Editor’s Note: The fall 2003 Issue of this Newsletter contained an article entitled “Can We Alter Long-Term Outcome? The Role of Anesthetic Management and the Inflammatory Response,” by Drs. Meiler, Moond, Mayfield, and Head. The following article is the second in this two-part series and provides a glimpse into evidence linking perioperative inflammation and long term morbidity and mortality. It raises some very interesting and provocative questions. As part of this initiative exploring perioperative inflammation, the APSF is organizing a multi-specialty Expert Panel entitled “Anesthetic Depth, Inflammation, and Surgical Outcomes,” to be held in Washington, DC, in September 2004, under the leadership of David Gaba, MD. A world-class group of investigators will meet to consider the current data, identify gaps in understanding, develop research strategies, and discuss the potential impact on perioperative management. Surgical leaders will participate, including Shukri Khuri, MD (VA), Thomas Russell, MD (ACS), and David Hunt (CMS), as well as anesthesiologists and scientists including Charles Serhan, MD, Simon Gelman, MD, Lee Fleisher, MD, Rober Legasse, MD, Marcel Durieux, MD, Steffen Meiler, MD, and Terri Monk, MD. Patient Safety experts will include Robert Stoelting, MD, and Jeffrey Cooper, PhD. This panel will be co-sponsored by the Joint Commission on Accreditation of Healthcare Organizations, represented by Jerold Loeb, PhD, and the National Quality Forum, represented by Kenneth Kizer, MD.
Our short-term interventions may have long-term consequences.
The first article in this two-part series introduced the APSF readership to the concept that medical decisions during the perioperative period may have long-term consequences for patient safety.1 The potential relationship between anesthesia management and long-term outcome is not intuitively obvious. Anesthesia practice typically functions within a “beat-to-beat” environment where intraoperative complications happen suddenly, anesthetic drugs wear off quickly, and patient outcomes are measured within hours or at most a few days following surgery. Most anesthesia patient safety initiatives have naturally focused on events that occur while anesthesiologists are actively involved with care—a period that has now become extraordinarily safe. This focus may need to shift as the risk of dying during the first postoperative year may be as high as 5% to 14% in certain patient populations. New evidence suggests that this mortality might be influenced by specific anesthetic interventions at the time of surgery.2-4
Recent biomedical research has demonstrated the importance of inflammation in the progression of chronic diseases such as atherosclerosis, cancer, and dementia. Anesthesia and surgery are also associated with a dramatically increased inflammatory response and concurrent suppression of cell-mediated immunity.5,6 Since most long-term deaths after surgery are due to cardiovascular events and cancer, it is reasonable to postulate that perioperative immune responses play some role in these outcomes. In this article we discuss some of the research on this topic, and briefly address three broad questions:
- What is the evidence that perioperative inflammation and immunity are determinants of long-term morbidity and mortality?
- Do we have evidence that anesthesia care is likely to affect this biology? Is it possible that some anesthetic approaches accelerate disease processes while others have protective effects?
- Will it be possible, by using these insights, to improve preoperative risk stratification and develop strategies that reduce long-term adverse events?
Inflammation: A Key Element in Disease
There is now a large and growing literature on the role of inflammation in the pathogenesis of atherosclerosis and cancer. Atherosclerosis is generally held to be an inflammatory disease of the vascular wall, and it responds to treatment with diverse classes of anti-inflammatory medications, such as aspirin, beta-blockers, HMG-CoA reductase inhibitors (statins), ACE-inhibitors, and activators of PPAR-a (fibrates).7,8 Statins, for example, have been shown in large trials to reduce mortality, but this benefit cannot be completely explained by their effects on cholesterol. It is likely that some of the benefit comes from their significant vascular anti-inflammatory effects.9,10 Another body of research suggests a link between inflammation and cancer. Many cancers not only arise from areas of chronic infection and inflammation (e.g., hepatitis/hepatocellular carcinoma or inflammatory bowel disease/colon cancer), but they often require a pro-inflammatory milieu to support their growth and metastatic spread.11 The fact that chronic use of aspirin and NSAIDs is associated with a reduced incidence of certain malignancies further substantiates the role of inflammation in the biology of cancers.12
“Inflammation is a local, protective response to microbial invasion or injury. It must be fine-tuned and regulated precisely, because deficiencies or excesses of the inflammatory response cause morbidity and shorten lifespan.”
