Last updated: July 27, 2023
| Translations Now Available: 日本語 (Japanese), Español (Spanish), 简体中文 (Chinese (Simplified)), Français (French), Português (Portuguese (Brazil)) |

Contributors: Jeffrey Feldman, MD, Professor of Anesthesiology and APSF Chair of Committee on Technology; Robert “Butch” Loeb, MD, Professor of Anesthesiology University FL; James Philip, MD, Professor of Anesthesiology Brigham & Women’s
Disclaimer: We aim to present our recommendations and a forum for questions. Given the novelty of COVID-19, best-available clinical evidence is supported from anecdotal reports from China, South Korea, Italy and previous epidemics like SARS and MERS. The APSF does not support or endorse any specific product, equipment or trademarked technique. We strongly promote consistency with your governing bodies and organizations such as the CDC, WHO, ASA, AANA, and AAAA. These guidelines were developed using the best judgement of clinicians with experience using anesthesia machines short-term in operating rooms. They are being frequently updated based on experience using anesthesia machines long-term for intensive care ventilation. The information and materials provided herein are provided only for information and educational purposes and do not establish a standard of care or constitute medical or legal advice. Readers are reminded to consult with their own institutions and medical/legal advisors.
International Disclaimer: The following FAQ information is intended to provide the best guidance possible for safe practices during the COVID 19 pandemic. Local conditions will almost certainly dictate modifications. Hopefully there is sufficient information to empower caregivers to make good decisions to keep patients, co-workers and themselves safe.
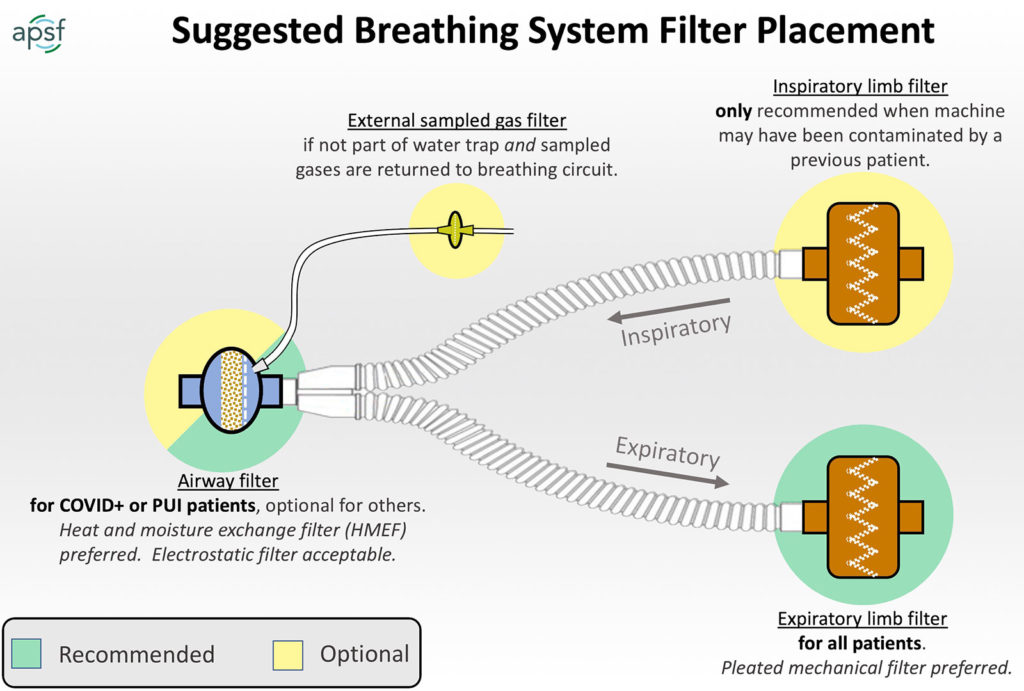
GRAPHICAL SUMMARY OF ANESTHESIA CIRCUIT FILTER RECOMMENDATIONS
See FAQ below for detailed information.
- Do I need to take any special precautions during the pandemic to protect my anesthesia machine from contamination while caring for patients who are presumed negative for SARS COV-2 infection?
- What is the best strategy for protecting the anesthesia machine from contamination by a potentially infected patient?
- What type of breathing circuit filter is likely to prevent passage of the SARS COV-2 virus from the patient to the anesthesia machine?
- Do I have to worry about transmitting the SARS COV-2 Virus from the Anesthesia Machine to a patient?
