging partnerships between obstetricians, hematologists, and anesthesia professionals could help address the timely diagnosis and management of maternal IDA.
Scope of the Problem
Anemia is one of the most common medical conditions in pregnancy, affecting nearly one in three pregnant women globally.6 The incidence and degree of iron deficiency anemia (IDA) vary significantly across the course of pregnancy and by population, reflecting the complex interplay of preexisting nutritional deficiencies and iron homeostasis during pregnancy. For example, in low and middle-income countries, IDA may affect up to half of pregnancies, regardless of gestational age.7,8 Even in the developed world, significant racial disparities exist: in one study, African American women had more than three times the rate of IDA as non-Hispanic white women.9 The connection between IDA and poor pregnancy outcomes is not hard to imagine: increases in hemorrhagic shock, cardiovascular failure, peripartum transfusion, and higher rates of infection all accompany maternal anemia.10-12 Additionally, emerging research is beginning to show that peripartum IDA affects not only the mother, but the fetus as well. A large, recent cohort study of over half a million children demonstrated an association between maternal anemia and neurodevelopmental disorders.13 These findings demonstrate the importance of managing this common condition.
Treatment of Iron Deficiency Anemia
The mainstay of IDA treatment in pregnancy is oral iron supplementation. Oral iron supplements are inexpensive, readily available, have simple storage requirements, and have an established safety record. However, oral iron supplementation is often limited by gastrointestinal side effects, such as nausea, dyspepsia, or constipation, which may occur in more than half of patients.14 Studies that have focused on improvements in maternal hematologic parameters (e.g., hemoglobin, ferritin) have found modest improvements in these values with oral iron supplementation.4 However, studies have failed to demonstrate consistent improvement in other maternal or fetal outcomes, such as decreased transfusion requirements, improved recovery, or higher birthweight.
The reasons for this may be severalfold. Diagnosis of iron-deficiency anemia may be delayed in pregnancy: an initial complete blood count (CBC) at a first trimester visit showing anemia may not be followed up with until iron studies can be performed at a subsequent visit, which may be a month or two later. Earlier intervention has clinical plausibility to have a greater impact on maternal and fetal outcomes. Also, studies in this area have generally been smaller, and have lacked consistent methodology.15 Additional large-scale studies with consistent treatment protocols may help establish a clinical benefit for early treatment of maternal iron deficiency.
Safety of Intravenous Iron
Intravenous iron has emerged as an alternative therapy for women with IDA who are intolerant of oral supplementation, have an insufficient response, require rapid correction of deficiency, or who have malabsorptive disorders. With the advent of newer, lower molecular weight iron dextran and non-dextran iron formulations, concerns about anaphylaxis with older parenteral iron formulations have given way to an established safety record in pregnancy.15 Adverse reactions occur less frequently than with oral iron supplementation, and tend to be minor (e.g., skin staining, transient bronchospasm).16 However, because these are parenteral formulations, they require at least one clinic visit, and are substantially more expensive than oral supplementation. Additionally, no guidelines exist about whether fetal heart rate monitoring should occur during infusion of these compounds, and few studies that study these medications in women comment on fetal heart rate monitoring.17 A single case study describes severe fetal bradycardia during infusion of an intravenous iron product (ferric derisomaltose), necessitating emergent cesarean delivery.18 Though a number of studies have established the safety of these compounds, more work needs to be done to establish protocols to ensure maternal and fetal safety during their administration.
Our Role
Reflex iron testing, developed in the pre-anesthesia clinic19 could be used by obstetricians to help diagnose and treat IDA earlier in pregnancy. Obstetricians could refer patients who require IV iron to perioperative anemia clinics, where monitoring and clinical infrastructure currently exist. Promising reductions in perioperative transfusions have been seen with ultra-short-term treatment of anemic cardiac surgery patients.20 Adapting these treatments to pregnant women may help limit the peripartum consequences of anemia. Several institutions have begun treating women in perioperative iron clinics, which presents additional opportunities for clinical research to evaluate the efficacy of this mode of care.
