To the Editor
We would like to report 2 look-alike medications. The purpose is to decrease the likelihood that accidental epidural injection by similarly packaged medications. The medications were local anesthetic vials of lidocaine HCl 2% with epinephrine 1:200,000 for “infiltration and nerve block including epidural and caudal”; and lidocaine HC1 2% with epinephrine 1:100,000 for “infiltration and nerve block” (NOT FOR EPIDURAL OR CAUDAL USE).
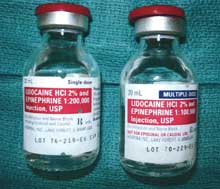
Read the fine print! Vial on left is preservative free local anesthetic approved for neuraxial anesthesia; look-alike vial on right is the same local anesthetic with methylparaben preservative.
These 2 look-alike medications were found in the labor and delivery suite of our institution where they are used typically for cesarean sections. The primary difference in the medications was the “multiple dose” indication for the lidocaine 2% with epinephrine in the 1:100,000 concentration. The preservative in this vial was methylparaben 1 mg.
Although the manufacturer lists the medications as separate for indications, it is interesting to note that methylparaben as a preservative has not been suggested to be non-toxic when given spinally in small doses.”1,2
A concern over accidental epidural use of the “multiple dose” vial with the methylparaben preservative for epidural analgesia is what initiated this letter. Our hope is that other anesthesiologists and providers will be made of aware of these differences.
Sincerely,
David Kim MD
Ihab Kamel MD
Temple University School of Medicine
Philadelphia, PA 19140
References
-
Hodgson PS, Neal JM, Pollock JE, Liu SS. The neurotoxicity of drugs given intrathecally (spinal). Anesth Analg 1999;88:797-809.
-
Gurun MS, Leinbach R, Moore L, Lee CS, Owen MD, Eisenach JC. Studies on the safety of glucose and paraben-containing neostigmine for intrathecal administration. Anesth Analg 1997;85:317-23.


 Issue PDF
Issue PDF