Dear Rapid Response:
At our institution, we recently encountered an issue with our HVAC system that left the operating room humidity at approximately 70%. Our operating rooms were closed because of this, and no cases were done while we were out of compliance. A joint statement published by multiple societies in 2015 recommends a relative humidity of 20%-60%.1–7 My question has two parts: (1) how was the upper limit of 60% decided, and (2) what are the real-life dangers of delivering an anesthetic and performing an operation in a setting with a humidity greater than 60% or less than 20%?
Thank you again for your time.
De-An Zhang, MD
Physician Director of Perioperative Services
Shriners For Children Medical Center Pasadena, CA
Dr. Zhang reports no conflicts of interest.
References
- ANSI/ASHRAE/ASHE. Standard 170-2013 ventilation of health care facilities. Atlanta, GA: ASHRAE; 2013.
- NFPA-99 Health Care Facilities Code, 2015 edition: National Fire Protection Association, Quincy MA.
- American Hospital Association. Guidelines for the design and construction of hospitals and outpatient facilities. Chicago, IL: American Hospital Association; 2014.
- American Society for Healthcare Engineering. Briefing for CMS on reduction of low-level humidity, outlines interim waiver process. 2010 http://www.ashe.org/resources/alerts/2012/pdfs/cms-humidity120118.pdf Accessed May 4, 2017.
- Burton, P.E. Lower humidity requirements in surgery and other spaces: why and what it means. 2010. http://www.fhea.org/ORHumReq.pdf. Accessed May 4, 2017.
- Joint Commission Online. CMS adopts ASHRAE-defined range for relative humidity in anesthetizing locations. 2013. http://www.jointcommission.org/assets/1/23/jconline_August_14_131.pdf. Accessed May 4, 2017.
- Association of Surgical Technologists. AST continuing education policies for the CST and CSFA. 2005. Revised July 2016. http://www.ast.org/webdocuments/CEpolicies/.Accessed May 8, 2017.
Reply:
The short answer is that a short-term increase in relative humidity (RH) above the limit of 60% has little impact on the safety of anesthesia care. In the long term, it may affect operating room and instrument sterility.
The current standard on operating room RH levels is set to be between 20% and 60% by the American Society of Heating, Refrigeration, and Air Conditioning (ASHRAE).1 Other standards writing organizations, such as the National Fire Protection Association and the Facility Guidelines Institute, refer to this standard.2,3 Both the Centers for Medicare & Medicaid Services (CMS) and the Joint Commission enforce these limits and require continuous monitoring of operating room temperature and humidity to insure they are in constant compliance when being used for their intended purpose; inspectors typically accept temporary deviations that are resolved within 24 hours.
The instruction for use of some supplies and equipment have even tighter RH limits for storage and/or use; and the facility should also monitor that the operating rooms and storage areas are within these limits. Justifiably, your institution chose not to do cases in operating locations that are outside of the recommended humidity limits.
The rationale for the current 20% lower limit is that Low RH:
- decreases the shelf life of certain supplies, such as biological indicators and chemical indicators used for sterilization monitoring, and electrocardiogram electrodes.
- increases the chance of electrostatic discharge that could harm or interfere with electromedical equipment,4 and potentially spark a fire (this is less of a recent issue because the risk of electrostatic discharge is less with nonflammable anesthetics and antistatic surgical gowns).
The rationale for the current 60% upper limit is that High RH:
- increases the chance of surface mold and mildew growth;
- may increase the risk of wound infections;
- is less comfortable for operating room personnel.
It should be noted that there is sparse experimental evidence documenting the exact levels where these risks increase.5–6 In 2008 the limits were relaxed because the HVAC equipment and fuel costs to meet the previously standardized limits of 35% to 60% were felt to be unnecessarily costly. There remains debate about the safety of the relaxed range (20-60%). A recent review of 10 years of reports to the FDA MAUDE database on electrostatic discharge by medical devices identified low RH (< 30%) as a contributing factor in some of these events.4 The authors also highlighted that instructions for use (IFUs) for most electronic devices recommend a minimum RH of 30%. Furthermore, CMS mandates conformance with the IFU recommendations.4 ASHRAE has yet to revise the standard to be consistent with the manufacturer recommendations.
Relative humidity is the amount of water vapor present in air expressed as a percentage of the amount needed for saturation at the same temperature. RH depends on the temperature and pressure of the system of interest. To acutely decrease the RH level of an operating room, one can either remove water vapor or increase temperature (it is unlikely that one would increase the atmospheric pressure). Cold operating rooms have a higher RH than do warm operating rooms with the same absolute humidity (Figure 1).