— Nature 2002;420:853
What about anesthesia and surgery? It is now well recognized that surgery frequently produces profound changes in both the innate and acquired immune systems.5,6 The duration and the extent of a surgical procedure as well as preoperative anxiety and postoperative pain can all influence this response. In the perioperative period, there is a heightened inflammatory state, orchestrated by the release of pro-inflammatory cytokines, reduced levels of anti-inflammatory cytokines, and increased production of arachidonic acid metabolites, superoxide radicals, and other mediators. Interestingly, individual genetic profiles, assessed by gene polymorphism, may also predispose certain patients to an enhanced inflammation risk.13 To put this concept into perspective, we propose a hypothetical model to explain how the perioperative immune response might impact long-term outcomes (Figure 1). This model is supported by the following rationale and studies that document the postoperative progression of disease known to be influenced by inflammatory processes:
- The typical pro-inflammatory mediators released in surgical patients overlap significantly with those involved in atherosclerosis and cancer.5-9
- The beneficial effects of beta-blockers on postoperative cardiac risk14 were initially attributed to altered hemodynamics; however, more recent studies demonstrated their significant anti-inflammatory properties.15,16
- Surgical patients demonstrate reduced lymphocyte counts and function, which could result in impaired tumor cell surveillance and elimination. Several studies have reported on the association of surgery and an increased incidence of metastatic tumor spread.17-19
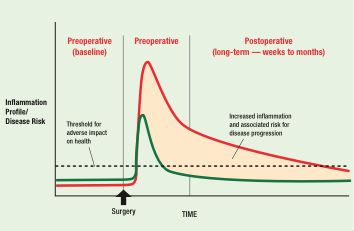
Figure 1. Perioperative Inflammation and Long-Term Risk: A Hypothetical Model. The green curve depicts the perioperative inflammatory response of an otherwise healthy individual, characterized by a transient elevation in various inflammatory mediators and then prompt return to a baseline state. In a high-risk patient (red curve), the amplitude and duration of the inflammatory response are prolonged. This sustained period of inflammation may contribute to new or accelerated disease risk.
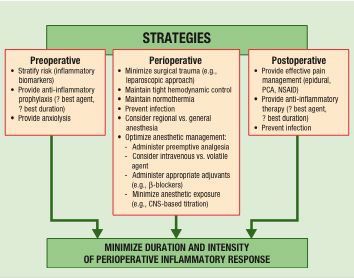
Figure 2. Conceptual framework of strategies that may minimize the perioperative inflammatory response and long-term adverse outcomes.
Possible Changes in Perioperative Management: Considering the Immune System At this point, the link between patient outcome and immune response is circumstantial, but credible. Enough is known to hypothesize a variety of clinical interventions that might improve or protect immune homeostasis (see Figure 2).
- The neuroendocrine response to stress is an important modifier of immune function, and anxiety, fear, and pain have been shown to be associated with adverse outcomes.20 There is a wealth of data on modification of stress hormone responses by anesthetic drugs, but very few studies relating hormone levels to long-term outcome. Stress hormones like norepinephrine can trigger a pronounced and immediate activation of pro-inflammatory cells and cytokines.21,22 The triggers for stress responses are both psychological and physical, so it seems reasonable that postoperative immune function could be improved by increased attention to perioperative anxiolysis and analgesia.
- Once adrenergic stress hormones have been released, their pro-inflammatory signals can be effectively intercepted with the use of beta- or alpha-blockers.22 Despite well-documented benefits for short- and long-term outcome, the perioperative use of beta-blockers remains disconcertingly low (the topic was previously discussed in this newsletter23,24). Given the success of beta-blocker treatment, similar outcome studies using other anti-inflammatory drugs (statins, COX-2 inhibitors, ACE-inhibitors, local anesthetics) deserve high priority.
- Perioperative temperature management is now known to play an important role in postoperative inflammatory processes. Maintenance of intraoperative normothermia improves wound healing and reduces wound infection,25 while mild hypothermia is known to be associated with lymphocyte suppression and increased stress hormone levels.26,27
- A small number of studies indicate that certain anesthetic agents exert direct toxic effects and may contribute to long-term risk. In some clinical studies, volatile anesthetics have been associated with a greater systemic inflammatory response compared to total intravenous anesthetics with adjuvant narcotic therapy.28,29 In vitro studies have shown that volatile anesthetics have substantial immunosuppressive effects by inducing programmed cell death in lymphocytes, diminishing lymphocyte function, and altering the distribution of lymphocyte cell subsets.30-33 In a study of metastatic melanoma in the mouse, volatile anesthetics alone (without surgery) caused a 2- to 3-fold increase in tumor burden.19 It is too early to link the immune-modulating effects of volatile anesthetics to an increased risk for tumor growth and spread in humans, but further work in this area is clearly needed.
