Unfamiliar Patient Care and Safety Issues Mandate Preparedness
Since September 11, U.S. intelligence agencies have issued alerts that terrorists continue to plan for further attacks. In the past months, U.S. intelligence agencies have uncovered plans of U.S. nuclear power plants at terrorist bases in Afghanistan. There is also evidence of plans designed to cause mass casualties and spread deadly radiological debris by a bombing or airline attack on a U.S. nuclear power plant or one of the Energy Department’s nuclear facilities. This type of attack, known as radiological warfare (RW), is the deliberate use of radiological materials to produce injury and death. The explosion of a radiological weapon, similar to that of an ordinary bomb, causes damage by the heat and blast liberated at the time of detonation. While such attacks have not occurred, many experts agree that it is a matter of “when” and not “if” such an event will occur. Unfortunately, the proliferation of nuclear material and technology has made the acquisition and terrorist use of ionizing radiation more likely than ever. Fortunately, the treatment of most radiation casualties is both effective and practical to decrease the morbidity and mortality from the use of nuclear and radiological weapons.
Currently there are 3 threat scenarios for radiological terrorism. The most probable scenario for the near future would be a radiological dispersion device. Such a weapon can be developed and used by any terrorist with conventional weapons and access to radionuclides. This is an expedient weapon, in that radioactive waste material is relatively easy to obtain from any location that uses radioactive sources. These sites could be a nuclear waste processor, a nuclear power plant, a university research facility, a medical radiotherapy clinic, or an industrial complex. The radioactive source is disseminated by using conventional explosives, and the debris is subsequently scattered across the targeted area. In 1996, Islamic rebels from Chechnya planted, but did not detonate, a device packed with Cesium 137, one of the most highly radioactive by-products of nuclear fission, in a Moscow park. Depending on the size of the explosive and the surrounding population density, the medical effects of the explosion could produce a significant number of deaths, while many thousands would suffer from radiation exposure.
A terrorist attack could also be made on a nuclear power plant using a commercial jet, heavy munitions, or internal sabotage. This type of attack would have an effect similar to a radiological bomb and could cause far greater casualties. If such an attack were to cause either a meltdown of the reactor core or a dispersal of the spent fuel waste, extensive casualties could be expected. To date, the significant medical effects of the radiological accident at Chernobyl provide the model for this type of radiological event, and the possibility that terrorists may attempt to attack such facilities has led to the implementation of more stringent security measures at nuclear facilities.
While the traumatic effects of blast and thermal injury are visible and tangible, the effects of radiation are not directly apparent and can only be discerned by the secondary effects. This is evident in the aftermath of the effects of the nuclear accident that took place in Chernobyl on April 26, 1986. On that day, an explosion secondary to loss of cooling capacity destroyed the nuclear reactor at Chernobyl. This explosion sent a cloud of radioactive material and gases 1 km high. Two workers died as a direct effect of the explosion. Those who remained in shielded areas of the plant survived while those who went to fight the fires eventually died of radiation effects. Sources of radiation exposure in this catastrophe came from the short-term gamma/beta emissions in the explosion and the subsequent gamma/beta radiation from the reactor core debris. Because of a lack of waterproof protective clothing and respirators, another principal source of radiation was from the deposition of particulate matter on the skin and mucous membranes of personnel in the area. The primary sources of residual radiation were due to iodine 131, strontium 90, and cesium 137.1,2 During the acute event in this low population density area, 29 casualties were evaluated in the first 30 minutes. In the next 24 hours, 140,000 people were evacuated from the 30 km surrounding Chernobyl, and potassium iodate tablets were distributed. Over