Humidity and Temperature from a single OR over 2-months
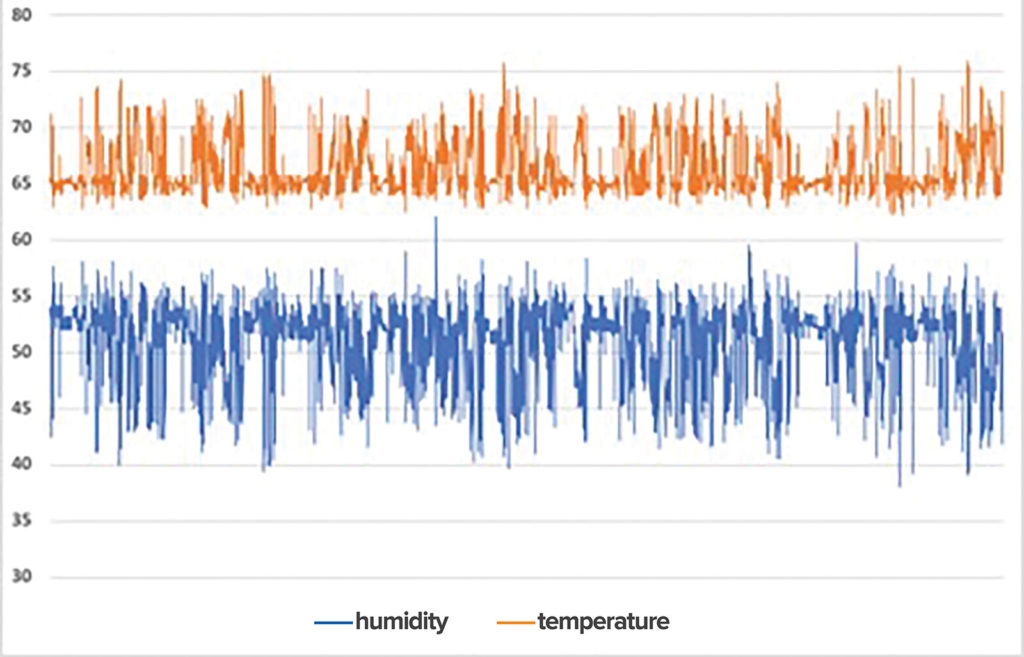
Figure 1: Humidity and temperature from a single OR over 2–months. Note the inverse relationship between temperature and relative humidity.
It is not uncommon for the OR environment to periodically exceed approved limits depending on geography and the type of cases. Northern and western climates often experience low RH, especially in the winter months, and have difficulty raising the RH to the lower limits. Conversely, hospitals in southern and coastal areas experience high ambient RH and systems are designed to remove humidity from the fresh air. While an OR operating at 70 degrees and 50% RH is well within standards, dropping the temperature to 60 degrees for case requirements or staff comfort would cause the RH to exceed the limit.
Whenever the RH of a room extends beyond the approved limits, a risk assessment (RA) of the event should be conducted to insure there is no adverse effect.6 A multidisciplinary group including physicians, surgical staff, Infection Control, OR management, Clinical Engineering, Supply Chain, and Facilities Engineering personnel should participate in the RA. A review of the event, and its impact on environmental or operational conditions should be evaluated and documented. The RA becomes the evidence that the operational breech had no negative safety consequence, and may also be used as a basis for future performance evaluations. This RA documentation also serves as a record of performance when authorities having jurisdiction perform inspections.
Robert G. Loeb, MD, is professor of Anesthesiology, University of Florida, Gainesville, FL.
Dr. Loeb serves as chair of the American Society of Anesthesiologists Committee on Equipment and Facilities. He is on the technical advisory committee of, and has been an invited speaker for, Masimo, Inc.
Bradley S. Pollitt, AIA
Vice President, Facilities
UF Health Shands Hospital, Gainesville
The authors have no conflicts of interest pertaining to this article.
References
- American Society of Heating, Refrigerating and Air-Conditioning Engineers. Standard 170-2017 ventilation of health care facilities. Atlanta, GA: ASHRAE; 2017.
- National Fire Protection Association. NFPA 99 health care facilities code. 2018 ed. Quincy, MA: NFPA; 2018.
- Facility Guidelines Institute. Guidelines for the design and construction of hospitals and outpatient facilities. 2018 ed. Chicago, IL: FGI; 2018.
- Kohani M, Pecht M. Malfunctions of medical devices due to electrostatic occurrences big data analysis of 10 years of the FDA’s reports. IEEE Access. 2018;6:5805-11.
- Wan GH, Chung FF, Tang CS. Long-term surveillance of air quality in medical center operating rooms. Am J Infect Control. 2011;39:302–308.
- American Society of Heating, Refrigerating and Air-Conditioning Engineers. Humidity control events in perioperative care areas. Atlanta, GA: ASHRAE, 2019.
The information provided is for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of APSF. It is not the intention of APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information.


 Issue PDF
Issue PDF