To the Editor

Figure 1. Large linear crack in intravenous acetaminophen vial.
A 59-year-old female was scheduled at our outpatient surgery center for a left breast lumpectomy for recurrent breast carcinoma. Our anesthetic plan included the intravenous administration of acetaminophen (Ofirmev®) as a pain adjuvant. Acetaminophen was obtained from the Pyxis® unit. Upon inspection of the acetaminophen bottle, a white powdery substance was noted to be adherent to the outside of the bottle and 2 linear cracks spanning the length of the vial were observed (Figure 1). In 2 unrelated incidents, previous to this, a cracked bottle of ketamine and a shattered bottle with spilled acetaminophen were found in 2 other Pyxis drawers (Figures 2 and 3).
A thorough literature search was conducted but few studies or reports were found concerning medication safety, packaging, or transportation and broken or cracked vials. These reports describe broken vials after obtaining access with a pressurized syringe and coring of the rubber stopper with the risk of embolization after intravenous administration.1,2,3 The integrity of glass vials recently came to the attention of the FDA and another manufacturer, Hospira. Subsequently, Hospira has recalled a lot of lidocaine vials in 28 states. The recall was initiated for a reddish-orange particulate found on the inner surface and floating in the solution thought to be related to a supplier’s glass defect.5,6
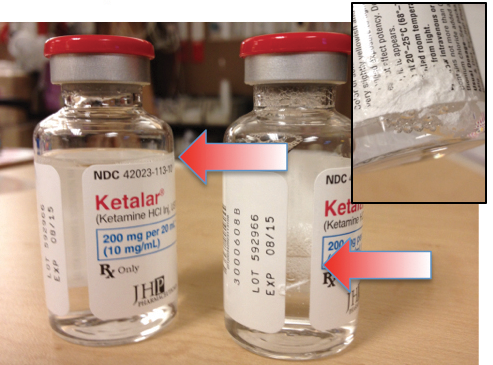
Figure 2. Diminished level of medication without any visibile cracks. Inset—White powdery substance adherent to outside of vial.
We hypothesized on possible mechanisms for the occurrence of bottle breakage: glass bottle manufacturing defect, transport damage (manufacturer to pharmacy and pharmacy to Pyxis®), and damage from repeated opening and closing of Pyxis® drawers. After discussion with our pharmacy, we learned Ofirmev® bottles are securely padded when shipped and that large cracks, when removing the bottles from the packaging or placing them in the Pyxis® drawers, have not been noted. As the lidocaine reference above suggests, defective manufacturing processes of glass vials can compromise medications in vials or lead to breakages similar to our recent findings. We have discussed these incidents with Ofirmev’s® manufacturer Cadence Pharmaceutical’s quality associates; they state cracks in the glass allow oxygen to enter the vial turning the solution yellow and permit possible particulate matter formation. Furthermore, the vials are inspected prior to shipment and any subsequent cracks are usually incurred during shipment of the medication.
In these cited cases, we report macroscopic bottle damages that compromised our medications’ sterility. These findings should make end-users aware of the possibility of microscopic bottle damage that may contribute to medication compromise and potential patient harm. One would surmise that the breakage of medication vials is more likely with more fragile glass vials, especially with pre-scored tops, than with these larger, sturdier glass bottles. However, as illustrated above, these larger bottles are also susceptible to breakage.

Figure 3. Shattered vial of intravenous acetaminophen.
Although medication safety and sterility should always be on the minds of anesthesia providers, The Joint Commission’s (TJC) recent emphasis on the strategic importance of medication management (which includes medication security, safety, and integrity) reconfirms our need for vigilance. In recent TJC medical facility accreditation site visits, one of the most common areas of non-compliance and, therefore, reasons for citation, is the secure storage of medications.4 To comply with this rule, many facilities are installing automated medication management systems in their operating rooms. Our experience underscores the need for pharmacy personnel to be attentive to the details of medication storage in these units. Because drawers in these units are accessed numerous times on a daily basis, the significance of medication alignment is obvious; proper attention and consideration to medication placement and alignment by the pharmacy staff during stocking should occur. TJC also states that the integrity of medications must be safeguarded.4 Thus, it is not only the pharmacists’ responsibility, but also that of all anesthesia providers to inspect bottles and vials for microscopic damage prior to medication administration. As we observed, a breach of a medication’s integrity may first be suspected by noting that a new medication vial is only partially full (In Figure 2, note the diminished medication level in the unopened ketamine vial). Equally concerning is the potential risk, primarily for anesthesia providers, of these cracked vials rupturing or splintering in their hands and cutting them when they attempt to spike or access a cracked bottle prior to medication administration.
With the assistance of our operating room pharmacists, we reconfigured our medication drawers to reduce medication movement during opening and closing of Pyxis® drawers. In addition, we have instructed all anesthesia providers to carefully inspect medication vials and bottles prior to medication administration. Confirming all medications’ integrity prior to administration is an important step we can and should take to provide safe anesthesia to all our patients. In doing this, we exemplify vigilance, the hallmark of safe patient care.
Andrew Crabbe, MD Thomas Tinker, MD Alberto DeArmendi, MD University of Oklahoma College of Medicine Oklahoma City, OK
References
- Chaudhry T, Serdiuk A. Coring and fragmentation may occur with rubber cap and blunt needles. ASPF Newsletter 2013; 27:68.
- Herd RS. Coring observed with large bore beveled needle. ASPF Newsletter 2013; 27:68.
- Moss D, Schumann R. Unusual complications and potential safety hazard from simply drawing up a medication in the operating room. Anesth Analg 2013; 116(2):504.
- Keinle P, Uselton, JP. Maintaining compliance with Joint Commission medication management standards. Patient Safety and Quality Healthcare [Internet]. 2008 July/August [cited 2013 Nov 30] Available from: http://www.psqh.com/julaug08/medication.html.
- Brooks M. Lidocaine injection recalled in 28 states [Internet]. 2013 December 24 [cited 2013 Dec 30] Available from: http://www.medscape.com/viewarticle/818299.
- Hospira issues a voluntary nationwide recall of one lot of lidocaine HCl injection, USP, 2%, 5 ml in 5 ml vial, due to presence of particulate matter [Internet]. Silver Springs (MD) 2013 Dec 23 [cited 2013 Dec 30] Available from: http://www.fda.gov/Safety/Recalls/ucm379739.htm.


 Issue PDF
Issue PDF