the next few weeks, 230 patients were hospitalized with priority given for the early onset of nausea and vomiting, skin and mucous membrane radiation burns, and a decrease in the lymphocyte count to <1000>6 Gray (Gy) irradiation. However, this did not seem promising, as 17 of 19 died due to the associated radiation burns. All told, radiation burns (40-90% BSA) contributed to the deaths of 21 patients. In addition, 82 patients had respiratory difficulty secondary to oropharyngeal radiation burns. Over the next 4 years, the average radiation exposure around Chernobyl was 4´ normal, primarily due to residual ground contamination with cesium 137. Despite the relatively low number of acute casualties given the magnitude of the accident, the long-term impact predicts 24,000 excess cancers in Europe and 280 in the region around Chernobyl.3-5
The worst scenario, and the least likely, is a terrorist organization diverting an existing nuclear device or procuring enough material and expertise to manufacture a nuclear device. In this scenario, a terrorist group could try to purchase a nuclear weapon, as the Japanese Aum Shinrikyo cult tried to do in Russia, or build a crude device on its own and utilize ground or ship transport to deliver the weapon to the point of detonation. Evidence suggests that some groups, including the Al-Qaeda network, have attempted to obtain weapons grade material. Since 1993, there have been 175 cases of trafficking in nuclear material, 18 of which involved substantial quantities of weapons-grade material. After acquiring fissionable nuclear material, sophisticated terrorists could design and fabricate a workable atomic bomb. The wake of a nuclear terrorist attack would be large numbers of casualties with combined injuries generated from the periphery of the lethal zone. Infrastructure, economic centers, and communications would be destroyed or disrupted by the electromagnetic pulse. The large numbers of fatalities and casualties in conjunction with the psychological effects and long-term radiation effects would impose a massive burden on available medical facilities. For example, a relatively small nuclear device of 15-kilotons detonated in Manhattan could immediately kill upwards of 100,000 inhabitants, followed by a similar number of deaths afterward. In addition, advanced medical care would be available only outside the area of immediate destruction and contamination. Consequently, the primary management importance would be placed on early evacuation of casualties to other available medical centers throughout the United States.
Because of the unique nature of radiological injury, the theory and treatment of radiological casualties is taught in the Medical Effects of Ionizing Radiation Course offered by the Armed Forces Radiobiology Research Institute at Bethesda, Maryland. In addition, the course content is published in The Medical Management of Radiological Casualities, which is available at http://www.afrri.usuhs.mil.
The key principle in managing radiation casualties is an understanding of the sources and effects of radiation exposure. Exposure to radiation may result from external and/or internalized radiation sources. External sources can be the radiation emitted during a nuclear explosion and the residual particulate matter that remains after the explosion. Neutrons in addition to, gamma, alpha, and beta radiation, can cause radiation injuries. During a nuclear detonation, gamma radiation and neutrons are the most serious radiation threats (neutron damage was not detected after the Chernobyl incident). Dust and weapons fragments from a nuclear explosion or radiation dispersal continue to emit alpha, beta, and gamma radiation. While the residual gamma radiation, which is similar to x-rays, is much less intense than that emitted during the first minute after a nuclear explosion, it is highly energetic, passes through matter easily, and causes whole-body exposure. Alpha particles are a negligible external hazard, but as an internalized radionuclide source, they can cause significant local damage. Beta particles are very light, charged particles that are primarily found in radiation fallout. These particles travel a short distance in tissue; but if large quantities are involved, they can produce radiation burns that are similar to a thermal burn. Sources of internal radiation come from radioactive particles absorbed through open contaminated wounds or by inhaled and ingested radioactive material.