- The studies of Weldon and Lennmarken show that deeper levels of anesthetic effect and, presumably, larger average doses of volatile anesthetics are associated with increased risk of mortality.2,3 This suggests a possible patient benefit from reducing the overall exposure to inhaled anesthetic agents. The anesthetic concentration needed to prevent consciousness (MAC-awake) is much less than that needed to prevent movement (MAC) or autonomic responses (MAC-BAR).34 Titrating volatile agents to suppress movement or to blood pressure and heart rate endpoints, not only results in the administration of higher doses, but may also be associated with underutilization of adjuvant therapy, such as beta-blockers and opioids. When anesthetic agents are titrated to hypnotic endpoints using EEG-based monitors, there is a significant reduction in anesthetic dosing.35-37
Regardless of the interventions we ultimately choose to investigate, it will be important to “measure” inflammation by analysis of appropriate serum markers. Measuring inflammation at preoperative baseline may significantly improve the accuracy with which we stratify patients for operative risk. The markers of inflammation, C-reactive protein and myeloperoxidase, have a high predictive utility in determining the clinical course and acute event rate of coronary artery disease.38,39 Preoperative measurements of these markers may provide a “window” on the inflammatory state or vulnerability of coronary lesions and improve outcome predictions. It is also possible that a postoperative profile of these bio-markers may identify patients who would benefit from more sustained anti-inflammatory treatment strategies. Similarly, these postoperative profiles may provide a means for optimizing anesthetic management.
In summary, the available evidence strongly suggests that immune system dysfunction in the perioperative period, with its combined pro-inflammatory and immuno-suppressive effects, can influence long-term disease progression, morbidity, and mortality. A substantial amount of research — much of it, we hope, by members of the APSF — is needed to establish whether changes in anesthetic practice can meaningfully reduce this risk. Should anesthesiologists really be looking at events 6 months or a year following surgery? The demographics are certainly compelling, since even a small improvement in 1-year outcome could mean thousands of lives saved each year and a significant reduction in economic burden. Although some of the observations shared in this article are still at the conceptual stage, we are confident that research on perioperative inflammation and immunity will yield significant improvements in long-term patient safety. Dr. Meiler is an Associate Professor and Vice Chair for Research in the Department of Anesthesiology and Perioperative Medicine, and Director of the Program for Molecular Perioperative Medicine & Genomics at the Medical College of Georgia. Dr. Monk is a Professor in the Department of Anesthesiology at the University of Florida College of Medicine. Dr. Mayfield is an Associate Professor and Vice Chair of Clinical Anesthesia in the Department of Anesthesiology and Perioperative Medicine at the Medical College of Georgia. Dr. Head is Professor and Chair of the Department of Anesthesiology and Perioperative Medicine at the Medical College of Georgia.
References
- Meiler SE, Monk TG, Mayfield JB, Head CA. Can we alter long-term outcome? The role of anesthetic management and the inflammatory response. APSF Newsletter 2003;18:33,35.
- Weldon C, Mahla ME, Van der Aa MT, Monk TG. Advancing age and deeper intraoperative anesthetic levels are associated with higher first year death rates. Anesthesiology 2002;97(Suppl):A1097.
- Lennmarken C, Lindholm ML, Greenwald SD, Sandin R. Confirmation that low intraoperative BIS levels predict increased risk of postoperative mortality. Anesthesiology 2003;99(Suppl):A303.
- Mangano DT, Layug EL, Wallace A, Tateo I. Effect of atenolol on mortality and cardiovascular morbidity after noncardiac surgery. Multicenter Study of Perioperative Ischemia Research Group. N Engl J Med 1996;335:1713-20.
- McBride WT, Armstrong MA, McBride SJ. Immunomodulation: an important concept in modern anaesthesia. Anaesthesia 1996;51:465-73.
- Salo M. Effects of anaesthesia and surgery on the immune response. Acta Anaesthesiol Scand 1992;36:201-20.
- Ross R. Atherosclerosis?an inflammatory disease. N Engl J Med 1999;340:115-26.
- Libby P. Inflammation in atherosclerosis. Nature 2002;420:868-74.
- Lefer AM, Scalia R, Lefer DJ. Vascular effects of HMG CoA-reductase inhibitors (statins) unrelated to cholesterol lowering: new concepts for cardiovascular disease. Cardiovasc Res 2001;49:281-7.
- Poldermans D, Bax JJ, Kertai MD, et al. Statins are associated with a reduced incidence of perioperative mortality in patients undergoing major noncardiac vascular surgery. Circulation 2003;107:1848-51.
- Coussens LM, Werb Z. Inflammation and cancer. Nature 2002;420:860-7.
- Baron JA, Sandler RS. Nonsteroidal anti-inflammatory drugs and cancer prevention. Annu Rev Med 2000;51:511-23.
- Tomasdottir H, Hjartarson H, Ricksten A, et al. Tumor necrosis factor gene polymorphism is associated with enhanced systemic inflammatory response and increased cardiopulmonary morbidity after cardiac surgery. Anesth Analg 2003;97:944-9.
