ASA and APSF Statement on Perioperative Testing for the COVID-19 Virus
June 15, 2022
Original December 08, 2020
Updated June 15, 2022


Patients who are infected with SARS-CoV-2, the virus responsible for the COVID-19 disease, have higher perioperative morbidity and mortality.1-5 Risk factors associated with SARS-CoV-2 infection that have been reported to increase perioperative morbidity and mortality include advanced age and other comorbities including higher ASA Physical Status, cardiac risk, and immunocompromise to list a few. Unexpected progression to acute respiratory distress syndrome, cardiac injury, kidney failure, and even death has been observed in patients infected with SARS-CoV-2 who have undergone surgical procedures.1,6-8 Additionally, aerosolizing procedures place operating room staff at greater risk of being infected with SARS-CoV-2. As a result, a robust screening and testing program to detect SARS-CoV-2 is essential for the safety of patients, health care workers, and the general public.
Screening for SARS-CoV-2 via careful symptom history is important, yet imperfect.9,10 There is high inter-patient variability in disease presentation and symptom severity. Screening should include an assessment of:11
- Close contact to someone diagnosed with COVID-19 in the past 10 days and
- Unexplained fever, cough, shortness of breath, chills, muscle pain, headache, sore throat, rhinorrhea, and/or new loss of taste or smell, nausea, vomiting, or diarrhea, have been reported.
The ability of testing to detect SARS-CoV-2 is dependent on sampling technique, fluid sampled, sampling site, the test performed and the timing of the test relative to the infectious course.12 The reported sensitivity of SARS-CoV-2 testing using polymerase chain reaction (PCR) testing is approximately 95 percent, meaning that some infected patients will be erroneously reported as free of the virus.13 Viral transmission may occur up to three days before patients become symptomatic. Healthy adult patients who have had a mild or moderate case of COVID-19 typically do not have replication-competent virus beyond 10 days following symptom onset. Viral particles may be detected by PCR for up to three months following these infections; however, these particles are presumed to be noninfectious.14 Importantly, replication-competent virus has been identified between 10 and 20 days post-symptom onset in patients who have had a severe case of COVID-19 or who are immunocompromised, and beyond 20 days in moderate to severely immunocompromised patients regardless of symptom severity.
The Centers for Disease Control and Prevention (CDC) guidance on discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings January 14, 2022 Update14 advises that symptom-based transmission-based precautions may be discontinued by health care facilities in patients with mild to moderate illness who are not severely immunocompromised once 10 days have passed since symptom onset, 24 hours have passed since last fever without the use of anti-pyretic medications, and improvement in symptoms. Time-based transmission-based precautions in symptomatic patients who have severe illness can have their transmission-based precautions discontinued when at least 10 days, and up to 20 days, have passed since symptom onset, 24 hours have passed since last fever without the use of anti-pyretic medications, and there is improvement in symptoms. Time-based transmission-based precautions in asymptomatic patients testing positive may be discontinued by healthcare facilities after 10 days. However, if symptoms develop after a positive test, day 0 changes to the first day of symptoms. Retesting patients is no longer a universally recommended approach for determining when someone is no longer infectious, keeping in mind the potential prolonged course of viral shedding. The exception are patients who are moderately to severely immunocompromised (regardless of COVID-19 symptoms or severity) for whom the CDC recommends a test based strategy.14 In addition, the CDC recommends consideration of a test-based strategy for patients who are severely ill and are not moderately to severely immunocompromised.
Antibody testing does not have a role in perioperative screening and risk stratification. Antibodies develop in the second week of symptoms and not all patients who are infected with SARS-CoV-2 develop detectable antibodies.15,16 Additionally, antibody tests have the potential of cross-reaction with other coronaviruses, resulting in false-positive results.16,17 As a result, antibody testing should not be performed during routine preoperative screening.
The Anesthesia Patient Safety Foundation (APSF) previously published and updated examples of SARS-CoV-2 preoperative testing protocols from around the U.S.18
Many institutions continue to require COVID preoperative testing for all patients, including those who have been fully vaccinated. Such concerns are expressed when the prevalence of new and evolving variants is increasing in the institution’s communities, in addition to CDC reports of breakthrough infections in fully vaccinated, asymptomatic individuals with waning immunity who have the potential to transmit SARS-CoV-2 to others, as well as the potential for reinfection. Therefore, we continue to recommend that unless the criteria listed below are met (e.g., low to moderate community transmission) that all patients undergoing an anesthetic, procedure, or surgery with the potential to generate aerosols should continue to have preoperative PCR testing for SARS-CoV-2, ideally not longer than three days prior to the procedure.