- Should the sampled gas be returned to the breathing circuit or scavenger?
- Do I need to replace the water trap for sampled gas after each patient?
- What is the proper method of cleaning the anesthesia machine between patients who are not known to be COVID + or under investigation (PUI)?
- What is the proper method of cleaning the anesthesia machine between patients who are known to be COVID + or under investigation (PUI)?
- How can the internal parts of the machine be sterilized?
- Can an anesthesia machine be used as an ICU ventilator?
- What are the recommendations for transporting intubated patients to and from the operating room?
- How can the endotracheal tube be safely disconnected from the anesthesia breathing circuit for transport, suctioning, or nebulizer therapy?
- We are running out of laryngoscope/GlidescopeTM blades. How do we clean and sterilize them?
Do I need to take any special precautions during the pandemic to protect my anesthesia machine from contamination while caring for patients who are presumed negative for SARS COV-2 infection?
Short Answer: No special precautions are needed for “negative” patients, although it is recommended to have an effective viral filter in the breathing circuit between the patient and the anesthesia machine.
A more complete answer to this question begins with a focus on how well “negative” patients can be identified. The reliability of local testing procedures and disease prevalence are important considerations. If testing is not considered to be very effective, or is not performed before a procedure, and/or the disease prevalence in the community is high, it would be prudent to treat all patients as potentially positive and follow guidelines for positive patients or PUI. All testing methods have a false negative rate and may not detect a patient who is infectious. Of course, it is possible to use maximal precautions for all patients, but that approach will consume more PPE resources, often for no reason. A practical approach is to establish criteria for defining what constitutes a “negative” patient and follow universal precautions for those patients.
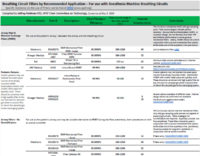
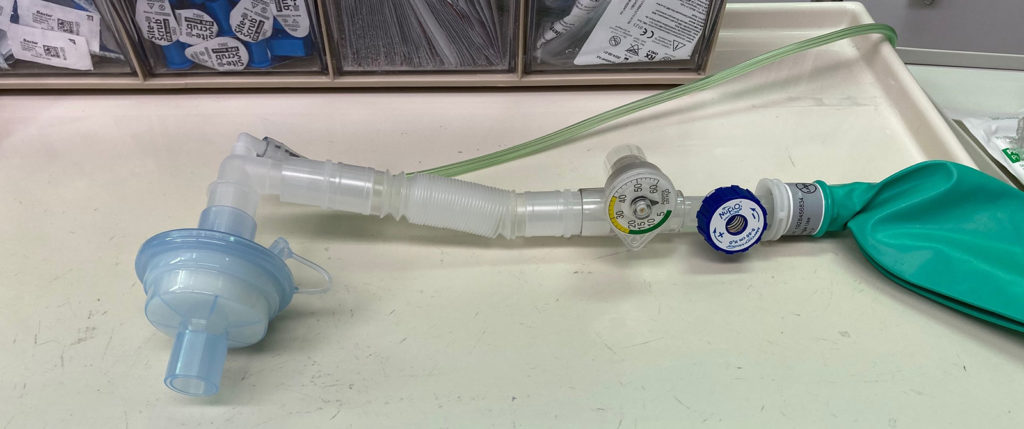
Universal precautions for preventing passage of respiratory pathogens into the anesthesia machine involve using a single high quality viral filter for the exhaled gas. This filter could be an HMEF (i.e., combination HME and filter) placed at the airway, or a breathing circuit filter placed at the connection between the expiratory hose and the machine. The evidence that respiratory pathogens can pass from one patient to another via the anesthesia machine is not very compelling, and indeed the risk is likely extremely low. Nevertheless, use of an inexpensive filter for the exhaled gas, especially if an HME will be used anyway, is a reasonable practice. A list of filter options is available here.
What is the best strategy for protecting the anesthesia machine from contamination by a potentially infected patient?
Short Answer:
- Place a “high quality” viral filter between the breathing circuit and the patient’s airway with the capability to sample gas from the machine side of the filter.
- HMEF is preferred to preserve humidification.
- If filter only is used, reducing fresh gas flow is an important strategy for preserving humidity. (1-2 L/min or less)
- Place a second filter at the end of the expiratory limb at the connection to the anesthesia machine.
- Local conditions of filter availability will determine what devices can be employed. Alternatives are described below.
