Episode #175 Remimazolam, A New Medication on the Anesthesia Block, PART 1
November 7, 2023Welcome to the next installment of the Anesthesia Patient Safety podcast hosted by Alli Bechtel. This podcast will be an exciting journey towards improved anesthesia patient safety.
We are jumping into the October 2023 APSF Newsletter today. Our featured article is “Remimazolam: Patient Safety Considerations of a Novel, Practice-Changing Drug in Perioperative Medicine” by Arnoley Abcejo and Miguel Teixeira.
Check out Table 1 in the article for a quick Remimazolam guide. Here it is:
Table 1: Quick Reference Guide for pharmacology and dosing for remimazolam.
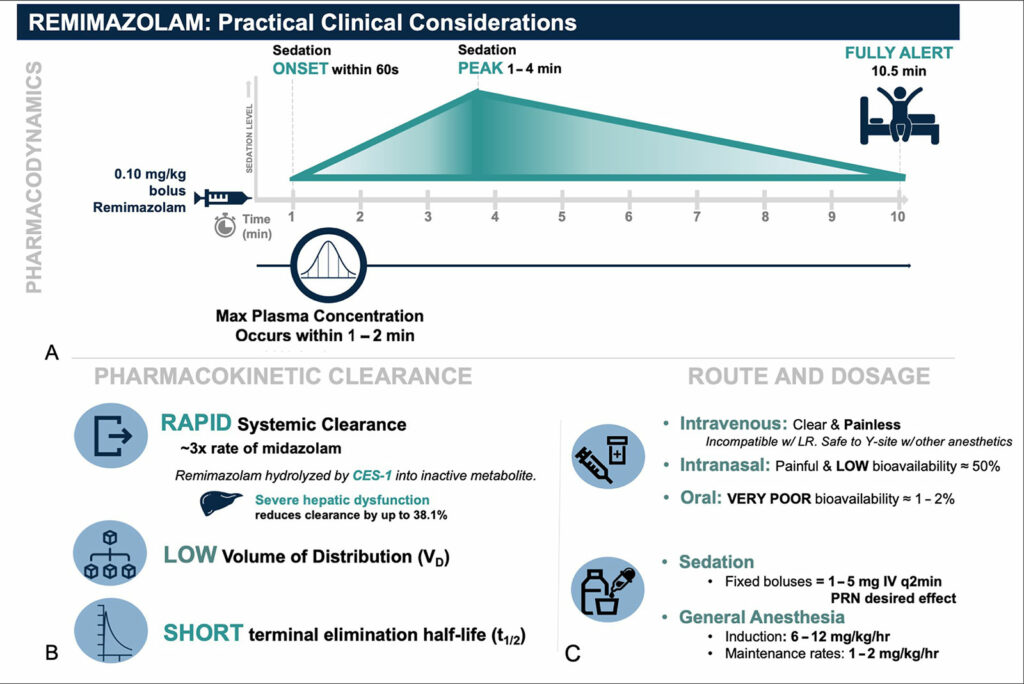
Figure 2: Remimazolam pharmacokinetic and pharmacokinetic profile adapted with permission from Figure 2 from Teixeira et al. “The role of remimazolam in neurosurgery and in patients with neurological diseases: a narrative review.” J Neurosurg Anesthesiol, May 31, 2023.
Abbreviations: mg = milligram, kg = kilogram, hr = hour.
Here are the protocols used at the Mayo Clinic:
- For procedural sedation: administration of 2 mg IV every 15 seconds as needed with or without analgesic adjuncts such as ketamine or opiates.
- For induction of general anesthesia: administration of a 0.2-0.4mg/kg induction dose followed by 1-2 mg/kg/hr.
