Report on the 7th Invited Conference by Cardinal Health
June 7-8, 2007, Center for Safety and Clinical Excellence, San Diego, CA
An invited conference sponsored by Cardinal Health Center for Safety and Clinical Excellence was held June 7-8 in San Diego, to review and summarize expert opinion on tight glycemic control (TGC) for acute hospitalized patients. Speakers included Simon Finfer, MBBS (Nice-SUGAR trial, Sydney, Australia) and Philippe Devos, MD (VISEP/Glucontrol trials, Belgium), with another 45 participants from across the United States and Canada. Thought leaders represented the disciplines of anesthesiology, intensive care, endocrinology, surgery, hospitalist medicine, medical genetics, nutrition, nursing, pharmacy, biostatistics, and healthcare biotechnology.
The central question, which anchored the conference, was whether ICU patients benefit most from “intensive” or “tight” glycemic control (usually defined as blood glucose in the range of 80-110 mg/dL) or “tighter” control (typically translated as blood glucose in the range of 110-150 mg/dL).
In the meantime, conference presentations yielded these highlights:
- Insulin is the most common drug reported as a medication error to the U.S. Pharmacopeia. Moreover, the error harm rate (categories E, F, H, I) = 9.3%.
- Data from hospitals using “smart” IV pumps documented a high frequency of averted insulin dosing errors in addition to high variability in concentrations and mixed use of weight-based and nonweight-based dosing units.
- The landmark ICU study by Dr. Greet Van den Berghe, Catholic University of Leuven in Belgium, targeted blood glucose of 80-110 mg/dL in the treatment group. She noted decreased mortality and morbidity (less renal failure, surgical wound infections, blood transfusions, ICU ventilator days, etc.) in surgical ICU patients.
- Two recent European glucose control studies—VISEP (488 patients in 17 German Centers) and GluControl (1,101 randomized patients across 21 ICU units in 7 European countries)—were both stopped due to unacceptably high rates of hypoglycemia and lack of beneficial effect.
- The on-going NICE-Sugar open-label, randomized stratified study in 25 Australian, 19 Canadian, and 2 American hospitals has a planned enrollment of 7,000 patients! This study will compare 2 target ranges for blood glucose (81-108 mg/dL vs. <180 mg/dL). Conference participants expect this to be the pivotal outcome investigation—and one that will likely define the target glucose for future ICU patients. The study is well on its way to 4,000 enrolled patients.
- Current data are insufficient to mandate TGC for patients in the operating room. A recent randomized study in cardiac surgery patients (Ann Int Med 2007;46:233) found no difference in ICU or hospital LOS despite TGC throughout the operative period.
- It is important to note that perioperative hyperglycemia occurring in “non-diabetics” may actually indicate undiagnosed Type II diabetes. Providers should consider hemoglobin A1c determinations in these patients to direct optimal metabolic management.
Additional insights based on the collective experience of participants included
- Prevalence of diabetes is increasing rapidly.
- TGC requires an interdisciplinary team approach, a culture of safety, and a focus on professional education. Moreover, benchmarks to evaluate effectiveness are needed.
- Current “paper” ICU protocols for TGC achieve target glucose concentrations about 40% of the time.
- Published TGC protocols differ significantly in insulin dosing recommendations.
- Computerized protocols improve the efficacy to 60%, and have the additional benefit of decreasing hypoglycemic episodes.
- Bedside glucometers rely on a number of different chemical reagents, and users should be aware of potential confounding variables—even including parameters like hematocrit, PaO2, and so forth. Interfering substances can generate seriously erroneous meter readings.
- Total nursing time for each point-of-care glucose determination varies from 3.5-9 min (median time = 4.7 min). The aggregate time and RN workload of applying TGC is substantial.
- Current continuous glucose monitors (CGM) measure glucose in the interstitial fluid, which lags behind blood concentrations by 3-10 min. Nonetheless, CGM appears to facilitate smoother, timelier titration of insulin infusions, and patients reach target glucose values more quickly. Coordination of this technology with a computerized protocol minimizes the incidence of hypoglycemia.
- Clinical experience suggests minimal risk of a single episode of hypoglycemia (FBS <= 40 mg/dL), if diagnosed and managed in a timely fashion.
- Transitions from ICUs to Medical/Surgical care units and from IV to oral feedings are especially problematic in maintaining TGC.
- Intensive insulin therapy and TGC is cost effective, but may be population and protocol sensitive.
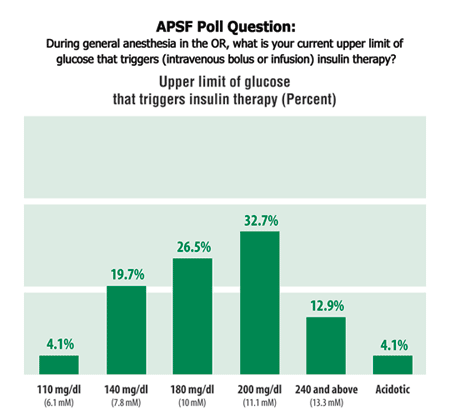
Results of independent APSF Poll regarding readers’ triggers for information of insulin therapy.
The optimal glucose threshold for TGC in ICU patients remains under investigation, and anesthesia providers debate how these principles should apply to patients in the operating room. More data are needed. An independent APSF poll regarding triggers for initiation of insulin therapy is also presented below.
Richard C. Prielipp, MD, MBA, FCCM
Professor and Chair of Anesthesiology
University of Minnesota
Minneapolis, MN
Carol S. Manchester, MSN, APRN, BC-ADM, CDE
Diabetic Clinical Nurse Specialist
UMMC-Fairview
Minneapolis, MN
Timothy W Vanderveen, PharmD, MS
Vice President
Cardinal Health Center for Safety and Clinical Excellence
San Diego, CA


 Issue PDF
Issue PDF