In 2012, Congress passed the Food and Drug Administration Safety and Innovation Act (FDASIA) granting the FDA more authority regarding drug shortages.1 The legislation, in part, mandated that manufacturers notify the FDA of potential discontinuations or interruption in the manufacture of drugs used to prevent or treat serious or life-threatening diseases. Within the first year after passage of the legislation the number of notifications to the FDA increased 6-fold. This bill also gave the FDA authority to expedite reviews and inspections to help mitigate drug shortages.
In 2013 the FDA issued its Strategic Plan for Preventing and Mitigating Drug Shortages.2 It had 2 goals. The first was aimed at further mitigation of drug shortages. The second was to develop long-term prevention strategies. The former targeted improvements to the FDA’s current mitigation activities for existing or imminent shortages. The latter concentrated on the root causes of shortages to better understand and eventually anticipate future drug shortages.
These measures have had an effect on the number of reported drug shortages. According to data from the University of Utah Drug Information Service,3 the number of new drug shortages per year from 2005 to 2011 had increased more than 5-fold, culminating in a maximum number of new drug shortages being reported in 2011. Since that time, there has been a downward trend that has resulted in more than a 50% reduction in new drug shortages through 2013. A review of these drug shortages show that certain classes of medications, sterile injectable drugs in particular, comprise the majority and have caused specialties such as anesthesiology to be especially vulnerable. And, while there has been a decrease in the number of new drug shortages, the number of existing drug shortages from 2012 to 2013 has increased slightly (Figure 1).3-5
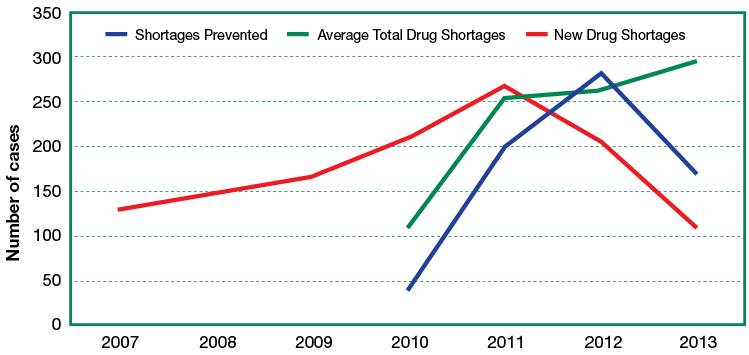
Figure 1: Drug Shortages in US: Prevented Shortages2 vs. New Shortages5 vs. Average Total Shortages.5
Drug shortages particularly affect anesthesiologists. The Government Accountability Office reports that anesthetics and central nervous system drugs account for 17% of all shortages and are among the classes of drugs that routinely experience the highest frequency of shortages.6 In a survey conducted by the American Society of Anesthesiologists in March 2012, 97.6% of responding anesthesiologists reported a shortage of at least 1 anesthesia-related drug.7 The drugs most likely to be reported were fentanyl, thiopental, and succinylcholine respectively. A majority of the anesthesiologists who responded reported less than optimal anesthetic outcomes with greater incidences of minor complications, including post-operative nausea and vomiting as well as prolonged times in the operating room and the recovery areas.
Drug shortages ultimately affect patient safety. Whenever a different brand or concentration of a drug must be purchased, prepared, or administered or whenever any clinician uses an unfamiliar alternative medication, the safety of the patient is threatened.3 Some practitioners, when confronted with a shortage or rationing of certain medications, may be tempted to forego the dispensing guidelines and use large volume single-dose vials multiple times. In 2010, Premier Health conducted a survey and, of the responding hospitals, 89% reported drug shortages that may have caused a medication safety issue or an error in patient care.8 A follow-up survey released in December 2013 assessed the effect of drug shortages on pharmacy directors. Of the respondents, 38% reported a history of patient complaints.9 Despite these survey results, currently no national database exists where patients and practitioners can report adverse effects, medication errors, and other patient outcomes that result from drug shortages.
Manufacturing problems are the most likely cause for drug production delays and usually result from quality problems with particulate matter or bacterial contamination.2 The International Society for Pharmaceutical Engineering (ISPE), the world’s largest not-for-profit pharmaceutical association that consists of engineers, microbiologists, chemists, suppliers, pharmacists and other professionals, represents many of the key stakeholders in the manufacture of drugs. The ISPE is acutely aware of the problem of drug shortages. Past president and former CEO Nancy S. Berg commented, “Everyone desires an industry that is free of shortages; after all we are all patients.”10
The ISPE released a report that focused on manufacturing in June 2013.11 In its report, the ISPE identified the lack of a “quality system” to be the most common cause of drug shortages for sterile products. It defined a quality system as a system that complies with regulations enforced by the FDA as well as internal procedures and specifications. In order to prevent or mitigate drug shortages the ISPE recommended that an effective quality system be implemented, including methods to ensure reliable manufacturing equipment since the actual equipment, not the cleaning or support of it, contributes most to drug shortages.