Here are the citations that we talked about on the show today:
- Teixeira MT, Brinkman NJ, Pasternak JJ, Abcejo AS. The role of remimazolam in neurosurgery and in patients with neurological diseases: a narrative review. J Neurosurg Anesthesiol. 2023;doi:10.1097/ana.0000000000000917 PMID: 37264540Online ahead of print. https://journals.lww.com/jnsa/Abstract/9900/The_Role_of_Remimazolam_in_Neurosurgery_and_in.67.aspx
- Kimoto Y, Hirano T, Kuratani N, et al. Remimazolam as an adjunct to general anesthesia in children: adverse events and outcomes in a large cohort of 418 cases. J Clin Med. 2023;12 PMID: 37373624
Subscribe to our YouTube Channel here: https://www.youtube.com/@AnesthesiaPatientSafety
Be sure to check out the APSF website at https://www.apsf.org/
Make sure that you subscribe to our newsletter at https://www.apsf.org/subscribe/
Follow us on Twitter @APSForg
Questions or Comments? Email me at [email protected].
Thank you to our individual supports https://www.apsf.org/product/donation-individual/
Be a part of our first crowdfunding campaign https://www.apsf.org/product/crowdfunding-donation/
Thank you to our corporate supporters https://www.apsf.org/donate/corporate-and-community-donors/
Additional sound effects from: Zapsplat.
© 2023, The Anesthesia Patient Safety Foundation
Hello and welcome back to the Anesthesia Patient Safety Podcast. My name is Alli Bechtel, and I am your host. Thank you for joining us for another show. We are jumping into the October 2023 APSF Newsletter today. (Splash) We have a novel, possibly practice-changing show all about a novel, possibly practice-changing medication, Remimazolam.
Before we dive into the episode today, we’d like to recognize Merck, a major corporate supporter of APSF. Merck has generously provided unrestricted support to further our vision that “no one shall be harmed by anesthesia care”. Thank you, Merck – we wouldn’t be able to do all that we do without you!”
Our featured article is “Remimazolam: Patient Safety Considerations of a Novel, Practice-Changing Drug in Perioperative Medicine” by Arnoley Abcejo and Miguel Teixeira. To follow along with us, head over to APSF.org and click on the Newsletter headings. First one down is the Current Issue. Then, scroll down until you get to our featured article today.
Before we dive into the article, we are going to hear from one of the authors. Here he is now.
[Teixeira] “Hello, my name is Miguel Teixeira and I’m an anesthesiologist and intensivist at the Mayo Clinic in Rochester, Minnesota. Thanks so much for having me.”
[Bechtel] To kick off the show, I asked Teixeira what got him interested in this topic. Let’s take a listen to what he had to say.
[Teixeira] “At first, I heard about Remimazolam from some colleagues at work. I was immediately intrigued by its pharmacokinetics and pharmacodynamics. Fast onset, short acting, reversible. As a neuro-anesthesiologist and intensivist, I knew benzodiazepines as a whole had an appealing profile.
Favorable effects on respiratory drive and hemodynamics, and its effects on neurophysiology, even. Yet I actually rarely used them. Why? Well, as a whole, benzodiazepines had a slower rate of loss of consciousness and a longer context sensitive half-life compared to other available agents. Also, I was concerned about the potential deleterious neurologic recovery side effect profile.
So, with that in mind, together with some colleagues in the Division of Neuro-anesthesia, we started researching Remimazolam’s role specifically as it pertains to neurosurgery and in those patients with neurologic diseases.”
[Bechtel] Now, we’re all excited to learn more about this new medication.
Have you used Remimazolam before? Do you have it available at your institution? Remimazolam is an IV, short-acting, and ultrafast onset benzodiazepine that acts as a potent sedative-hypnotic, anxiolytic, anticonvulsant, and muscle relaxant. The name was designed to remind you of the therapeutic effects of midazolam and the unique metabolism of remifentanil.
Remimazolam has been used for procedural sedation in Asia and Europe since it was first released in China in 2019 for use in gastrointestinal endoscopy. The use has expanded to general anesthesia in Japan and Korea as well as IV sedation in Belgium. In July 2020 in the United States, the FDA approved remimazolam for induction and maintenance of sedation for adults undergoing procedures lasting 30 minutes or less. There are many reported off-label uses as well. Over the past 3 years, this drug has not been acquired by many institutions or used in a large clinical practice.