- Breathing circuits should be discarded after every patient.
(See next FAQ for details on which filtration devices to use and special considerations for small patients)
There are two reasons to protect the anesthesia machine from contamination by a potentially infected patient. First, if pathogens are allowed to enter the internal parts of the machine, they could be passed on to a subsequent patient. Second, respiratory gases sampled for gas analysis can pass pathogens on to other patients or healthcare workers after leaving the gas analyzer if not managed properly.
The good news is that the same precautions can be applied to all patients. The strategy is not different depending upon the patient’s risk of infection. A “high quality” filter placed between the breathing circuit and the patient’s airway will protect the machine from contamination and also gases sampled for analysis. Heat and moisture exchange filters (HMEFs) are a good choice since they preserve airway humidity, and are designed so that sampled gas is filtered before it enters the gas analyzer (note that HMEs without filters provide no protection to the anesthesia machine or gas analyzer). It is possible to use a filter only at the airway that is not also a heat and moisture exchanger, but strategies like low flow anesthesia should be employed to preserve humidity. If a filter only is used at the airway, lower fresh gas flows (1-2 L/min or less) are desirable during maintenance of anesthesia to preserve humidity in the circuit.
While a high quality filter at the airway will protect both the sampled gas and the anesthesia machine, there are reasons to consider adding a second filter at the end of the expiratory limb. The airway filter can become soiled and lose effectiveness and in small patients, airway filters are designed to minimize dead space and as a result are generally less effective. Not only is the second filter a reasonable backup to protect the machine from any particles that pass the primary filter, but it significantly amplifies the effectiveness of the first filter.
What can I do if the two-filter option is not possible due to limited supplies?
- Can I change filters less frequently?
How often the filters need to be changed will depend upon the type of filter and clinical use. Filters mounted at the airway are susceptible to progressive occlusion by secretions and may need to be changed if resistance to flow and airway pressures become too high. Filters at the expiratory limb are less likely to need replacement due to occlusion. Manufacturers typically have recommended maximum intervals for changing filters. These recommendations should be observed unless shortages make replacement difficult or impossible, in which case continuing to use the filter is better than nothing. As long as the filter is not soiled, viral filtration effectiveness should be maintained.
- The airway mounted filter will need to be changed between every patient, The patient side of the filter contains whatever particles the patient exhaled, including virus if present.
- The expiratory limb filter seems like an option to leave in place between patients if the filter supply is constrained due to the directional flow of gas away from the patient. It is not known how long the filter can be used before performance declines. It is prudent to change as frequently as supplies allow in accord with the manufacturer’s recommendations. The outside surface should be wiped between patients.
- Reusing breathing circuit hoses is potentially more problematic because the distal inspiratory hose can become contaminated despite unidirectional flow and the outside surface of the circuit is difficult to clean reliably.
- It may be necessary to use only one filter. Unfortunately, we do not have any data on the minimum amount of filtration efficiency that will be protective. (see next FAQ) As a single solution, the airway mounted filter only could be sufficient if it is a high quality filter. A single filter at the end of the expiratory limb will protect the anesthesia machine but not the sampled gas which may not be a problem if the sampled gas does not return to the breathing circuit.
Do I need to filter the gases sampled for analysis?
It depends upon the design of your anesthesia machine. Some machines route the gas to the scavenging system whereas others direct it to the breathing circuit.
If the sampled gases end up in the scavenging system, no filtering is needed. If you are using a gas analyzer that is not integrated into the anesthesia machine it is easy to trace the exhaust gas and it should go to an active (not passive) scavenging system, not the room. For integrated gas analyzers, the connections are usually hidden. The following table indicates the output destination of the integrated gas analyzer by manufacturer:
GE: Aisys CS2 version 11 software is the only machine that we factory fit sample gas returning to the breathing system. All of these machines can be re-plumbed in the field to return the sample gas to scavenge. It would require some trained personnel to remove a panel and make the change.
