Hospital inpatients represent a large constituency in the health care system—the National Center for Health Statistics estimated a total of 34.7 million discharges in 2005.1 Accordingly, much effort has historically been expended to keep these patients safe during their stay. In its 2001 report “Crossing the quality chasm: A new health system for the 21st century,”2 the Institute of Medicine identified failure to rescue—first defined by Silber in 1992 as hospital deaths following adverse occurrences such as post-surgical complications3—as a primary patient safety target.
The Anesthesia Patient Safety Foundation in “Essential Monitoring Strategies to Detect Clinically Significant Drug-Induced Respiratory Depression in the Postoperative Period” conference summary stated that, “The consensus of conference attendees was that continual electronic monitoring should be utilized for inpatients receiving postoperative opioids.”4
Patient surveillance or continuous monitoring on normally unmonitored wards5 is a departure from the concept of optimized individual care to optimized population care. It is a necessary conceptual paradigm shift for anesthesiologists, but common practice in preventative medicine. This change in approach became necessary because of the documented failure of successfully identifying patients at risk for adverse events.6 Historically, postoperative monitoring was electively used on some patients perceived to be at a particular risk (e.g., patients with sleep apnea), a strategy based on condition monitoring. Equally important, retrospective reviews demonstrated that adverse events are preceded by a period of physiologic instability of 6-8 hours.7,8 Therefore, identification of at-risk patients by spot checks every 6 hours for 10 minutes, which observes vital signs only 5% of the time, begs for improvement. Hence, patient surveillance was introduced with the full understanding that we must do better. While monitoring cannot prevent all physiologic deterioration, it can function as a “patient safety airbag.”
Patient surveillance (PS) is still in its infancy. While there are initial encouraging results,9 there are some common misunderstandings regarding the concepts and many questions remain. Thus, we appreciate the invitation by the APSF to provide more information in this newsletter on our use of patient surveillance since 2007.
In this report we will summarize the Dartmouth experience in the following areas:
- Are alarm settings for heart rate (HR) and oxygen saturation (SpO2) transferable among different surgical populations, or even between surgical and medical populations?
- Were our initially reported results reproducible on other units?9
- Is patient surveillance cost-effective?
- What are the next steps we should implement?
Universal Alarm Settings?
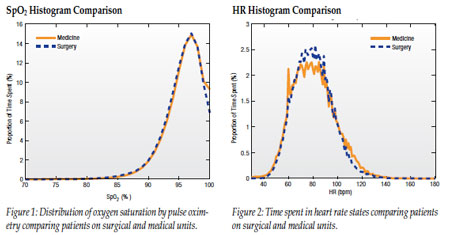
Patients on medical and surgical floors show remarkable similarities regarding their physiological status. Knowledge of these similarities allows the use of similar static alarm settings when introducing patient surveillance systems. Only minor observable differences exist between different surgical and medical wards (Table 1, Figures 1 and 2). Patients spent about 6% of the time with oxygen saturations of <90% and 13% at <93% SpO2. Heart rates were >80 bpm 50% of the time for all units; in medicine 14% of the time was spent >100 bpm, while in surgery the figure was 11%. Mean SpO2 and HR were very similar among surgical units and between surgical and medical wards.
SD: standard deviation. DRG index: diagnosis related group relative weight index (an indicator for severity of illness with higher numbers reflecting higher severity).
With the exception of the pediatric unit, we used the same alarm settings as in our original description (SpO2 <80%, HR <50 or >140),9 with alarm adjustments by nursing staff of ±10%, and further adjustments with a physician order. All medical and surgical patients at Dartmouth have been continuously monitored since 2010.