The authors share the experience at their institution, the Mayo Clinic, which is one of the first major academic centers in the United States to incorporate remimazolam in perioperative and periprocedural practice including over 5,000 patients and over 20,000 doses administered. We are going to take a journey through the literature and the clinical experience at the Mayo Clinic to discuss the unique pharmacokinetics and pharmacodynamics as well as the known limitations, adverse events, and contraindications. The authors also provide key clinical practice implications and help to identify existing knowledge gaps for safe, widespread adoption.
First up, let’s talk about the pharmacology. The mechanism of action is to enhance the gamma-aminobutyric acid type A (GABAA) inhibitory receptor leading to increased frequency of opening of ligand-gated chloride ion channels. Following administration, there is minimal cardiac or respiratory depression. When compared to Midazolam, there is faster onset and dose-dependent sedation and about half the potency for procedural sedation. The sedative effects can be reversed with flumazenil.
Table 1 in the article is a great quick reference guide for remimazolam which includes the pharmacology and dosing. I will include it in the show notes as well. Let’s go through it now. Remimazolam is a benzodiazepine with the following characteristics:
- Sedation time: 11-14 minutes
- Time of peak effect: 3-3.5 minutes following 1 dose and 11-14 minutes following multiple doses
- Half-life elimination: 37-53 min
- Metabolism is esterase dependent.
- Distribution: 0.76-0.98L/kg
- Excretion is via urine.
- Protein-binding is greater than 91%, primarily to albumin.
What all this means is that remimazolam has a relatively high clearance, a small steady-state volume of distribution, shorter elimination half-life, and a short context sensitive half time compared to other benzodiazepines or propofol. This medication is highly bound to protein and metabolized by liver carboxylesterase before being excreted in the urine. Remimazolam is water-soluble. After dilution into a solution, it is painless when injected. Remimazolam is most soluble in slightly acidic solutions and can precipitate in lactated or acetated Ringer’s solution. Check out figure 1 in the article which depicts remimazolam precipitation in plasmalyte. You can administer Remimazolam through y-site co-administration with other common anesthetic medications including remifentanil, fentanyl, dexmedetomidine, midazolam, rocuronium, and vecuronium. The remimazolam approved in the United States is ByFavo which is prepared in a 20mg powder vial which is meant to be drawn up into 8.2ml sterile 0.9% sodium chloride which results in 2.5mg/ml after being reconstituted. The FDA labeling recommends the following:
Inject 2.5-5mg over one-minute followed by supplemental doses of two 1.25-2.5mg doses IV over a 15-second time period after at least two minutes.
The authors share their experience for procedural sedation which includes administration of 2mg IV every 15 seconds as needed with or without analgesic adjuncts such as ketamine or opiates. Induction of general anesthesia involves the administration of a 0.2-0.4mg/kg induction dose followed by 1-2 mg/kg/hr.
There is another great reference in the article, Figure 2, Practical Clinical Considerations. Don’t worry I will include it in the show notes, and we will go through it now.
First, the pharmacodynamics. Following administration of a 0.1mg/kg bolus of remimazolam, onset of sedation is within 60 seconds, maximum plasma concentration occurs within 1-2 minutes, and peak sedation lasts for 1-4 minutes with the patient being fully alert in 10.5 minutes.
Next, pharmacokinetic clearance. There is rapid systemic clearance that is three times faster than midazolam. For bonus points, remember that remimazolam is hydrolyzed byCES-1 into inactive metabolite, and severe hepatic dysfunction will reduce clearance by about 38%. There is a low volume of distribution and a short terminal elimination half-life.
Now, let’s do a quick review of route and dosage. Intravenous is clear and painless, but it is incompatible with lactated ringers and safe to administer by y-site with other anesthetics. Intranasal is painful and has a bioavailability of about 50%. Oral route has very poor bioavailability of only 1-2%. For sedation, fixed boluses may be administered in 1-5mg IV every 2 minutes as needed for desired effect. For general anesthesia, induction doses range from 6-12 mg/kg/hr with maintenance rates of 1-2 mg/kg/hr.