| GE Anesthesia Model | Integrated or Modular Gas Measurement | Sample Gas returned to | Can sample gas return be changed by customer. |
| Aestiva | No | N/A | N/A |
| Aespire | No | N/A | N/A |
| Avance | Yes | Scavenge | Engineer in field |
| Aisys | Yes | Scavenge | Engineer in field |
| Carestation 600 | Yes | Scavenge | Engineer in field |
| Avance CS2 | Yes | Scavenge | Engineer in field |
| Aisys CS2 | Yes | Scavenge | Engineer in field |
| Aisys CS2 Software V11 (Note) | Yes | Patient Circuit | Engineer in field |
Draeger
| Anesthesia machines with integrated gas analyzers | Where gas analyzer output goes |
| Apollo | Returned to breathing circuit |
| Perseus | Returned to breathing circuit |
| Fabius/Tiro/Fabius MRI – not integrated gas analysis | Scavenger |
- If sampled gases are returned to the breathing circuit they need to be filtered. Water traps have built in filters and the viral filtration efficiency (VFE) determines the effectiveness. The GE DFend Pro water traps include a 0.2 micron filter with a VFE of 99.999%. The Draeger Watertrap 2 uses a 0.2 micron filter with a VFE of 99.99981%. If an airway filter option is not available, and the water trap filter VFE cannot be confirmed, a 0.2 micron drug injection filter similar to that used in epidural kits can be placed at the water trap and used between patients after wiping the surface.
- If the sampled gas is routed to the scavenging system, additional filtration may not be necessary as there are standards for managing biohazards in the central suction system or waste anesthetic gas system (WAGS). Check with the local facilities manager to confirm the risk of biohazard in the suction system.
- Unfiltered Sampled gas should not be exhausted directly into the OR environment or a passive scavenging system.
Do I need a filter on the inspiratory limb?
Adding another filter between the machine and the inspiratory limb is also done sometimes but is not necessary to protect the patient if the machine is kept clean. The airway mounted filter and/or expiratory limb filters achieve this already. The main reason to add an inspiratory limb filter is to eliminate the chance of error by placing a single filtered limb on the inspiratory rather than expiratory port. If there is concern that the internal parts of the machine have become contaminated, it is important to place a high quality filter at the inspiratory limb.
If filter supplies are very short, what is the most conservative approach to reusing supplies?
The following protocol from Dr Jan Hendrickx, OLV Hospital, Aalst Belgium outlines how many components of the circuit can be reused after caring for low risk patients.
- Protecting the Anesthesia station – A strategy for reusing supplies [PDF]
- ARC Medical – Circuit designed for reuse in patients who are NOT covid positive or PUI (https://www.arcmedical.com/products/circuitguardtrade.php)
What type of breathing circuit filter is likely to prevent passage of the SARS COV-2 virus from the patient to the anesthesia machine?
Short Answer:
- The minimum viral filtration efficiency (VFE) required to guarantee that SARS COV-2 virus does not pass from the patient to the anesthesia machine is not known.
- Mechanical (pleated) filters perform better than electrostatic filters and are less affected by water vapor.
- A VFE rating of 99.99% is commonly available in both filters and HMEFs and should provide good protection.
- The minimum tidal volume rating of the device placed at the airway should be considered to minimize the impact of dead space on ventilation. A rule of thumb is to consider three times the internal volume of the device as the minimum tidal volume.
There are no data testing the efficacy of breathing circuit filters for preventing transmission of SARS COV-2 to healthcare workers or patients. All breathing circuit filters decrease viral transmission, but we do not know the minimum performance specification that will be protective. The information presented here is based upon the best reference material identified to date. Pleated (mechanical) filters are preferred over electrostatic filters since the latter can lose effectiveness with humidity. Electrostatic HMEFs and filters are commonly used and may be “good enough,” but are not recommended by some sources for use in a circle system or with active humidification. Mechanical filters are larger and have lower resistance to breathing, but the dead space increases as well, which can be a problem for small patients.* There are no mechanical filters available with a recommended minimum tidal volume less than 150 mls.
Performance of filters designed to protect against viruses is measured using the Viral Filtration Efficiency or VFE and is reported for all commercial filters. VFE is determined using standardized testing in accord with the ASTM F2101 standard. The particle size tested is on average 3 microns which is accepted to approximate a viral particle suspended in a droplet. A VFE of 99.99% means that only one particle in 10,000 (104) will pass through the filter under standard test conditions that control flow rate. 30 L/minute is a commonly used flow for adult condition testing. Increased flow rate reduces the VFE.
It is not known what VFE is needed to guarantee that viral particles are not passed from an infected patient to the anesthesia machine. Combining filters in series will amplify the VFE significantly which is another compelling reason to use two filters as described above. If each filter is capable of filtering all but 1 in 104 particles (VFE=99.99%), the combined effectiveness is 1 in 108. High quality filters are readily available that meet or exceed the VFE rating of 99.99%.