Results on Other Units
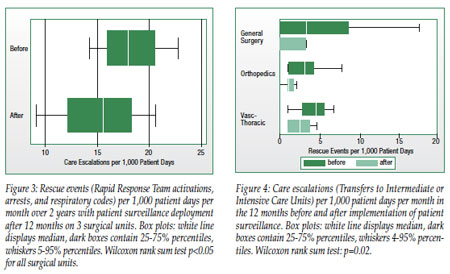
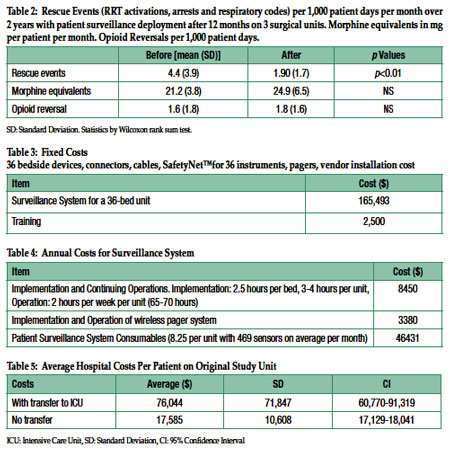
Expansion of patient surveillance using SafetyNet™, Version 2.0.1.3 (Masimo Corp., Irvine, CA) to other units had positive effects on outcomes on all surgical, but not medical units. Figure 3 demonstrates a reduction in average rescue events on the surgical units. This was accompanied by a reduction in care escalations to units of higher intensive care (intermediate and intensive care units), as seen in Figure 4. We use rescue events identified as Rapid Response Team (RRT) activations for cardiopulmonary and respiratory arrests as our main measure of success of early intervention prompted by continuous monitoring. In contrast to measuring escalation of care to intermediate or intensive care units (ICU), the triggering of the rescue team is not dependent on resource availability (ICU beds) or institutional practice patterns. Therefore, we find reduction of rescue events to be a more meaningful measure of early interventions that also makes comparisons among institutions possible.
We have seen institutional reductions in rescue events (0-65%) and in ICU transfers (0-50%). Greater reductions are seen on wards with higher utilization of the system, greater baseline risks, and higher opioid consumption. Use of opioids and number of opioid reversals have not changed (Table 2); opioid antagonists are given for respiratory rates of 5 or less and are administered by nurses per our protocol. However, no patients have suffered irreversible severe brain damage or died since PS was instituted on the original study unit in December of 2007 as a result of respiratory depression from opioids. On surgical units, opioid consumption is greater than on medical units, and the majority of rescue events (>75%) are respiratory in nature.
These results have prompted our institution to mandate continuous monitoring of all patients when they are not being directly observed by a health care provider. If patients refuse such monitoring, they are asked to acknowledge the increased risk using an institutional refusal form.
Cost-Effectiveness
We also conducted a cost-effectiveness analysis for continuous patient monitoring. The costs we used included hardware costs, hospital charges, and fees, and should be considered estimates. These costs are dependent on institutional factors such as volume purchasing and discounts and would vary from institution to institution. The cost-effectiveness depends upon the impact of patient surveillance. For purposes of simplicity, the model presented here is based on reduction of ICU transfers and days spent in ICU. We do not try to estimate other cost opportunities such as medicolegal cost or reduced utilization of rapid response teams, nor did we use a financial penalty for adverse outcomes as is commonly done in cost-effectiveness studies. These potential costs could dramatically increase the cost savings shown in this article. The study of the relationship of quality improvement and cost savings is complex; thus, we are giving a broad overview of cost opportunity without trying to attempt an estimate of various levels of realizable cost reductions.10
Initial implementation costs for a 36-bed unit amounted to $167,993 (Table 3), plus annual costs of $58,261 (Table 4). The cost per patient per hospital episode is $85 for the implementation year and $22 for subsequent years. Averaged hospital costs for a patient on the original study unit without an ICU Transfer were $17,585 vs. $76,044 with an ICU transfer (Table 5).
Prior to introduction of patient surveillance the length of stay (LOS) of a patient with ICU transfer was 24.39 days (7.67 days in ICU plus 16.72 days on the regular floor), afterwards average LOS was19.32 days (5.87 days in ICU plus 13.45 days).
Annual opportunity cost savings11 due to decreased ICU transfer rate amount to $1,479,012 for the initial study unit (as described in reference 9). These opportunity cost savings at DHMC helped address the ICU capacity limitations that were leading to missed opportunities to care for patients in addition to the financial impact. On the other end of the spectrum we had increased cost in a medical unit where the introduction of surveillance was not associated with any change in outcome (implementation and ongoing maintenance costs of the system).
Sensitivity Analysis. Varying the baseline ICU transfer rate demonstrated a greater effect of using patient surveillance as the baseline ICU transfer rate increases. Varying the relationship between ICU transfers with and without PS showed equality when the rate (per 1000 patient days per month) of ICU transfers on the patient surveillance unit is 1.09 (9% higher) that of the non-PS unit.
Cost-effectiveness on other units depend primarily on incidence of adverse events and reduction of event rate. Our thoraco-vascular unit had a higher baseline event rate than the original study unit with a smaller reduction of ICU transfers by about 30% and rescue events by 50%. Because of the higher incidence of transfers, a total of 168 days in the ICU were saved in the 12 months after implementation of PS compared to before, about 10 more days than the original study unit. On some medical units with low event rates and smaller or no change with PS, cost-effectiveness is neutral or even negative when using this opportunity cost-based analysis. Due to high utilization of patient bed capacity (98% at DHMC), standard bed monitoring capacity in all medical and surgical beds allows a flexible floating team of nurses that can provided care for patients on a temporary, as-needed, basis. This flexibility assists our management of our entire inpatient census.