Treatment of prenatal anemia doesn’t always succeed, and hemorrhage can occur unexpectedly in the peripartum setting, even in patients without pre-existing anemia. This requires anesthesia professionals and obstetricians to make collaborative decisions on when it is appropriate to transfuse a peripartum patient. In large studies of nonobstetrical patients, there were no benefits (and an increase in harms) associated with a liberal transfusion strategy.21 Similar findings have been observed in peripartum patients.22 This has led both the American Society of Anesthesiologists and the American Association of Blood Banks to issue recommendations in favor of conservative blood transfusion strategies (i.e., transfusion threshold Hb < 7 vs. <10).23,24 However, there are limits to threshold-based approaches to transfusion. Postpartum hemorrhage often occurs quickly, and requires an anesthesia professional to weigh the benefits of transfusion (i.e., improved tissue perfusion) against the potential risks (e.g., infection, TACO, TRALI). Other perioperative strategies, like the use of blood salvage25 and antifibrinolytic therapy,26 have the potential to decrease the rate of peripartum transfusion even further.
Combining these strategies into comprehensive patient blood management programs can help institutions protocolize the treatment of anemia early in pregnancy that may reduce the number of unnecessary blood transfusions. One such program included clinical pathways for diagnosis, educational materials, laboratory protocols, and standardized iron prescriptions (Figure 1).27 After implementation, the rate of ferritin testing increased tenfold, and the number of transfusions fell by 50%. Similar programs have demonstrated a decrease in rates of anemia at admission and a reduction in the rate of transfusion.28,29 These and other such programs may finally help providers realize the potential of antenatal iron deficiency treatment to improve maternal and fetal outcomes.
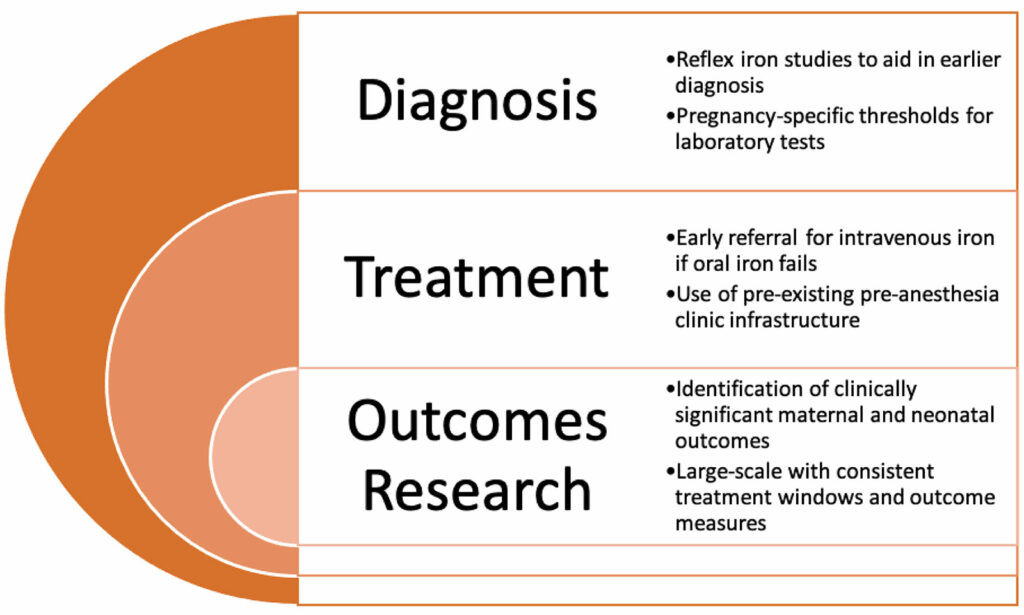
Figure 1: Opportunities for improvement in the diagnosis, treatment, and study of maternal iron deficiency anemia.