APSF does not promote any particular product or manufacturer. For reference, the following are some sources for filters that would satisfy the requirements described above. Additional resources will be added as they become available.
- Summary Spreadsheet of Filter Options – Not inclusive of all available products: Click Here.
SAMPLING OF FILTER MANUFACTURERS
- Arc Medical: https://www.arcmedical.com/products/circuitguardtrade.php
- Draeger Medical: Breathing Filter/HME Guide [PDF]
- MEDLINE: https://www.medline.com/category/HMEs-and-Filters/Z05-CA05_11_01
- Medtronic:
- PALL: https://medical.pall.com/en/mechanical-ventilation/coronavirus.html
- Pall Ultipor 25
- Pall Ultipor 100
- TELEFLEX: https://www.apsf.org/wp-content/uploads/patient-safety-resources/covid-19/TELEFLEX-Gibeck-Filters.pdf
- Westmed: https://westmedinc.com/hmes-and-filters/
*A NOTE ON PEDIATRIC PATIENTS:
HMEFs that minimize dead space are typically used for pediatric patients to provide humidification and filtration. These are all electrostatic filters with VFE ratings that are not as good as the larger mechanical (pleated) filters, and the VFE is degraded by humidity. While use of an HMEF is desirable to maintain humidification, it is not clear to what degree any decrease in the VFE increases the risk of viral transmission. In particular, gas sampled from the HMEF may or may not be protected from viral transmission to the equipment and may need to be filtered separately if returned to the breathing circuit. The anesthesia machine should be protected by placing a high quality mechanical viral filter between the end of the expiratory limb and the machine. The minimum safe rating for this viral filter in the absence of a reliable airway filter is unknown, but quality filters with VFE ratings of 99.999% and above are available and should be used if possible. There is no impact on dead space by the filter placed at the end of the expiratory limb.
Recommendation for small patients where dead space imposed by an airway filter is a concern.
- Use an HMEF with acceptable dead space on the airway to insure humidification. Available devices typically have a dead space of 10 mls and a minimum tidal volume of 30 mls (5 Kg patient).
- Place a viral filter at the end of the expiratory limb with a VFE of 99.999% or greater. Change as often as supplies permit and wipe surface if not changed between patients.
- If the filter in the water trap is confirmed to have an effective VFE, gases sampled from the airway do not require additional filtering. (see above) Otherwise placing a 0.2 micron drug injection filter at the entry to the water trap will provide an added measure of protection. This is the same filter that is typically placed in an epidural tray for filtering epidural injections. Drug filter should be changed between patients.
References:
- Wilkes, A. R. (2002). Measuring the filtration performance of breathing system filters using sodium chloride particles. Anaesthesia, 57(2), 162–168.
- Wilkes, A. R. (2010). Heat and moisture exchangers and breathing system filters: their use in anaesthesia and intensive care. Part 1 – history, principles and efficiency. Anaesthesia, 66(1), 31–39.
- Wilkes, A. R. (2010). Heat and moisture exchangers and breathing system filters: their use in anaesthesia and intensive care. Part 2 – practical use, including problems, and their use with paediatric patients. Anaesthesia, 66(1), 40–51.
- Fehr, A. R., & Perlman, S. (2015). Coronaviruses: An Overview of Their Replication and Pathogenesis. In Coronaviruses(Vol. 1282, pp. 1–23). New York, NY: Springer New York.
- Sprung, C. L., Zimmerman, J. L., Christian, M. D., Joynt, G. M., Hick, J. L., Taylor, B., et al. (2010). Recommendations for intensive care unit and hospital preparations for an influenza epidemic or mass disaster: summary report of the European Society of Intensive Care Medicine’s Task Force for intensive care unit triage during an influenza epidemic or mass disaster. Intensive Care Medicine, 36(3), 428–443.
Do I have to worry about transmitting the SARS COV-2 Virus from the Anesthesia Machine to a patient?
Short Answer: As long as the machine is protected during every case with high quality filters as described in the previous FAQs there is no risk to subsequent patients.
None of the manufacturers are recommending cleaning procedures that involve the internal components of the machine as long as high quality filters are used with each patient to prevent exhaled virus from entering the machine and gas sampling lines are connected to the machine side of the filter.