- Auerbach AD, Goldman L. Beta-blockers and reduction of cardiac events in noncardiac surgery: clinical applications. JAMA 2002;287:435-44.
- Jenkins NP, Keevil BG, Hutchinson IV, Brooks NH. Beta-blockers are associated with lower C-reactive protein concentrations in patients with coronary artery disease. Am J Med 2002;112:269-74.
- Anzai T, Yoshikawa T, Takahashi T, et al. Early use of beta-blockers is associated with attenuation of serum C-reactive protein elevation and favorable short-term prognosis after acute myocardial infarction. Cardiology 2003;99:47-53.
- Vallejo R, Hord ED, Barna SA, et al. Perioperative immunosuppression in cancer patients. J Environ Pathol Toxicol Oncol 2003;22:139-46.
- Tsuchiya Y, Sawada S, Yoshioka I, et al. Increased surgical stress promotes tumor metastasis. Surgery 2003;133:547-55.
- Moudgil GC, Singal DP. Halothane and isoflurane enhance melanoma tumor metastasis in mice. Can J Anaesth 1997;44:90-4.
- Page GG, Ben-Eliyahu S. The immune-suppressive nature of pain. Semin Oncol Nurs 1997;13:10-5.
- Maes M, Song C, Lin A, et al. The effects of psychological stress on humans: increased production of pro-inflammatory cytokines and a Th1-like response in stress-induced anxiety. Cytokine 1998;10:313-8.
- Bierhaus A, Wolf J, Andrassy M, et al. A mechanism converting psychosocial stress into mononuclear cell activation. Proc Natl Acad Sci U S A 2003;100:1920-5.23.
- Royster RL. Perioperative beta-blockade can reduce morbidity and mortality. APSF Newsletter 2002;17:21,23.
- Royster RL. Perioperative beta-blockade II: practical clinical application APSF Newsletter 2002;17:54.
- Sessler DI. Mild perioperative hypothermia. N Engl J Med 1997;336:1730-7.
- Frank SM, Higgins MS, Breslow MJ, et al. The catecholamine, cortisol, and hemodynamic responses to mild perioperative hypothermia. A randomized clinical trial. Anesthesiology 1995;82:83-93.
- Beilin B, Shavit Y, Razumovsky J, et al. Effects of mild perioperative hypothermia on cellular immune responses. Anesthesiology 1998;89:1133-40.
- Schneemilch CE, Band U. Release of pro- and anti-inflammatory cytokines during different anesthesia procedures. Anaesthesiol Reanim 2001;26:4-10.
- Crozier TA, Muller JE, Quittkat D, et al. Effect of anaesthesia on the cytokine response to abdominal surgery. Br J Anaesth 1994;72:280-5.
- Matsuoka H, Kurosawa S, Horinouchi T, et al. Inhalation anesthetics induce apoptosis in normal peripheral lymphocytes in vitro. Anesthesiology 2001;95:1467-72.
- Karabiyik L, Sardas S, Polat U, et al. Comparison of genotoxicity of sevoflurance and isoflurance in human lymphocytes studied in vivo using the comet assay. Mutat Res 2001;492:99-107.
- Brand JM, Kirchner H, Poppe C, Schmucker P. The effects of general anesthesia on human peripheral immune cell distribution and cytokine production. Clin Immunol Immunopathol 1997;83:190-4.
- Corsi M, Mariconti P, Calvillo L, et al. Influence of inhalational, neuroleptic and local anaesthesia on lymphocyte subset distribution. Int J Tissue React 1995;17:211-7.
- Stanski DR. Monitoring depth of anesthesia. In: Miller RD, ed. Anesthesia, 5th ed. New York: Churchill Livingstone, 2000: 1087-116.
- Recart A, Gasanova I, White PF, et al. The effect of cerebral monitoring on recovery after general anesthesia: a comparison of the auditory evoked potential and bispectral index device with standard clinical practice. Anesth Analg 2003;97:1667-74.
- Luginbuhl M, Wuthrich S, Petersen-Felix S, et al. Different benefit of bispectal index (BIS) in desflurane and propofol anesthesia. Acta Anaesthesiol Scand 2003;47:165-73.
- Guignard B, Coste C, Menigaux C, Chauvin M. Reduced isoflurane consumption with bispectral index monitoring. Acta Anaesthesiol Scand 2001;45:308-14.
- Ridker PM, Morrow DA. C-Reactive Protein, inflammation, and coronary risk. Cardiol Clin 2003;21:315-25.
- Brennan ML, Penn MS, Van Lente F, et al. Prognostic value of myeloperoxidase in patients with chest pain. N Engl J Med 2003;349:1595-604.

