New and Different Patient Safety Challenges Must Be Anticipated
In the aftermath of the September 11 tragedies and subsequent anthrax mail deliveries, society and physicians scrambled to become familiar with the identification and treatment of the most likely bioterrorist agents.1-12 It is no longer a question of if bioterrorism will occur, but when will it happen again, and how will we recognize and manage such occurrences. Future bioterrorist events may be overt and publicized or they may be covert and associated with delays in identification, containment, and treatment. Either overt or covert attacks may stress medical systems dramatically and overwhelm our existing healthcare resources.
The development of new, emerging, or re-emerging modified pathogens is also on the horizon. It is frightening to realize how easily highly infectious agents can be produced, transported, and deployed. The elimination of sources of bioterrorist weapons, prevention of dissemination, and early recognition of attack are keys to limiting the devastating physical, emotional, and financial effects of these weapons of mass destruction.
This article will selectively discuss potential bioterrorist-initiated infections, address initial triage and pathogen identification, and touch on specific concerns for anesthesia and critical care providers in the management of biological exposures. Immediate access to state-of-the-art care is facilitated by information available on reliable web sites from agencies such as the Centers for Disease Control and Prevention (CDC), public health organizations, medical societies, and the military and Defense Department. Therefore, this review closes with a list of readily obtainable publications and web-based guides on bioterrorism, since the information about specific approaches to bioterrorism is likely to be in rapid flux.
Properties of the ideal bioterrorist infectious agent are outlined in Table 1. Tragically, a host of agents are readily available and additional ones are currently under development. In June 1999, a group of national experts was convened to assess the potential threat of biological agents that might be used in bioterrorist attack or biowarfare as a prelude to public health preparedness efforts. The group categorized potential agents as Category A, B, or C, with Category A agents having the greatest potential for adverse public health impact with mass casualties and requirements for broad-based public health preparedness efforts (Table 2). Category A agents also have moderate to high potential for large scale dissemination or heightened general public awareness that could cause mass public fear and civil disruption. Category B agents are a slightly lower priority, and Category C are likely to be emerging biologics. Tables 3 and 4 outline some basic epidemiologic and therapeutic approaches to several group A infections.




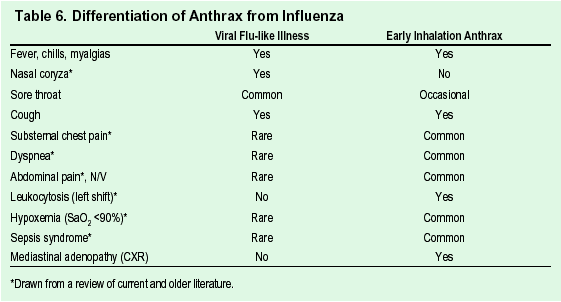
Healthcare providers must remain alert to illness patterns and diagnostic clues that might signal an unusual infectious process secondary to a bioterrorist attack (e.g., 2 or more patients presenting with signs and symptoms of fever, cough, sepsis, and rapidly progressive respiratory failure with marked mediastinal adenopathy on chest x-ray as occurred in the recent cases of inhalational anthrax). Healthcare members who suspect such a cluster of infectious diseases or unusual illness should immediately contact their local and state health department. Various factors that might indicate the intentional release of a biologic agent are listed in Table 3.