Postpartum Anemia
Though relatively poorly studied, postpartum anemia is linked to a number of adverse maternal outcomes including fatigue, depression, and impaired cognition. It should come as no surprise that antenatal anemia and postpartum hemorrhage are significant predictors of postpartum anemia.30 And unlike antepartum IDA, postpartum IDA often occurs suddenly, with a significant loss of iron stores at or around delivery. Treatment of postpartum IDA with oral iron is subject to the same compliance and tolerance issues of antepartum therapy. Because of this, intravenous iron therapy is emerging as a first-line therapy to rapidly restore iron lost at delivery, and has already been shown to elicit a faster and more significant rise in hemoglobin when compared with oral iron therapy.3
Administration of intravenous iron may help avoid some of the morbidity associated with blood transfusion, given evidence that some portion of postpartum transfusions of packed red blood cells may be inappropriate.31,32 Namely, a recent non-inferiority trial of over 500 women with non-symptomatic postpartum anemia were randomized to RBC transfusion or non-intervention.22 This study suggested only slightly lower fatigue scores in transfused women. In the future, direct comparison of transfusion and intravenous iron treatment may help determine optimal treatment of women with postpartum IDA while minimizing unnecessary blood transfusions.
Conclusion
Peripartum IDA remains a common and significant problem, linked to a variety of detrimental clinical outcomes for women and their babies. Yet, iron supplementation has not been consistently shown to ameliorate those outcomes. Opportunities for collaboration between anesthesia professionals, obstetricians, and hematologists are many, and leveraging the tools of the perioperative home may provide avenues for improving maternal and fetal clinical outcomes. In the meantime, awareness of the safety issues surrounding the consequences and treatment of peripartum IDA is paramount for anesthesia professionals responsible for the care of these patients.
Jack M. Peace, MD, is an assistant professor at Temple University.
Jennifer M. Banayan, MD, is an associate professor in the Department of Anesthesiology at Northwestern University Feinberg School of Medicine.
Jack Peace, MD, has no conflicts of interest. Jennifer Banayan, MD, serves as editor of the APSF Newsletter.
References
- Prefumo F, Fichera A, Fratelli N, Sartori E. Fetal anemia: diagnosis and management. Best Pract Res Clin Obstet Gynaecol. 2019;58:2–14.
- Butwick AJ, McDonnell N. Antepartum and postpartum anemia: a narrative review. Int J Obstet Anesth. 2021:102985.
- Sultan P, Bampoe S, Shah R, et al. Oral vs intravenous iron therapy for postpartum anemia: a systematic review and meta-analysis. Am J Obstet Gynecol. 2019;221:19-29.e13.
- Abraha I, Bonacini MI, Montedori A, et al. Oral iron-based interventions for prevention of critical outcomes in pregnancy and postnatal care: an overview and update of systematic reviews. J Evid Based Med. 2019;12:155–166.
- Warner MA, Shore-Lesserson L, Shander A, et al. Perioperative anemia: prevention, diagnosis, and management throughout the spectrum of perioperative care. Anesth Analg. 2020;130:1364–1380.
- Stevens GA, Finucane MM, De-Regil LM, et al. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995-2011: a systematic analysis of population-representative data. Lancet Glob Health. 2013;1:e16-25.
- Black RE, Victora CG, Walker SP, et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. The Lancet. 2013;382:427–451.
- McLean E, Cogswell M, Egli I, et al. Worldwide prevalence of anaemia, WHO Vitamin and Mineral Nutrition Information System, 1993-2005. Public Health Nutr. 2009;12:444–454.
- Mohamed MA, Ahmad T, Macri C, Aly H. Racial disparities in maternal hemoglobin concentrations and pregnancy outcomes. J Perinat Med. 2012;40:141–149.
- Milman N. Anemia—still a major health problem in many parts of the world! Ann Hematol. 2011;90:369–377.
- VanderMeulen H, Strauss R, Lin Y, et al. The contribution of iron deficiency to the risk of peripartum transfusion: a retrospective case control study. BMC Pregnancy Childbirth. 2020;20:196.
- Drukker L, Hants Y, Farkash R, et al. Iron deficiency anemia at admission for labor and delivery is associated with an increased risk for cesarean section and adverse maternal and neonatal outcomes. Transfusion. 2015;55:2799–2806.
