“Mother dies when given two ampoules of Adrenalin in mistake for Pitocin.”
This 1984 report of an anesthesia fatality highlighted the problem of poor legibility of many labels (Fig. 1). Not only is the lettering small but it is printed on clear glass. Thus, legibility depends upon having good lighting and a contrasting background.
The solution to this legibility problem is contained in ASTM Standard D4267 which had been published in 1983. This requires that the drug’s name and dosage be legible at arm’s length in the dimmest lighting permitted in hospital corridors (20 ft. candles). As the new D4267 standard labels have been adopted (Fig. 2) them has been a marked improvement in legibility.
Anesthesiologists Boardman Wang and Herman Turndorf of New York University in the 1970’s had publicized that up 12% of doses of drugs administered in hospitals could be in error. After failing to get action from the FDA, PMA, AMA, or ASA, in 1981 a group consisting of anesthesiologists, nurses, pharmacists, manufacturers, and FDA label experts formed Subcommittee DIO.34 of the American Society of Testing and Materials (whose subcommittee DIO.32 had previously produced the child resistant medication closure). The objective was to eliminate as many hazards as possible by enhancing the legibility of the labels and coordinating the use of colors.
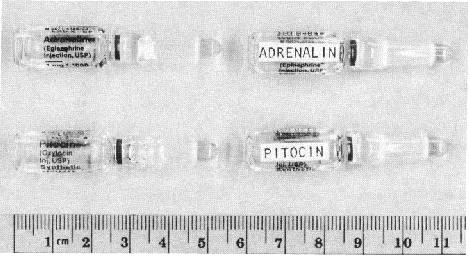
Figure 1 The poorly legible Adrenalin and Pitocin labels (left), contrasted (right)’With the bold print on an opaque background required by ASTM Standard D42671983.
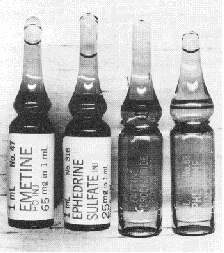
Figure 2 The new D4267 standard labels for emetine and ephedrine are clearly legible in contrast to the old labels.

Figure 3 Samples of the standard user-applied syringe labels. When seen in color, the yellow thiopental label is clearly different from the fluorescent red of the muscle relaxants. Succinylcholine is printed with a distinctive black surround to separate it from the other relaxants. The antagonists have stripes of the same color as their agonists red for neostigmine, blue for naloxone and violet for nitroglycerine
“Patient paralyzed by succinylcholline given by anesthesiologist before induction of anesthesia in mistake for Sublimaze.”
In the absence of an understanding for the coherent use of colors and generic drug names, the syringe labels used in this hospital for these two drugs were of similar color and the printed names had similar outlines. The anesthesiologist concerned has since become a strong supporter of the proposed standard’s use of different bold colors to separate the various drug groups from each other (Fig. 3). In this system, labels for thiopental and other induction agents are yellow, muscle relaxants are fluorescent red (succinylcholine is distinguished by a distinctive black surround), fentanyl and other narcotics are light blue, tranquilizers are orange, vasopressors are violet, local anesthetics are gray, atropine and other anticholingeric agents are green, and all other drugs are printed in black on a white background.
Prefilled syringe chaos
Another problem is that emergency drug-filled syringes, intended for use under difficult conditions when the prompt injection of the correct drug may save a life, all look alike and their labels are poorly legible. The proposed standard labels using bold print 5 to 7 mm high on an opaque background permit the drug’s name and dosage to be read at 5 feet, even through the plastic barrel of the syringe (Fig4)
“Nurse flushes Heplock with potassium chloride in mistake for sodium chloride kills patient.”
Manufacturers have enthusiastically adopted colored snap-off caps for their vials as an aid in identifying the various drugs they supply. However, the manufacturers’ color codes differ. So the system only works if a hospital buys all its preparations from a single manufacturer. Any change of supplier may end in tragedy as in this case where the new potassium chloride vials had the same colored cap and a similar label to the former supplier’s sodium chloride vials.
The solution proposed by the subcommittee is for drugs that must be diluted for safe use to be fitted with a distinctive black metal closure with a black or natural plastic cap, both to be inscribed “Dilute Before Use” or similar instructions. Even in a drug tray as disorganized as that shown in Fig. 5, these vials would readily be identifiable. Ampoules containing drugs to be diluted before use would be distinguished by a broad black band above the neck of the ampoule
By taking multiple bites out of this problem, it is hoped to reduce the chance a user will make a mistake. However, there still remains the manufacturers’ tendency to adopt labels designed to identify clearly all their own products rather than using different designs for each group of products. This makes the user’s task unnecessarily difficult and stressful. Just as it would be unreasonable to have all road signs octagonal in shape with red backgrounds expecting the user to pick out the STOP sign from a mishmash of other logos so the manufacturer should follow the safety engineer’s lead and differentiate between groups of products. This would help the fatigued user at the end of 24 hour emergency call to use outline shape, color and bold text at least to differentiate safely between Adrenaline and Pitocin.
Leslie Rendell-Baker, M.D. is Professor of Anesthesiology, Lcma Linda (CA) University and a long time activist for safer anesthesia apparatus
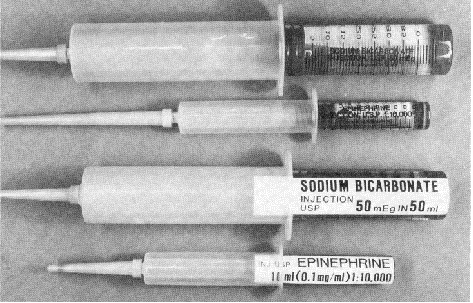
Figure 4 -‘Me poor legibility of the existing pre-filled syringe labels is in stark contrast to the proposed standard labels that can even be read through the plastic syringe barrel at 5 feet.
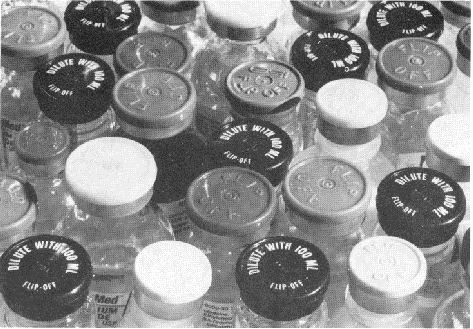
Figure 5 The drugs which must be diluted for safe use with their black snap-off caps and closures can readily be distinguished even in this mixed-up tray.