Recommendations:
We acknowledge that the science regarding pre-procedural testing for SARS-CoV-2 has yet to fully mature. However, today, healthcare organizations are faced with real decisions regarding the appropriateness of testing for SARS-CoV-2. We offer the following recommendations to facilitate this decision making.
A population risk assessment identifying the prevalence of SARS-CoV-2 should be reviewed. It is plausible that SARS-CoV-2 will become endemic and we will have to learn to adapt to various levels of community transmission. As we enter this next phase we recommend the following when there is local or regional community transmission19,20 of SARS-CoV-2:
- All patients should be screened for symptoms and exposures prior to presenting to the health care facility. Patients reporting symptoms should be referred for additional evaluation. Unless the exceptions listed below (number 3) are met, all other patients should undergo nucleic acid amplification testing (e.g., PCR tests) prior to undergoing non-emergent surgery.
- If a patient tests positive for SARS-CoV-2, elective surgical procedures should be delayed per our recent statement on elective surgery after a patient has COVID-19.7
- We continue to review updated information concerning the increased transmissibility, the potential risk of longer symptoms (Long COVID) and postoperative complications from the evolving variants of SARS-CoV-2.7 The updated recommendations below should be reviewed along with previous recommendations to operationalize a robust and safe perioperative screening and testing program for our patients, our healthcare workers, other hospital patients and the general public.
- Population risk assessment data is now available via two metrics from the CDC data tracker: Community Levels and Community Transmission. Healthcare facilities should utilize the Community Transmission metric (based on cases/100K in the last 7 days and % test positivity in the last 7 days to evaluate their perioperative testing strategy.21 (see table)
- Low to moderate community transmission: If the patient is asymptomatic, is up-to-date in vaccination and is having a lower-risk procedure, facilities could consider a more permissive approach to perioperative testing. Those facilities should consider if preoperative testing may still inform such infection prevention decisions such as room assignment/cohorting after surgery.
- Substantial or high community transmission: Facilities should continue pre-procedural testing as recommended in earlier updates.
- Pre-procedural testing has imperfect sensitivity. A negative test result should not preclude implementation of infection prevention measures such as use of respirators for healthcare personnel performing aerosol-generating procedures.
- Substantial or high community transmission: Universal use of NIOSH-approved N95 or equivalent or higher-level respirators for healthcare workers during perioperative encounters can simplify implementation.
- For patients who have tested positive for SARS- CoV 2, previous recommendations advised against retesting patients and relied solely on time-based or symptom-based empiric strategies. Those recommendations have been updated by the CDC.
- Patients who are moderately to severely immunocompromised may produce replication-competent virus beyond 20 days after symptom onset. Therefore, for this particular subset of patients, the CDC has updated its recommendations and advises a test-based strategy and (if available) recommends consultation with an infectious disease specialist to determine when Transmission-Based Precautions could be discontinued for these patients.
- Patients who are severely ill and not moderately or severely immunocompromised may produce replication-competent virus beyond 10 days. The CDC states that a test-based strategy can be considered in consultation with infectious disease experts.
- Other situations where the option of a test-based strategy might be used are:
- Patients who are symptomatic: Resolution of fever without the use of fever-reducing medications and symptoms (e.g., cough, shortness of breath) have improved, and results are negative from at least two consecutive respiratory specimens collected ≥24 hours apart (total of two negative specimens) tested using an antigen test or nucleic acid amplification test (NAAT).
- Patients who are not symptomatic: Results are negative from at least two consecutive respiratory specimens collected ≥24 hours apart (total of two negative specimens) tested using an antigen test or NAAT.
- Patients who have recurrence of symptoms after treatment with oral therapies such as paxlovid.
Table. Determining Community Transmission Risk of COVID-19. If the two indicators suggest different transmission levels, the higher level is selected.
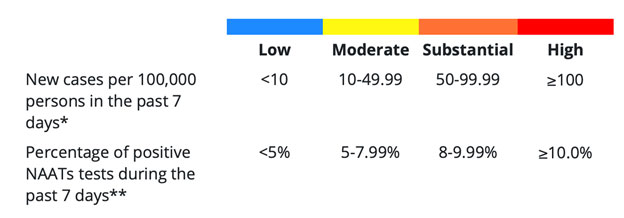
*Calculated by adding the number of new cases in the county in the last 7 days divided by the population in the county and multiplying by 100,000.
**Calculated by dividing the number of positive tests in the county during the last 7 days by the total number of tests resulted over the last 7 days.
Table was developed by the CDC and is available free of charge at: https://covid.cdc.gov/covid-data-tracker/#county-view?list_select_state=all_states&list_select_county=all_counties&data-type=Risk
Use does not imply endorsement by CDC of the material within this statement.