- Wiegersma AM, Dalman C, Lee BK, et al. Association of prenatal maternal anemia with neurodevelopmental disorders. JAMA Psychiatry. 2019;76:1294–1304.
- Cancelo-Hidalgo MJ, Castelo-Branco C, Palacios S, et al. Tolerability of different oral iron supplements: a systematic review. Curr Med Res Opin. 2013;29:291–303.
- Qassim A, Grivell RM, Henry A, et al. Intravenous or oral iron for treating iron deficiency anaemia during pregnancy: systematic review and meta-analysis. Med J Aust. 2019.
- Govindappagari S, Burwick RM. Treatment of iron deficiency anemia in pregnancy with intravenous versus oral iron: systematic review and meta-analysis. Am J Perinatol. 2019;36:366–376.
- Jose A, Mahey R, Sharma JB, et al. Comparison of ferric Carboxymaltose and iron sucrose complex for treatment of iron deficiency anemia in pregnancy-randomised controlled trial. BMC Pregnancy Childbirth. 2019;19:54.
- Woodward T, Kay T, Rucklidge M. Fetal bradycardia following maternal administration of low-molecular-weight intravenous iron. Int J Obstet Anesth. 2015;24:196–197.
- Okocha O, Dand H, Avram MJ, Sweitzer B. An effective and efficient testing protocol for diagnosing iron-deficiency anemia preoperatively. Anesthesiology. 2020;133:109–118.
- Spahn DR, Schoenrath F, Spahn GH, et al. Effect of ultra-short-term treatment of patients with iron deficiency or anaemia undergoing cardiac surgery: a prospective randomised trial. Lancet. 2019;393:2201–2212.
- Carson JL, Stanworth SJ, Roubinian N, et al. Transfusion thresholds and other strategies for guiding allogeneic red blood cell transfusion. Cochrane Database Syst Rev. 2016;10:Cd002042.
- Prick BW, Jansen AJ, Steegers EA, et al. Transfusion policy after severe postpartum haemorrhage: a randomised non-inferiority trial. BJOG. 2014;121:1005–1014.
- Management ASoATFoPB. Practice guidelines for perioperative blood management: an updated report by the American Society of Anesthesiologists Task Force on Perioperative Blood Management. Anesthesiology. 2015;122:241–275.
- Carson JL, Guyatt G, Heddle NM, et al. Clinical practice guidelines from the AABB: red blood cell transfusion thresholds and storage. JAMA. 2016;316:2025–2035.
- Goucher H, Wong CA, Patel SK, Toledo P. Cell salvage in obstetrics. Anesth Analg. 2015;121:465–468.
- O’Brien KL, Shainker SA, Lockhart EL. Transfusion management of obstetric hemorrhage. Transfus Med Rev. 2018;32:249–255.
- Abdulrehman J, Lausman A, Tang GH, et al. Development and implementation of a quality improvement toolkit, iron deficiency in pregnancy with maternal iron optimization (IRON MOM): a before-and-after study. PLoS Med. 2019;16:e1002867.
- Flores CJ, Yong A, Knights E, et al. Maternity iron, anaemia and blood management in South Australia: a practice-based evidence for clinical practice improvement. Vox Sang. 2020;115:735–744.
- Guinn NR, Cooter ML, Maisonave Y, et al. How do I develop a process to effectively treat parturients with iron deficiency anemia? Transfusion. 2020;60:2476–2481.
- Butwick AJ, Walsh EM, Kuzniewicz M, et al. Patterns and predictors of severe postpartum anemia after Cesarean section. Transfusion. 2017;57:36–44.
- Butwick AJ, Aleshi P, Fontaine M, et al. Retrospective analysis of transfusion outcomes in pregnant patients at a tertiary obstetric center. Int J Obstet Anesth. 2009;18:302–308.
- Parker J, Thompson J, Stanworth S. A retrospective one-year single-centre survey of obstetric red cell transfusions. Int J Obstet Anesth. 2009;18:309–313.