Although anesthesia and critical care personnel may not be at the point of origin of a biologic attack or initiating primary assessment of patients with suspected biological toxicity, they may be involved in the care of acutely ill victims in the emergency room, operating room, and intensive care unit. These clinicians may be responsible for the provision of life-sustaining therapy such as airway management, resuscitation, hemodynamic monitoring, ventilatory support, and initiation of definitive antimicrobial therapy. Anesthesia and critical care personnel so involved will be at increased risk for inhaled exposure, direct contact with pathogens, or spread of blood-borne infection. In times of emergency, it behooves the acute care anesthesia healthcare provider and intensivist to be familiar with basic isolation as well as decontamination techniques and to strictly adhere to them. It is crucial to have a suitable index of suspicion of a bioterrorist event and to rapidly isolate, decontaminate, and triage potential patients. Concurrently, it is essential to notify proper public health and defense authorities to initiate appropriate actions that include techniques to control the source of exposure, provide prophylaxis for exposed individuals (including healthcare providers), vaccinate as needed, and identify and eliminate the source of current and additional biological threats. If patients arrive at your emergency department with suspected exposure to biological agents, it is important to triage them outside normal patient care areas and take them to a designated decontamination area to prevent secondary exposures.
Specific Agents
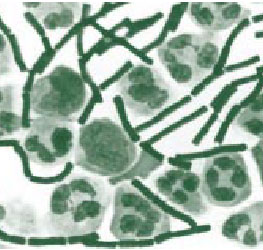
Figure 1. Anthrax Bacillus
Anthrax (Table 5), a gram-positive bacillary infection (Fig. 1), varies as to mode of delivery of infection. It may be via a respiratory (inhaled), gastrointestinal (GI), or contact (cutaneous) vector. Inhalational anthrax begins with a brief prodrome that appears to be similar to a viral upper respiratory infection with fever, myalgias, and malaise (Table 6 differentiates anthrax from influenza). A brief period of improvement may then occur, but is quickly followed 2 to 4 days later by hypoxia, dyspnea, meningitis, and radiographic evidence of widening of the mediastinum (Fig. 2). Inhalational anthrax is the most lethal of the 3 forms of anthrax. Incubation prior to active infection averages 1-7 days, but may be as long as 60 days. Prophylaxis or prior immunization may alter the incubation period. Patients with GI anthrax develop severe generalized abdominal pain followed by fever and signs and symptoms of sepsis. It is usually associated with having eaten raw or undercooked meat. The incubation period after exposure is 1-7 days. GI anthrax can develop anywhere along the GI tract from the oropharynx to the lower bowel. Cutaneous anthrax is characterized by a painless papule that becomes vesicular en route to forming a depressed eschar (Fig. 3). The incubation period for cutaneous anthrax ranges from 1 to 12 days. Patients frequently develop fever, headache, malaise, and lymphadenopathy. In contradistinction to inhalational anthrax, appropriate antibiotics are associated with significant improvement in survival for patients with cutaneous anthrax.
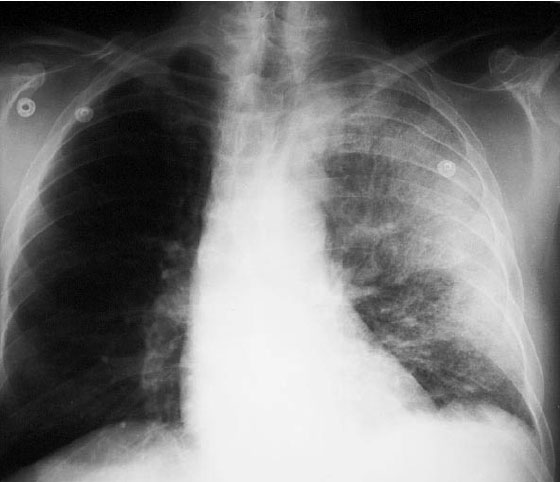
Figure 2. Chest XRay of Inhalation
Smallpox (Table 7), caused by the variola virus (a DNA virus) of the orthopoxvirus family, has been absent from the world-wide flora since the last outbreak in Somalia in 1977. However, there are reports of stockpiles of smallpox that have been bioengineered to be more virulent and contagious. Smallpox spreads readily from person-to-person via droplet nuclei or aerosols from the oropharynx of infected patients or by direct contact from contaminated clothing or bed linen. Infectivity wanes quickly in infected patients as lesions scab over. After a 3-4 day asymptomatic period, when the virus spreads hematogenously from the oropharynx, a second viremia occurs by day 12 to 14, and results in high fever, malaise, and prostration with headache, backache, severe abdominal pain, and in some patients, delirium. A widespread maculopapular rash becomes vesicular and subsequently pustular. “Hemorrhagic” smallpox is a more abrupt and usually fatal form of the disease to which pregnant woman are particularly susceptible. “Malignant” smallpox is frequently fatal as well, with rapid onset of symptoms, but it rarely progresses to pustular skin lesions. Strict isolation of suspected or known cases of smallpox is crucial to limit the spread of this highly contagious virus. Supportive care for affected patients is the backbone of therapy while timely identification and vaccination of exposed individuals should be undertaken as quickly as possible.