An ISPE Report in June 2013 examined manufacturers in the industry who were best able to mitigate drug shortages.11 Those manufacturers that could not manage drug shortages, according to the report, had focused more on building information technology to identify potential shortages or had expanded efforts to establish redundancy in the supply chain. The companies that were most successful at mitigating drug shortages had implemented most, if not all, of the processes listed below. One interesting finding that came out of the study was that the majority of the manufacturing representatives believed improvements to new production lines to increase capacity should be the primary area of focus for the industry.
The findings by the ISPE were published in the Drug Shortage Prevention Plan,12 an industry roadmap for improvement. This plan provides information to manufacturing organizations for ways to prevent drug shortages. More specifically, the plan recognizes the unique role the industry plays in drug shortages and offers guidance to help discover the root causes of these shortages and develop quality systems to ensure a robust, resilient, and reliable supply of medications.
Cited Factors in Successful Prevention of Drug Shortages
- Strong Quality Systems that lead to compliance with manufacturing regulations
- Documented corporate goal to avoid drug shortages
- Strong Quality Systems track record and Current Good Manufacturing Practice Inspection History
- Corporate Goals Tagged to Drug Shortage Prevention
- Ability to Quickly React to Drug Shortages
- Strong Relationship With Regulatory Authorities
- Strong Communication Link With Regulatory Authorities
- Dedicated Resources Focused on Preventing Drug Shortages
- Incentives Tied to Preventing Drug Shortages
- Metrics Defined Around Drug Shortages.
A newly updated 2014 ISPE Drug Shortage Prevention Plan12 focuses on a multidimensional “Hexagon Model” to ensure a sustainable drug shortage prevention plan by using building blocks to serve as a roadmap. The blocks consist of Corporate Quality Culture, Robust Quality System, Metrics, Building Capability, Business Continuity Planning, and Communication with Authorities.
The FDA is also a major stakeholder in the drug shortage problem. In order to be more responsive about drug shortages, the FDA has upgraded its drug shortage website13 to include a searchable database with therapeutic categories that allows subscribers to receive RSS feeds specific to an area of practice as well as the planned future development of a smartphone application to make the information more readily available. The FDA has also begun to include notices from pharmacists and other health care professionals who report price increases for drugs sold by third parties on the “gray market.” While the FDA can mandate reporting of drug shortages, it has no authority to regulate the quality of manufacturing. To address this problem, according to Stephen King, the public affairs specialist at the Center for Drug Evaluation and Research, the FDA is exploring ways to incentivize and prioritize manufacturing quality.
Professional organizations have also been active in addressing drug shortages. The American Society of Anesthesiologists is committed to the implementation of the drug shortage provisions of the FDASIA and working with stakeholders and the FDA to prevent and mitigate drug shortages. The American Society of Health-System Pharmacists (ASHP) most recently collaborated with the Assistant Secretary for Preparedness and Response to develop a resource for managing critical shortages of IV fluids. During a recent national shortage of 0.9% injectable sodium chloride, the ASHP conducted a survey and found that 75% of the respondents reported the product to be in short supply.14 The ASHP conveyed this information to the FDA and other health care organizations and encouraged conservation efforts that included patient triage and dosage changes.
Anesthesiologists, Nurse Anesthetists and Anesthesiologist Assistants are integral to the fight against drug shortages. The ASHP has developed a Drug Shortage Team Reporting Form on its website for health care professionals to provide valuable information regarding the sense of urgency and the magnitude of drug shortages that may affect patients at the point of care.15
Hospitals, as institutions that reward manufacturers with their business, are also active stakeholders in the fight against drug shortages. Presently, drug suppliers are not required to list where a drug has been made or which factory manufactured it. Hospitals, therefore, cannot be assured either of the drug’s quality or the consistency of its supply. Dr. Erin Fox, the director of the University of Utah Drug Information Service, believes hospitals may purchase more drugs from manufacturers when they are assured of a steady supply and a high quality product.
One such measure that may improve transparency and influence ordering patterns is the Drug Quality and Security Act (DQSA) signed by President Obama in November 2013.16 The DQSA will require all pharmaceutical drugs to have a complete list of transaction information whenever the drugs are bought or sold. Starting in July 2015, hospital pharmacies will have to reject drug products without an accompanying transaction history. In addition, within the next 4 years drug manufacturers must add serial numbers to all drugs packaged, and within the next 10 years the industry must implement electronic codes to track medications that travel from the manufacturing facility to hospital pharmacies.