Clinical experience does not suggest that transmission of pathogens from the machine to the patient is a high risk. APSF has provided guidance on this question in the past. (1,2) Pathogens must travel through the caustic environment of the CO2 absorbent, survive the low humidity of fresh gas entering the circuit, and traverse the pathways of the inspiratory side of the ventilator and breathing circuit. That said, one study showed that bacterial pathogens are capable of traversing the space between absorbent granules.(3) Further, no one would be comfortable with connecting a machine that had become contaminated with a pathogen to another patient without adequate cleaning. Manufacturers are the best source of information on cleaning procedures for specific devices. (Additional information on cleaning machines can be found at: https://www.asahq.org/-/media/files/spotlight/anesthesia-machines-as-icu-ventilators-5-07.pdf)
In the absence of data, manufacturers’ recommendations for cleaning may be excessively restrictive due to medicolegal concerns. Placing a high quality viral filter on the inspiratory limb that can be reused between patients is a viable option for putting a machine back in service quickly after following sterilization procedures.
References:
- 2009 Discussion of this topic from APSF: https://www.apsf.org/article/cross-contamination-via-anesthesia-equipment/
- 2009 Discussion of Gas Sampling Contamination from APSF: https://www.apsf.org/article/cross-contamination-via-gas-sampling-lines/
- Dryden, G. E. (1969). Risk of contamination from the anesthesia circle absorber: an evaluation. Anesthesia & Analgesia, 48(6), 939–943.
Should the sampled gas be returned to the breathing circuit or scavenger?
Short Answer:
- Gas sampled from the machine side of an HMEF is not contaminated and can be returned to the breathing circuit or the scavenging system.
- Gas sampled without the benefit of a reliable filter may be contaminated. Some water traps for the sampled gas include effective bacterial and viral filters (see below). If the effectiveness of the water trap filter cannot be verified, sampled gases should not be returned to the breathing circuit but may be returned to the scavenging system. Local processes for handling bioburden in central suction should be confirmed. Current standards for medical grade suction consider infectious risks.
- A 0.2 micron drug injection filter (eg. epidural type) can be placed at the connection to the water trap to filter sampled gases but should be changed between patients. Capnography is possible but the quality of the capnogram may be affected and should be confirmed before clinical use.
Inspired and expired gases should be routinely sampled during anesthesia care for measurement of gas concentrations. High quality filters and HMEFs are readily available that include a port for sampling gas on the machine (protected) side of the filter. Gas can then be routed through the gas analyzer to either the breathing circuit or scavenging system, there is no risk of contamination. Routing gas back into the breathing circuit has the advantage of reducing waste.
If the gas sampling line is connected to an elbow or otherwise unfiltered, for example to obtain a better capnogram, potentially contaminated gases will enter the gas analyzer typically through a water trap. There is a filter in the water trap and some of these can have be very effective for blocking viruses. The GE DFend Pro water traps include a 0.2 micron filter with a VFE of 99.999%. The Draeger Watertrap 2 uses a 0.2 micron filter with a VFE of 99.99981%. Medtronic produces the Filterline watertrap for their capnographs and are an OEM for Philips patient monitors. The Medtronic Filterline watertrap is a 0.2 micron hydrophobic filter that has been tested and shown to prevent bacterial spread. The VFE has not been documented for the Filterline but it is similar in design to the Draeger and GE products. Of note, the In Vivo Expression MRI compatible monitor which is sold by Philips uses a 0.45 micron hydrophobic filter to filter gases entering the water trap. VFE data are not available for that filter. Exhaust gas from the analyzer should be routed to a scavenging system if inhaled anesthetics are used and will prevent environmental contamination.
If the HMEF is bypassed by the sampled gas, it is recommended that sampled gas not be returned to the breathing system unless the VFE of the water trap filter can be confirmed. Sampled gas can be returned to a scavenging system designed to handle bioburden safely.
If the effectiveness of the filters in the water trap cannot be confirmed or are felt to be inadequate a 0.2 micron drug injection filter (eg. epidural type) can be placed at the connection to the water trap. This would only be necessary if the sampled gas is returned to the breathing circuit or is not routed to a scavenging system. The drug filter should be changed between patients. Capnography is possible when using the drug filter but the quality of the capnogram may be affected.
References:
- 2009 guidance on this topic from APSF: https://www.apsf.org/article/cross-contamination-via-gas-sampling-lines/
Do I need to replace the water trap for sampled gas after each patient?
Short Answer: Yes, if used with a COVID + patient or a potentially infected patient. No, if the sampled gas passes through a high quality viral filter.