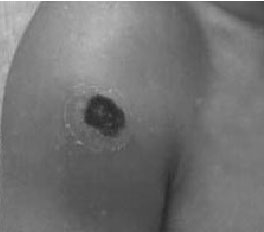
Figure 3. Cutaneous Anthrax
Although modern public health practices, advances in sanitation, and modern antibiotics have limited widespread outbreaks of plague (Table 8), it remains an important potential aerosolized biologic that is no longer dependent on the flea as a vector for transmission directly to humans or rodents. The pneumonic form of disease is the most likely result of inhalation of this gram-negative bacillus, which causes a reported mortality of 25%. The aerosolized plague bacillus is viable for up to an hour after being released, and infected patients may spread additional bacilli by coughing. Patients with pneumonic plague initially present with signs and symptoms of severe respiratory infection such as tachypnea, dyspnea, and cyanosis. Examination of the chest suggests consolidation. Patients may rapidly progress to shock and end-organ failure. Patients with advanced disease may develop purpuric lesions and necrotic digits.
Bioterrorist-mediated tularemia (Table 9) would be caused by aerosolization of this small gram-negative coccobacillus resulting in a plague-like respiratory illness characterized by fever, pharyngitis, bronchitis, pneumonia, pleuritis, and hilar lymphadenitis, developing 3-5 days after exposure. It is less fulminate and less lethal than infections secondary to Yersinia pestis. Transmission of tularemia from person to person has not been documented. A relapsing and debilitating illness may develop in undiagnosed and untreated patients.
Botulinum (Table 10) toxin is odorless, tasteless, and colorless. It is formed by the spore forming gram-positive bacillus, C. botulinum, and is the most poisonous substance known. It irreversibly blocks the vesicular release of acetylcholine at the neuromuscular junction. This results in an acute, afebrile, symmetric, descending flaccid paralysis that always starts in the bulbar musculature as evidenced by visual abnormality, and difficulty with speech and swallowing. The onset of symptoms is related to the dose of toxin absorbed and may vary from hours to up to 8 days. The disease varies from those with mild weakness to those who appear comatose with dense plegia. Axonal regeneration, which takes weeks to months, is required to reverse the effects of the toxin.
The Viral Hemorrhagic Fever (VHF) Syndrome (Table 11) is the clinical entity used to describe an infectious process caused by a group of RNA viruses that includes Ebola, Lassa, Marburg, and the South American hemorrhagic viruses. Although mainly spread in nature by arthropod vectors, these viruses are highly infectious when weaponized as aerosols. The syndrome causes an abrupt onset febrile illness characterized by malaise, prostration, generalized signs of vascular permeability, and blood pressure compromise. The various hemorrhagic viruses have an incubation period of 2 to 19 days. A truncal maculopapular rash starts approximately 5 days after the onset of acute illness followed by petechiae, ecchymoses, and frank hemorrhage as the disease progresses. Fulminate VHF typically evolves to shock and generalized mucous membrane hemorrhage. VHF is accompanied by evidence of direct neurological, hematopoietic, or pulmonary involvement, while hepatic dysfunction and renal failure develop in proportion to cardiovascular compromise.