We are going to switch our focus from clinical use of remimazolam to discuss the unknown patient safety considerations. This is not a medication with a long track record of use. It is a new medication on the scene. Fortunately, it appears to be a relatively safe medication, but we probably do not know that full impact of this medication on the clinical outcomes after use for specific surgeries or procedures or for specific patient populations. It is so important that any unexpected serious adverse events following remimazolam administration are reported to help improve our understanding of this novel medication in clinical practice…and to help keep patients safe. The authors provide a list of important patient safety considerations or questions that we need to figure out going forward. Let’s review it now.
- Recovery in neurologically vulnerable patients:
- We know that benzodiazepine may increase the risk for perioperative delirium and may need to be avoided for neurologically vulnerable patients, especially the elderly. Postoperative delirium following remimazolam administration only has not been extensively studied and the results may not be generalizable to larger populations or procedure types. At this time, we just do not know what the relationship is between remimazolam administration and long-term postoperative neurocognitive disorder.
- Check out the most recent literature on remimazolam published this year in the journal of neurosurgical anesthesiology by Teixeira and colleagues. Don’t worry, I will include a link in the show notes as well.
- Adverse reactions in specific patient populations and surgical subtypes:
- The pharmacokinetic properties of remimazolam are not significantly altered in elderly patients or patients with higher ASA scores. Keep in mind that according to the FDA recommendations, you may need a slight decreased dose for these patients as well as a reduced dose for patients with severe hepatic impairment as evidenced by a child-Pugh score greater than or equal to 10 due to the concern or decreased drug clearance. For remimazolam, you do no need to change the dose for patients with severe kidney disease. Currently, there are no recommendations for administration for pediatrics, but you can find off-label case reports of remimazolam as an adjunct for general anesthesia in the literature. There are also no case reports of the use of this medication in pregnant patients. If you take care of pediatric patients, we hope that you will check out the June 2023 article in the Journal of Clinical Medicine, “Remimazolam as an Adjunct to General Anesthesia in Children: Adverse Events and Outcomes in a Large Cohort of 418 Cases” by Kimoto and colleagues.
- Spoiler alert the authors of that study conclude that remimazolam administration to pediatric patients as an adjunct to general anesthesia may be associated with hemodynamic variability which may require treatment as well as a rapid return to responsiveness and ability to meet discharge criteria. The authors leave us with a call to action that large, multicenter trials are needed to learn more about remimazolam administration safety for pediatric patients. I will include the link in the show notes as well.
We still have so much more to talk about when it comes to what we don’t know about remimazolam as well as what we do know about adverse reactions and contraindications, and clinical implications. It promises to be another exciting remimazolam show and we are going to hear from another APSF author who was responsible for this great article. It’s a show you don’t want to miss.
Before we wrap up for today, I also asked Teixeira, “What’s next for your research on remimazolam?” Let’s take a listen to what he had to say.
[Teixeira] “So far, here at the Mayo Clinic, we have amassed over 20, 000 uses of remimazolam in over 5, 000 patients. The vast majority of these are for procedural sedation, but we’ve also used remimazolam for the induction and maintenance of general anesthesia. I want to retrospectively review the key clinical outcomes, including adverse events.
We then hope to analyze their data in a way that might help other centers. I’m also interested in remimazolam use for those with very poor cardiopulmonary reserve and as an agent to treat refractory status epilepticus.”
[Bechtel] Thank you so much to Teixeira for contributing to the show today and we are very excited about your future research and learning more about remimazolam.
If you have any questions or comments from today’s show, please email us at [email protected]. Please keep in mind that the information in this show is provided for informational purposes only and does not constitute medical or legal advice. We hope that you will visit APSF.org for detailed information and check out the show notes for links to all the topics we discussed today.
The all-new October 2023 APSF Newsletter is here! We are so excited to continue to talk about the new articles with exclusive content from the authors right here on this podcast in the upcoming weeks!! In the meantime, we hope that you check out the new newsletter. For more anesthesia patient safety content all week long, we hope that you will follow us on twitter, @APSForg or you can like and share our posts on Instagram and Facebook. Before you know it, the next podcast episode will be ready to drop!
Until next time, stay vigilant so that no one shall be harmed by anesthesia care.
© 2023, The Anesthesia Patient Safety Foundation


