To the Editor
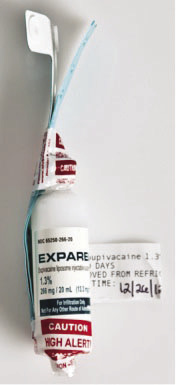
Figure 1. “High-Alert” labeling to prevent look-alike error.
The FDA approved a new formulation of bupivacaine called Exparel™ in Fall 2011. Exparel™ is a liposome injectable suspension of bupivacaine used to infiltrate surgical sites to provide up to 72 hours of local anesthesia. The potential benefits of this new analgesic compared to other analgesics for postsurgical analgesia include that it is not a narcotic, can decrease narcotic use, can decrease the potential for drug interactions, and it has the convenience of being a single administration medication. The decreased use of narcotics may also result in reduced side effects (e.g., dizziness), which may translate to reduced incidences of falls after surgery. However, even though Exparel™ has many innovative benefits, some of the characteristics result in 2 distinct medication safety challenges that should be considered by facilities using the product.
One of the major medication safety challenges related to Exparel™ is its potential for a look-alike error. Since Exparel™ is a liposome suspension, it has a milky-white emulsion appearance much like propofol. Exparel™ and propofol are both used in the operating room and come in glass vials of similar sizes. Therefore, it is very likely that one drug could accidently be selected with the intent of selecting the other. Propofol has a different indication and route of administration than Exparel™ in that it is used for sedation and administered intravenously. Exparel™, however, is infiltrated into the surgical site. Selecting the inappropriate product can result in a sentinel event such as fatality. This is especially true in the event Exparel™ is administered intravenously.
To prevent a look-alike error related to Exparel™ and propofol, our facility has implemented several processes. First, the 2 agents are stored in 2 separate locations within the pharmacy. This is easy to do as Exparel™ is stored in the refrigerator and propofol is stored at room temperature on IV shelves and in medication carousels.
However, Exparel™ is stable at room temperature for 30 days. Due to Exparel’s stability at room temperature, it is commonly filled in medication dispensing units (e.g., Pyxis units) within the surgical area. Therefore, both propofol and Exparel™ can be retrieved from the same medication dispensing unit. The second safety measure we have in place is to avoid accidently pulling the wrong medication from the dispensing unit. A Pyxis Clinical Alert has been programmed within the Pyxis unit that states, “Confirm removing of Exparel™ for SQ infiltration at surgical site.” This alert brings to the user’s attention that they are selecting Exparel™ and reminds them of the appropriate route of administration. There is still potential for a user to select the wrong drug if both agents are stored in an open access drawer (e.g., a matrix drawer within a Pyxis unit). To avoid the potential for error, Exparel™ is not stored in these drawers and can only be accessed in a carousel (pie) drawer within the Pyxis unit so the user only has access to Exparel™ when pulling the medication from the Pyxis unit. Another safety measure that was implemented to help differentiate Exparel vials from propofol was the addition of a “High Alert” shrink wrap seal covering the cap of the Exparel™. Finally, there is always the potential for user error. To help counteract user error, we educated pharmacy and surgical staff on Exparel™ regarding the potential for a look-alike error, the consequences of an error, and the procedures to follow to avoid an error.
The second major medication safety challenge involving Exparel™ is that it is a high-risk medication. Exparel™ is a long-acting formulation of bupivacaine which results in plasma levels for a duration of 96 hours. No formulation of bupivacaine should be administered within 96 hours of Exparel™, due to potential risks of a significant overdose and systemic side effects. However, it can be difficult to identify whether a patient was given Exparel™, and when it was administered. Often, patients receive Exparel™ during a same-day procedure and have no way to identify that they received this agent. This is an issue since no one outside the surgical facility will be aware that ExparelTM was administered to the patient and the patient could potentially receive additional bupivacaine if he/she receives medical care within the 96-hour period.
To properly identify each patient who has received Exparel™ in a standardized process is not an easy task. Our facility wanted a “fool-proof” process that would identify each patient that received Exparel™ when it was administered. Due to the complexity of many patients quickly leaving the surgical facility, we needed an identifier that would follow the patient once they left our facility. We decided to utilize the Exparel™ wristband produced by the manufacturer of Exparel™. The wristband has the Exparel™ name preprinted on the band and a location to write in the date/time it was administered. This wristband is placed on the same arm as the patient’s identification band. This helps surgical and post-op staff identify that the patient received Exparel™ and prompts them to educate the patient on the necessity of wearing the wristband for a full 96 hours after their procedure. It is our process that anyone wearing an Exparel™ wristband in our facility is verbally educated on the medication, instructed to wear the wristband for a full 96 hours, and informed on the potential adverse events that could occur if additional bupivacaine is administered during the 96 hour time period. Patients also receive standardized discharge instructions with this same information.
The only way the wristband procedure can be a success is if it is a reliable process that can be done on every patient who receives Exparel™. To assure a wristband is placed on each patient that receives Exparel™, the vials are specially processed before ever leaving the pharmacy. Each vial has an Exparel™ wristband attached to it with 2 “High Risk” shrink-wrap labels. This process assures a wristband is readily available at each administration and performs a double duty of further identifying the vial contains Exparel™ to prevent a look-alike error (see Figure 1). In the event that a wristband would fall off the vial or a wristband needed to be removed temporarily from the patient, the surgical staff has access to additional Exparel™ wristbands that are stored in the automated dispensing device.
Exparel™ is a novel anesthetic that provides long-acting pain relief and may help decrease the use of narcotics. However, its use is not without caution. Exparel™ has the potential to cause sentinel events via look-alike medication errors, especially with propofol. It is also a high risk medication due to plasma levels of bupivacaine persisting for 96 hours. Therefore, no additional bupivacaine is to be administered during this time period without significant risk of adverse events. To safely use Exparel™ within our facility, many standardized processes were implemented prior to its use in the operating room to avoid medication safety events.
Elizabeth Gripentrog, PharmD Sanford USD Medical Center, Sioux Falls, SD
Megan Maddox, PharmD, BCPS Medication Safety Officer Sanford USD Medical Center, Sioux Falls, SD
Rhonda Hammerquist, PharmD, BCPS Medication Safety Officer Sanford USD Medical Center, Sioux Falls, SD
All authors have no disclosures to report.


 Issue PDF
Issue PDF