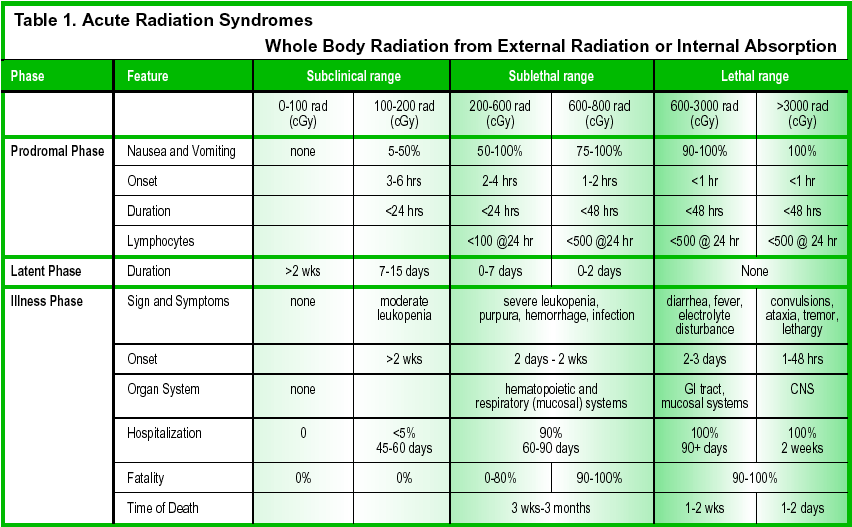
After exposure, the radiation effects can be grouped into acute and latent effects and are dependent on the radiation dose (Table 1). In the United States, the radiation absorbed dose (rad) is the measure of absorbed radiation. However, this is being replaced by the International System unit for radiation absorbed dose, the gray (Gy) (1 joule/kg); 1 Gy = 100 rad; 1 centigray (cGy) = 1 rad. The earliest effects of radiation exposure are limited to early transient incapacitation (ETI) during extensive exposure and nausea and vomiting during lesser exposures. ETI is associated with very high, acute doses of radiation (20 to 40 Gy) and has only occurred during fuel reprocessing accidents. This level of exposure is unlikely in a terrorist attack. After an initial brief loss of consciousness during ETI, the patient lapses into coma within 1-3 days and dies from vascular instability. The severity and onset of the other effects after radiation exposure are predictable. The 3 most significant radiosensitive organ systems in the body are the skin and mucosa, hematopoietic, and the gastrointestinal systems. The specific effects that occur after a variable latent phase of days to weeks are 1) thermal burn-like effects to skin and mucosa, 2) gastrointestinal enteritis, 3) bone marrow suppression with immunological dysfunction and secondary infections, and 4) hemorrhagic complications from thrombocytopenia.
While information regarding the comprehensive medical management of radiation injury is extensive, there are general guidelines that apply to decontamination, diagnosis, and management of radiological and combined injuries. Ideally, decontamination should be performed outside the hospital. Since this will not always be possible, decontamination procedures should be part of the operational plans of any treatment facility. Decontamination consideration for non-injured casualties requires standard universal precaution and removal of patient clothing. Contaminated clothing should be carefully removed, placed in marked plastic bags, and removed to a secure location within a contaminated area. Passing a radiation detector over the entire body can readily assess the presence of radiological contamination. If present, decontamination of the skin and hair is accomplished by washing. However, open wounds should be covered before decontamination. If practical, the decontamination effluent should be sequestered and disposed of appropriately.
In the case of injury and radiological exposure, aggressive therapy will be required to allow survival. Surgical priorities for acute or life-threatening injury must precede any treatment priority for associated radiation injury. Because radiologic contamination poses little risk to healthcare providers, these patients are prioritized by standard trauma protocols. In the presence of traumatic injury, hypotension must be considered to be due to hypovolemia and not radiological injury. While the skin is impermeable to most radionuclides, particles can be absorbed through wounds. Therefore, contaminated wounds should be decontaminated with copious irrigation. It should be noted that any residual fluid in the wound might hide weak beta and alpha emissions from detectors. Because wound healing is markedly compromised by radiation injury, open wounds that are allowed to heal by secondary intention will serve as a potentially fatal nidus of infection in the radiologically injured patient. If possible, all wounds should be extensively debrided and closed as soon as possible.
For internal contamination, chelation therapy may be indicated and recommendations can be obtained by a Radiation Safety Officer or Nuclear Medicine Physician. If radioiodine (from a reactor accident) is suspected, prophylactic potassium iodide (Lugol’s Solution) should be administered within the first 24 hours in order to be efficacious. After inhalation, particles less than 5 microns in diameter can be deposited in the alveoli. Larger particles will be limited to the mucociliary apparatus of the oropharynx. In either area, soluble particles will be absorbed into the blood stream and the lymphatic system. Insoluble particles will continue to irradiate surrounding tissues until cleared from the respiratory tract. This will cause inflammation and result in fibrosis and scarring. Absorption of ingested radioactive particles depends on the solubility of the contaminant. Iodine 131 and cesium 137 are rapidly absorbed while plutonium, radium, and strontium are not. The lower GI tract is the target organ for insoluble particles that pass unchanged in the feces.