Summary
Americans are revisiting the long history of biological warfare that dates back to the Dark Ages when plague-infected corpses were catapulted into besieged cities. The scourge of biological warfare has grown dramatically during the past century through modern biological warfare research and weaponization of previously controlled or emerging pathogens. We must combine prevention of attack and solid epidemiologic vigilance with local, regional, and national preparation that includes education, public awareness, development of vaccines, and ready transport and availability of evolving therapies. As the Steve Miller Band said, “We’re traveling fast to things in the past, it’s a brave new world.”
Dr. Coursin is a Professor of Anesthesiology and Medicine and Dr. Ketzler is an Assistant Professor of Anesthesiology at the University of Wisconsin School of Medicine, Madison, WI. Dr. Kumar is an Assistant Professor of Medicine at Rush Medical College, Chicago, IL. Dr. Maki is the Ovid S. Meyer Professor of Medicine and Chief of Infectious Disease and Infection Control at the University of Wisconsin School of Medicine, Madison.
References
- Franz DR, Jahrling PB, Friedlander AM, et al. Clinical recognition and management of patients exposed to biological warfare agents. JAMA 1997;278:399-411.
- Lane HC, Fauci A. Bioterrorism on the home front: a new challenge for American medicine. JAMA 2001;286:2595-7.
- CDC. Biological and chemical terrorism: strategic plan for preparedness and response. MMWR 2000;49 (no. RR-4).
- Recognition of illness associated with the intentional release of a biologic agent. MMWR 2001;50:893-7.
- Henderson DA. The looming threat of bioterrorism. Science 1999;283:1279-82.
- Barbera J, Macintyre A, Gostin L, et al. Large-scale quarantine following biological terrorism in the United States. Scientific examination, logistic and legal limits, and possible consequences. JAMA 2001;286:2711-7.
- Swartz MN. Recognition and management of Anthrax—an update. N Engl J Med 2001;345:1621-6.
- Inglesby TV, Henderson DA, Bartlett JG, et al. Anthrax as a biological weapon: medical and public health management. JAMA 1999;281:1735-45.
- Henderson DA, Inglesby TV, Bartglett JG, et al. Smallpox as a biological weapon: medical and public health management. JAMA 1999;281:2127-37.
- Dennis DT, Inglesby TV, Henderson DA, et al. Tularemia as a biological weapon: medical and public health management. JAMA 2001;285:2763-73.
- Arnon SS, Schechter R, Inglesby TV, et al. Botulinum toxin as a biological weapon: medical and public health management. JAMA 2001;285:1059-70.
- Drugs and vaccines against biological weapons. Med Lett 2001;43:87-9
Biological and Chemical Terrorism Websites
(We suggest that information from these sites be downloaded as a PDF file so that symbols, figures, and tables are accurately reproduced.)
- www.bt.cdc.gov
CDC Bioterrorism and Response Web Page. Accessed 2/24/02. - http://www.cdc.gov/ncidod/EID/index.htm
The website of the new Emerging Infectious Diseases journal sponsored by CDC. Accessed 2/21/02. - www.hopkins-biodefense.org/index.html
A plethora of information based at Johns Hopkins. Accessed 2/24/02. - www.ama-assn.org/ama/pub/category/6206.html
Up-to-date resource sponsored by the AMA; a CD-ROM of useful sites and information is also available. Accessed 2/24/02. - www.apic.org/bioterror/news/
Timely resource. Accessed 2/24/02. - www.bioterrorism.slu.edu/
In-depth and current resource sponsored by St. Louis University. Accessed 2/24/02.
Important Emergency Contacts
FBI Special Information Operations Center
203-324-0259
FEMA 24-hour hotline
800-424-8802
USPHS Office of Emergency Preparedness
800-USA-NDMS
CDC (to report an incident)
770-488-7100
Defense Threat Reduction Agency (DTRA)
800-424-8802

