Recently, two colleagues wrote to the APSF that they had observed a discrepancy between end-tidal and arterial carbon dioxide tensions (PETC02 and PaCO2) in children during inhalation anesthesia. The table summarizes their data. It shows large PaCO2-PETC02 gradients during general anesthesia with use of a mask, and quite normal gradients once the children had been intubated and their lungs mechanically ventilated.
The colleagues wondered why the capnometer gave such misleading information during inhalation anesthesia with mask and bag. They also asked whether hypercarbia, as observed by them during induction of anesthesia in children, has deleterious effects.
The measurement of carbon dioxide in respired gas has its vicissitudes. A brief review brings things into focus.
It is often recommended to sample gas for analysis as close to the mouth as possible. Indeed, sampling gas in the airway itself would be ideal. After all, we want information about alveolar gas, because that Ls the best way to represent arterial gas.
Of the exhaled gas, that at the end of a large tidal volume best represents alveolar gas. The first part of the exhalation will come from the mouth and trachea and bronchi, that is from dead space that will have been filled with the last part of the preceding inspiration. We can increase this dead space by putting a mask over the patient’s face. With a large dead space and a small tidal volume, the end-expired gas may not contain much or even any alveolar gas.
Anesthesia maneuvers that increase dead space and at the same time decrease ventilation and tidal volume will not only prevent us from detecting alveolar gas, they will also cause arterial carbon dioxide tensions to increase. The difference between P,,CO2 and POC02 will increase, just as it did in the cases reported above. Such findings are particularly likely in patients with relatively small tidal volumes. Furthermore, a leak around the mask will cause dilution of the expired C02 and will increase PaCO2-PETC02.
How can it be detected that there is not a good sample of end-tidal gas? Inspection of the capnogram provides clues. A normal capnogram has a box-like appearance [see figure (reduced dead space, normal tidal volume)]. If the transitions from phase 11 (the upstroke representing the appearance of carbon dioxide in expired gas) to phase III (the plateau), and from phase III to phase IV (the downstroke representing the appearance of fresh gas in inspired gas) are rounded [see figure (increased dead space, reduced tidal volume)], the tidal volume is likely to be too small to bring forth alveolar gas in the end-tidal gas. If phase III of the capnogram has not reached a plateau before it is interrupted by phase IV, expiration may not have run its course, and end-tidal C02 tensions will underrepresent arterial C02 tensions.
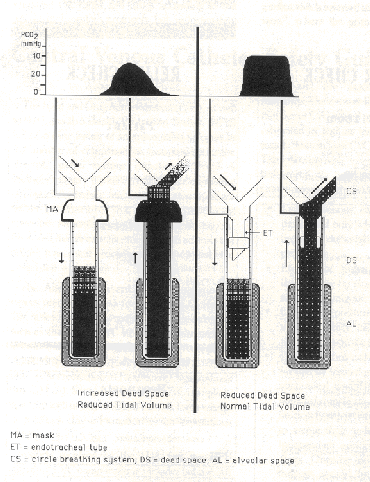
Diagram of depressed ventilation and increased dead space (left pair) and normal ventilation and dead space (left pair). At top are capnograms from breaths with reduced and normal tidal volumes, respectively Observe the box shape with plateau (phase III) of the normal capnogram on the right and the rounded shape of the abnormal capnogram on the left. End-tidal tensions of the normal capno8ram are greater than those of the abnormal capnogram.
The diagrams represent gas in the lung and in the Y-piece of a circle system. Of each pair the left shows inspiration, the right, expiration. Fresh gas (not shaded), deposited during inspiration, fills the anatomic dead space. Observe that the alveolar concentration of C02 is higher with increased dead space and reduced tidal volume.
A capillary taps into the gas column just outside the anatomic dead space and represents the site of collecting gas for capnometry. ]’he capillary points to the capnogram corresponding to the phase of ventilation. Observe that the site of collecting gas for capnometry sits on top of additional dead space due to a mask (left pair). For the right pair an endotracheal tube is depicted.
Because of the reduced tidal volume and the added dead space from the left pair, the high alveolar concentration of carbon dioxide never reaches the sampling capillary. The corresponding capno8ram, therefore, fails to reflect the fact that in this instance, alveolar (and, hence, arterial) C02 tension exceeds that of the right pair, even though end-tidal C02 tension is higher for the right pair than for the left pair.
Inspection of the capnogram, therefore, is essential during capnometry. Many older capnometers do not offer capno8rams for inspection; instead, they simply report the highest expired C02 tension in a digital readout. This explains why these so-called “end-tidal” tensions are quite different from arterial tensions at times.
Other disturbances can magnify the gradient between end-tidal and arterial PCO2 (see Gravenstein IS, Paulus DA, Hayes TI: Capnography in Clinical Practice, Stoneham, Massachusetts: Butterworths, 1989, available through the APSF office). However, in the pediatric patients described hem where improved ventilation and gas samp4ng corrected the problem, the simple mechanisms (increased anatomic dead space and shallow ventilation) are very likely the cause.
Is hypercarbia deleterious? In healthy patients the answer is a qualified no. As long as hypercarbia does not become excessive, increased arterial PCO2 is not dangerous. There are three concerns, though:
1. As the alveolar concentration Of C02 increases, the alveolar concentration Of 02 must decrease. On room air, hypoventilation leading to hypercarbia is hypoventilation leading to hypoxemia. However, an atmosphere enriched with oxygen can prevent the hypoxemia.
2. Hypercarbia may lead to increased sympathetic activity and, thus, set the stage for arrhythmias, for example during halothane anesthesia.
3. Hypercarbia raises brain blood flow. Any patient at risk of increased intracranial pressure should not be exposed to hypercarbia.
Is hypercarbia during routine induction of inhalational anesthesia in children avoidable? Probably, but perhaps not always. Premedication with inspiratory depressants, breath holding with induction of inhalation anesthesia, and respiratory depression with intravenous induction agents contribute to hypercarbia. Meticulous attention to the airway and assisted or controlled ventilation (as indicated) help maintain relative normocarbia. Once the endotracheal tube is in place, depressed ventilation can be corrected easily; at the same time, sampling of end-tidal gas for capnography becomes easier. Intubation dramatically reduces dead space, and tidal volume can be adjusted as desired based on the more correct end-tidal C02.
Dr. Gravenstein, in addition to co-authoring the book on capnography, is Graduate Research Professor of Anesthesiology at the University of Florida and a member of the APSF Executive Committee.