For all patients with confirmed or suspected exposure, a complete blood count should be obtained on presentation and after 24 hours to determine the absolute lymphocyte count. At 24 hours, an absolute lymphocyte count <1000>
The initial care of radiologic casualties with moderate and severe radiation exposure should include early measures to reduce pathogen acquisition. These could include low-microbial-content food, clean water supplies, frequent hand washing, and air filtration. When possible, oral feeding is preferred to intravenous feeding to maintain the immunologic and physiologic integrity of the gut.
During the neutropenic phase of the radiation syndrome, the prevention and management of infection is the mainstay of therapy. These patients should be treated with a hospital’s standard regimen for neutropenic patients.6-8 Empiric antibiotic regimens should be selected based on the pattern of bacterial susceptibility and nosocomial infections in each institution. In addition, hematopoietic growth factors, such as filgrastim (Neupogen¨), a granulocyte colony-stimulating factor (G-CSF), and sargramostim (Leukine¨), a granulocyte-macrophage colony-stimulating factor (GM-CSF), are potent stimulators of hematopoiesis and may shorten the duration of neutropenia and thus reduce morbidity and mortality.9-11 As with all neutropenic patients, blood products administered should be fresh, irradiated, and CMV negative.
In summary, it is obvious that terrorist groups have investigated actions using radiological material or nuclear devices. If such an attack were to occur, the strain on medical resources will be significant due to the severity of bone marrow suppression that occurs after even moderate exposure to radioactive substances. However, the relatively slow onset of the syndromes and the advances in medical care will dramatically improve the survivability of such injuries.
Paul D. Mongan, MD, LTC MC (US Army), is an Associate Professor, and Cynthia Shields, MD, LTC MC (US Army), and Darin Via, MD, LCDR MC (US Navy) are Assistant Professors in the Department of Anesthesiology, at The Uniformed Services University of the Health Sciences in Bethesda.
References
- Kashparov VA, Oughton DH, Zvarich SI, et al. Kinetics of fuel particle weathering and 90Sr mobility in the Chernobyl 30-km exclusion zone. Health Phys 1999;76:251-9.
- Takatsuji T, Sato H, Takada J, et al. Relationship between the 137Cs whole-body counting results and soil and food contamination in farms near Chernobyl. Health Phys 2000;78:86-9.
- Abbott A, Barker S. Chernobyl damage “underestimated.” Nature 1996;380:658.
- Ivanov VK, Gorski AI, Maksioutov MA, et al. Mortality among the Chernobyl emergency workers: estimation of radiation risks (preliminary analysis). Health Phys 2001;81:514-21.
- Mettler FH, Becker DV, Wachholz BW, et al. Chernobyl: 10 years later. J Nucl Med 1996;37:24N, 26N-27N.
- Greene JN, Linch DC, Miller CB. Current treatments for infection in neutropenic patients with hematologic malignancy. Oncology Hunting; 2000;14:31-4.
- Klastersky J. Empirical treatment of sepsis in neutropenic patients. Hosp Med 2001;62:101-3.
- Reeves GI: Radiation injuries. Crit Care Clin 1999;15:457-73.
- Freyer G, Ligneau B, Trillet-Lenoir V. Colony-stimulating factors in the prevention of solid tumors induced by chemotherapy in patients with febrile neutropenia. Int J Antimicrob Agents 1998;10:3-9.
- Rubenstein EB. Colony stimulating factors in patients with fever and neutropenia. Int J Antimicrob Agents 2000;16:117-21.
- Serke S. Hematopoietic growth factors as an adjunct for neutropenic patients in the ICU: still a controversial issue. Intensive Care Med 1999;25:901-2.
Radiological Casualty Related Websites |