The water trap has a continuous flow of gas from the patient into the machine. If that gas was filtered using a quality viral filter, the water trap is not contaminated and does not need to be changed. It is recommended to change the gas sampling tubing and wipe off the external surface of the water trap between patients.
If the water trap is used with a COVID + patient or a potentially infected patient, it seems reasonable to change it as part of a terminal cleaning procedure.
Water traps should, of course, be changed according to the recommended schedule.
What is the proper method of cleaning the anesthesia machine between patients who are not known to be COVID + or under investigation (PUI)?
Short Answer: Cleaning procedures are the same for all patients who are not known to be COVID + or PUI. Depending upon the local disease prevalence or testing resources, all patients may be treated as COVID +. High quality viral filters are recommended at all times to protect the machine from contamination. Between patients, discard disposable items – breathing circuit, reservoir bag, patient mask, gas sampling tubing, filters placed at the airway and elsewhere if supply is sufficient. Wipe all exposed surfaces. Manufacturers’ cleaning recommendations are useful for individual devices.
It is a local decision to decide if patients can be reliably tested in advance of a procedure to rule out the possibility of being COVID +. Some institutions are using negative test results to guide use of standard precautions and preserve the inventory of PPE for known patients with unknown COVID status. As noted previously, a high quality viral filter will protect the internal components of the anesthesia machine from contamination. Single use items must be discarded and not used between patients. If testing cannot be performed or local standards include treating all patients as COVID +, see the next FAQ on cleaning after COVID + patients. In general, the practice of using an HMEF and reusing the breathing circuit between patients is not desirable in part because it is virtually impossible to decontaminate the external surfaces of the circuit. Some practices in Europe are reusing circuits by just changing the airway filter after use with patients at low risk for COVID 19 infection. Manufacturers typically have recommendations for cleaning solutions that are safe and effective for cleaning between patients. Usual practices for surface cleaning are acceptable.
What is the proper method of cleaning the anesthesia machine between patients who are known to be COVID + or under investigation (PUI)?
Short Answer: Specific guidance on this topic can be found at: https://www.asahq.org/-/media/files/spotlight/anesthesia-machines-as-icu-ventilators-5-07.pdf.
Recommendations for using high quality viral filters are intended to eliminate the risk of contaminating the anesthesia machine internally so special cleaning procedures should not be needed. Although it is unlikely that the machine would be contaminated and capable of infecting a subsequent patient, there are no objective data that recommended practices can absolutely prevent cross infection of patients. Further, if the machine is used for long term ICU ventilation of a COVID + patient, it is reasonable to assume that the risk of internal machine contamination is increased. If internal contamination of the machine is suspected, the manufacturers’ recommendations for cleaning and sterilization should be followed. This can be a time consuming, labor intensive process, and may result in keeping the machine out of service longer than desired.
Manufacturers’ recommendations can be overly restrictive due to medicolegal concerns. For example, Draeger and Mindray recommend quarantining the machine for 21-28 days before re-use. There are no data to support this recommendation or any modification thereof. Fortunately, many machines are designed to be able to sterilize the internal components. It is preferable to use machines with this capability especially for COVID + patients and PUI. An alternative to consider is to sterilize internal components in accord with the manufacturers’ procedures and then place a high quality viral filter on the inspiratory limb. See https://www.asahq.org/-/media/files/spotlight/anesthesia-machines-as-icu-ventilators-5-07.pdf for additional information.
How can the internal parts of the machine be sterilized?
Short Answer: Follow the manufacturer’s recommendations.
Manufacturer’s all provide reference information and customer support contacts to answer questions on cleaning procedures for specific devices.
References:
- GE HEALTHCARE: 24×7 phone support (800) 345-2700
- DRAEGER Medical: (800) 437-2437
- MINDRAY: (800) 288-2121 or (877) 913-9663
- GETINGE
Can an anesthesia machine be used as an ICU ventilator?
The APSF and ASA have collaborated on guidance for utilizing anesthesia machines as ICU ventilators. Click here: https://www.asahq.org/ventilators
Short Answer: Anesthesia machines generally have highly capable ventilators that should meet the needs of a large majority of patients with respiratory disease requiring mechanical ventilatory support. Anesthesia machines are not typically approved for use as ICU ventilators but during the pandemic, the US FDA has provided Emergency Use Authorization. Manufacturers have also provided guidance on using their devices for this purpose. Anesthesia professionals should provide consultation and support to critical care teams that will not be familiar using and maintaining anesthesia machines.