Next Steps
Despite our best efforts, patients still have adverse events requiring rescue interventions and escalations of care. PS as an airbag has worked; we have had no death on the original PS unit since 2007.
We are investigating the use of acoustic respiratory monitoring in addition to our current pulse oximetry network to determine if it has an impact on overall outcome and to identify population groups at risk that would have the greatest benefit from additional monitoring (such as postoperative patients on supplemental oxygen). Early results show that the monitors are relatively well-received by patients. These monitors are better tolerated than our earlier trials in the immediate postoperative phase with chest straps for respiratory rate monitoring or nasal cannulas for end tidal CO2 monitoring, but not as well as finger pulse oximeter probes. Patient comfort and acceptance and minimizing false positive alarms are of great importance when evaluating continuous surveillance devices. In the future we will likely see pulse oximetry surveillance for all, and additional monitoring for some until monitors with the accuracy and comfort of pulse oximetry become available.
Static alarm triggers need to be combined with smart alarms, which have the ability to identify and track patterns associated with clinical deterioration. Our early results are encouraging, while the ability to identify patients likely to deteriorate remains challenging. In a recent roll-out of continuous monitoring in a pediatric unit we have started to use patient dependent alarm settings (age-dependent heart rate alarms). Ideally, systems could be integrated and exchange information between electronic record systems and bedside monitors to allow the seamless calculation of early warning scores based on physiologic, demographic, and comorbidity data, without the need for the user to provide additional input or set alarms and triggers.
Finally, we would like to emphasize the importance of strong collaboration between engineers, nurse, physicians, and technology providers to make patient surveillance work. All stakeholders must be engaged and work together to facilitate the establishment of a safer clinical environment.
Andreas H. Taenzer, MS, MD, is an Associate Professor of Anesthesiology and Pediatrics, and George T. Blike, MD, is Professor of Anesthesiology at The Geisel School of Medicine at Dartmouth, Dartmouth Hitchcock Medical Center. This article was an invited submission in followup to the APSF Consensus Conference on “Essential Monitoring Strategies to Detect Clinically Significant Drug-Induced Respiratory Depression in the Postoperative Period.”
References
- DeFrances CJ, Cullen KA, Kozak LJ. National Hospital Discharge Survey: 2005 annual summary with detailed diagnosis and procedure data. Vital Health Stat 13. 2007;(165):1-209.
- Institute of Medicine: Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academies Press; 2001.
- Silber JH, Williams SV, Krakauer H, Schwartz JS. Hospital and patient characteristics associated with death after surgery. A study of adverse occurrence and failure to rescue. Med Care 1992;30:615-29.
- Weinger MB, Lee LA. No patient shall be harmed by opioid-induced respiratory depression. APSF Newsletter 2011;26:21,26-28. Available at: https://www.apsf.org/newsletters/pdf/fall_2011.pdf. Accessed April 5, 2012.
- Taenzer AH, Pyke JB, McGrath SP. A review of current and emerging approaches to address failure-to-rescue. Anesthesiology 2011;115:421-31.
- Galhotra S, DeVita MA, Simmons RL, Dew MA; Members of the Medical Emergency Response Improvement Team (MERIT) Committee. Mature rapid response system and potentially avoidable cardiopulmonary arrests in hospital. Qual Saf Health Care 2007;16:260-5.
- Buist MD, Jarmolowski E, Burton PR, Bernard SA, Waxman BP, Anderson J. Recognising clinical instability in hospital patients before cardiac arrest or unplanned admission to intensive care. A pilot study in a tertiary-care hospital. Med J Aust 1999;171:22-5.
- Buist M, Bernard S, Nguyen TV, Moore G, Anderson J. Association between clinically abnormal observations and subsequent in-hospital mortality: a prospective study. Resuscitation 2004;62:137-41.
- Taenzer AH, Pyke JB, McGrath SP, Blike GT. Impact of pulse oximetry surveillance on rescue events and intensive care unit transfers: a before-and-after concurrence study. Anesthesiology 2010;112:282-7.
- Rauh SS, Wadsworth EB, Weeks WB, Weinstein JN. The savings illusion—why clinical quality improvement fails to deliver bottom-line results. N Engl J Med 2011;365:e48.
- Weinstein MC, Siegel JE, Gold MR, Kamlet MS, Russell LB. Recommendations of the panel on cost-effectiveness in health and medicine. JAMA 1996;276:1253-8.


 Issue PDF
Issue PDF