Pharmacies, too, play an integral role in addressing drug shortages. The University of Utah Health Care system is an example of what can be done: alternative drugs are purchased when available; proactive plans are developed; pharmaceutical drug needs are prioritized; physicians and anesthesia professionals are educated about alternatives; and physicians are kept informed about current and impending drug shortages. About a quarter of hospitals pharmacy directors report that they have added at least one full-time position in order to manage and ameliorate drug shortages.9
Looking to the future, more information is needed from patients about how drug shortages affect them. A reporting system for patients, similar to the FDA’s post-marketing surveillance program, may provide valuable information. In addition, pharmacies, physicians and and anesthesia professionals, and professional organizations can reach out to patients to assess how drug shortages have affected their health and safety. Finally, identification of locations most affected by drug shortages may help to focus efforts to address new and ongoing drug shortages.
Drs. Orlovich and Kelly are both with UC Irvine Health, Orange County, CA. Neither author has financial conflicts of interest to report.
References
- Food and Drug Administration Safety and Innovation Act. Public Law 112-144, July 9, 2012, 126 Stat. 993. http://www.gpo.gov/fdsys/pkg/BILLS-112s3187enr/pdf/BILLS-112s3187enr.pdf.
- United States Food and Drug Administration. Strategic plan for preventing and mitigating drug shortages. http://www.fda.gov/downloads/Drugs/DrugSafety/DrugShortages/UCM372566.pdf. Accessed December 9, 2014.
- Fox ER, Sweet BV, Jensen V. Drug shortages: a complex health care crisis. Mayo Clin Proc. 2014 Mar;89:361-73.
- United States Food and Drug Administration: FDA Voice. FDASIA (Food and Drug Administration Safety and Innovation Act) at year two. http://blogs.fda.gov/fdavoice/index.php/tag/drug-shortages/. Accessed December 9, 2014.
- University of Utah Drug Information Service. Current drug shortage statistics [data on file]. October 1, 2013.
- United States Government Accountability Office. Drug shortages: Public health threat continues, despite efforts to help ensure product availability. GAO-14-194. http://www.gao.gov/assets/670/660785.pdf. Published February 2014. Accessed November 22, 2014.
- 2012 ASA drug shortage survey results. https://www.asahq.org/For-Members/Advocacy/Washington-Alerts/2012-ASA-Drug-Shortage-Survey-Results.aspx. Published April 17, 2012. Accessed November 22, 2014.
- Cherici C, Frazier J, Feldman M. Navigating drug shortages in American healthcare: A Premier healthcare alliance analysis. March 2011.
- McLaughlin M, Kotis D, Thomson K, et al. Effects on patient care caused by drug shortages: A survey. JMCP Mission Statement and Editorial Policy. 2013;19:783-788.
- Interview with Nancy S. Berg. President & CEO, International Society for Pharmaceutical Engineering.
http://www.nnepharmaplan.com/insights/angle-magazine/us-life-science-industry/articles/interview-with-nancy-s-berg-ispe-president-and-ceo/?utm_source=facebook.com&utm_medium=social&utm_content=Interview+with+Nancy+S.+Berg&utm_campaign=Angle+Jun-13. Published June 2013. Accessed November 22, 2014. - International Society for Pharmaceutical Engineering. Report on ISPE Drug Shortages Survey. http://www.ispe.org/drug-shortages/2013junereport. Published June 2013. Updated May 8 2014. Accessed November 22, 2014.
- International Society for Pharmaceutical Engineering. ISPE Drug Shortages Prevention Plan. A holistic view from root cause to prevention. http://www.ispe.org/drugshortagespreventionplan.pdf. Published October 14, 2014. Updated October 14, 2014. Accessed November 22, 2014.
- United States Food and Drug Administration. Drug Shortages. http://www.fda.gov/Drugs/drugsafety/DrugShortages/default.htm. Updated November 26, 2014. Accessed December 3, 2014.
- Traynor K. Saline shortage prompts conservation efforts. American Journal Of Health-System Pharmacy. 2014;71:520-522.
- American Society of Health-System Pharmacists. Report to ASHP’s Drug Shortage Team. http://ashp.az1.qualtrics.com/SE/?SID=SV_25KOx5N9FJ Yhuyp. Accessed December 3, 2014.
- Drug Quality and Security Act. Public Law 113-54, November 27, 2013, Stat. 587. http://www.gpo.gov/fdsys/pkg/BILLS-113hr3204enr/pdf/BILLS-113hr3204enr.pdf.


 Issue PDF
Issue PDF