References
- Aminian A, Safari S, Razeghian-Jahromi A, Ghorbani M, Delaney CP. COVID-19 outbreak and surgical practice: unexpected fatality in perioperative period. Ann Surg 2020;272:e27-e29
- Tuech J-J, Gangloff A, Di Fiore F, Michel P, Brigand C, Slim K, et al. Strategy for the practice of digestive and oncological surgery during the Covid-19 epidemic. J Visc Surg 2020;157:S7-S12
- Kayani B, Onochie E, Patil V, Begum F, Cuthbert R, Ferguson D, Bhamra JS, Sharma A, Bates P, Haddad FS. The effects of COVID-19 on perioperative morbidity and mortality in patients with hip fractures: a multicentre cohort study. Bone Joint J 2020;102-B(9):1136-45
- Knisely A, Zhou ZN, Wu J, Huang Y, Holcomb K, Melamed A, Advincula AP, Lalwani A, Khoury-Collado F, Tergas AI, St Clair CM. Perioperative Morbidity and Mortality of Patients With COVID-19 Who Undergo Urgent and Emergent Surgical Procedures. Ann Surg 2021;273:34-40
- Liang W, Guan W, Chen R, Wang W, Li J, Xu K, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol 2020;21:335-7
- Lei S, Jiang F, Su W, Chen C, Chen J, Mei W, et al. Clinical characteristics and outcomes of patients undergoing surgeries during the incubation period of COVID-19 infection. EClinicalMedicine. 2020;21:100331.
- American Society of Anesthesiologists and Anesthesia Patient Safety Foundation Joint Statement on Elective Surgery and Anesthesia for Patients after COVID-19 Infection. https://www.apsf.org/news-updates/asa-and-apsf-joint-statement-on-elective-surgery-and-anesthesia-for-patients-after-covid-19-infection/ (Accessed June 8, 2022)
- COVIDSurg Collaborative, Machine learning risk prediction of mortality for patients undergoing surgery with perioperative SARS-CoV-2: the COVIDSurg mortality score, Br J Surg 2021;108:1274–1292
- Gudbjartsson DF, Helgason A, Jonsson H, Magnusson OT, Melsted P, Norddahl GL, et al. Spread of SARS-CoV-2 in the Icelandic Population. N Engl J Med 2020 11;382:2302-15
- Sutton D, Fuchs K, D’Alton M, Goffman D. Universal screening for SARS-CoV-2 in women admitted for delivery. New England Journal of Medicine. 2020.
- Centers for Disease Control and Prevention. Symptoms of Coronavirus. Updated 2/22/21. https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html Accessed June 8, 2022
- He X, Lau EH, Wu P, Deng X, Wang J, Hao X, et al. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat Med 2020:26:672-75
- IDSA Guidelines on the Diagnosis of COVID-19: Molecular Diagnostic Testing. https://www.idsociety.org/practice-guideline/covid-19-guideline-diagnostics/ Accessed June 8, 2022
- Interim Guidance on Ending Isolation and Precautions for Adults with COVID-19. Updated January 14, 2022. https://www.cdc.gov/coronavirus/2019-ncov/hcp/duration-isolation.html Accessed June 8, 2022
- Zhao J, Yuan Q, Wang H, Liu W, Liao X, Su Y, et al. Antibody responses to SARS-CoV- 2 in patients of novel coronavirus disease 2019. Clinical Infectious Diseases. 2020.
- Okba NM, Muller MA, Li W, Wang C, GeurtsvanKessel CH, Corman VM, et al. SARS- CoV-2 specific antibody responses in COVID-19 patients. medRxiv. 2020.
- World Health Organization. Advice on the use of point-of-care immunodiagnostic tests for COVID-19 [updated 4/8/2020. https://www.who.int/news-room/commentaries/detail/advice-on-the-use-of-point-of-care-immunodiagnostic-tests-for-covid-19 Accessed June 8, 2022
- Preoperative COVID Testing: Examples from Around the U.S. https://www.apsf.org/novel-coronavirus-covid-19-resource-center/preoperative-covid-testing-examples-from-around-the-u-s/ Accessed June 8, 2022
- Centers for Disease Control and Prevention. Overview of Influenza Surveillance in the United States. https://www.cdc.gov/flu/pdf/weekly/overview-update.pdf Accessed June 8, 2022
- Interim Infection Prevention and Control Recommendations for Healthcare Personnel During the Coronavirus Disease 2019 (COVID-19) Pandemic. Updated February 2, 2022. Available from https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html
- CDC Covid Tracker. (accessed May 27, 2022) https://covid.cdc.gov/covid-data-tracker/#county-view?list_select_state=all_states&list_select_county=all_counties Accessed June 8, 2022