What are the recommendations for transporting intubated patients to and from the operating room?
Short Answer: Keep a high-quality HMEF or filter on the airway at all times. Clamp the endotracheal tube whenever it is disconnected from the filter to maintain a closed system. Mapleson D systems can eliminate concern about dead space imposed by the filter if the filter is placed on the device side of the fresh gas flow.
Patient transport is a common part of anesthesia practice and will almost certainly be required when COVID + patients require an urgent procedure in the operating room. Institutions should have local guidelines for the preferred transport routes and use of PPE for the patient and health care workers during transport. Intubated patients will require continuous ventilation during transport. Transport ventilators and self-inflating resuscitation bags should be used with a high-quality viral filter mounted at the airway. Ventilators typically exhaust exhaled gas to the immediate environment so added protection would include placing a high-quality viral filter at the end of the expiratory limb.
For patients where the deadspace imposed by the filter is a concern, a Mapleson D type transport circuit eliminates concern about the impact of additional deadspace IF the filter is placed on the device side of the fresh gas flow inlet and fresh gas flow is set to exceed minute ventilation.
How can the endotracheal tube be safely disconnected from the anesthesia breathing circuit for transport, suctioning, or nebulizer therapy?
Short Answer: When initiating the transport process, leave the viral filter in place on the endotracheal tube and connect the filter to the transport ventilation device. Use in-line suction and nebulization devices. Clamp the endotracheal tube whenever it is disconnected from the filter to maintain a closed system.
Once a “high-quality” viral filter is in place on the endotracheal tube, the breathing circuit can be disconnected from the filter while moving a patient or to reconnect to a different ventilation device. The filter will reliably stop viruses exhaled by the patient from contaminating the room.
Suctioning the endotracheal tube or administering nebulized medications is a different problem. If suctioning is anticipated, a closed suction system (like those used in intensive care) should be placed between the endotracheal tube and filter as soon as the endotracheal tube is inserted. In line nebulizers will need to be placed on the patient side of the filter to deliver medication effectively. If closed suction is not already in place or an inline nebulizer adapter is needed, the ventilator should be paused and the endotracheal tube clamped whenever it is disconnected from the HMEF.
We are running out of laryngoscope/GlidescopeTM blades. How do we clean and sterilize them?
We do not have guidance on individual laryngoscope processing or high level disinfection processes. To our knowledge, none of the four major U.S. manufacturers of disposable videolaryngoscope blades (covers) recommend cleaning or sterilization of their disposables for re-use. However, a number of U.S. medical centers are using various techniques to process and re-use these covers. ASA and APSF both recommend the use of videolaryngoscopes instead of typical laryngoscopes in an effort to keep the faces of anesthesia professionals further away from the airways/mouths of their patients during this pandemic. These medical centers that are re-processing their disposable videolaryngoscopic blades are trying to conserve blades in anticipation of shortage because of limited supplies or supply maldistribution.
Below are several examples of techniques used by medical centers to process blades for re-use. Neither ASA nor APSF endorse any specific company or technique; these examples are provided only for information.
- Some medical centers use techniques commonly used to clean endoscopes (e.g., http://www.medivators.com/products-and-services/endoscope-reprocessing). This equipment type can be thought of as a super medical dishwasher. A number of centers that have used this technique have reported that they have not seen damage to any of their disposable blades.
- Some medical centers use vaporized hydrogen peroxide (e.g., https://www.steris.com/healthcare/knowledge-center/sterile-processing/hydrogen-peroxide-sterilization). This is a low temperature sterilization process. A number of centers that have used this process also have reported no damage to any of the disposable blades.
- Some medical centers are using manual cleaning and autoclaves (i.e., https://www.cdc.gov/infectioncontrol/guidelines/disinfection/sterilization/steam.html) and have reported that they have not seen damage to their disposable blades.
To conserve videolaryngoscopy equipment, remember that routine equipment can be used with patients with Mallampati Class 1 and 2 straightforward appearing airways. Regardless, please refer to CDC and FDA regulations at: https://www.cdc.gov/infectioncontrol/guidelines/disinfection/reuse-of-devices.html
We also recommend that you contact the manufacturer for cleaning guidance in the face of shortages. You may also wish to review recent recommendations on Infection Prevention in the Operating Room Anesthesia Work Area from the Society for Healthcare Epidemiology of America (SHEA).