This paper highlights the existence of a substantial body of literature demonstrating that mortality following major surgery can be significantly reduced by the adoption of certain goal-directed approaches to perioperative hemodynamic management. The argument will be presented in four stages. Firstly we will report the incidence of mortality following surgery and propose that this represents a major public health issue. Secondly we will suggest that patients at higher risk of mortality or major morbidity following surgery can be identified and describe a number of methods to achieve this aim. Thirdly we will review the literature supporting the claim that perioperative goal directed hemodynamic management can prevent a proportion of this mortality. Finally we ask the question: can we defend not adopting this management strategy in our patients?
History
Death and major morbidity following surgical procedures should be of major concern to anesthesiologists and surgeons. Historically anesthesiologists have focused on mortality directly attributable to anesthesia such as anaphylactic reactions to anesthetic drugs, perioperative cardiac arrest and mortality associated with failed intubation. This philosophy has led to the development of critical incident reporting systems1 and the ASA Closed Claims Project (in response to rising medical insurance costs for anesthesiologists in the USA).2,3 Improvements in patient care resulting from this approach include the introduction of the ASA difficult intubation algorithm and the establishment of national minimum monitoring standards. Death rates directly attributable to anesthesia of between 1 in 10,000 and 1 in 250,000 are commonly quoted for this type of risk.4,5
However the definition of what is directly attributable to anesthesia is tight. The reported mortality rate is at most 1% of the total number of deaths following surgery. Although not commonly discussed it is important to consider the possibility that anesthetic practice might be a factor in some of this “non-attributable” mortality. In order to address this proposition it is necessary to answer two questions. Firstly the size of the problem must be defined. Secondly any evidence suggesting that a proportion of this mortality be either broadly attributable to a facet of anesthetic practice or that it might be preventable by a change in this practice must be considered. We contend both that the postoperative mortality is a significant public health issue and that a large body of evidence supports the assertion that specific approaches to cardiovascular management can reduce this mortality.
Population based data reporting mortality following surgery is scarce but where present suggests that the risks of surgery are greater than commonly perceived. At present defining how often these serious adverse outcomes occur, and to which patients, is difficult. Case series reporting outcome following specific procedures are commonly published from Specialist Centers with an established interest and expertise in a specific field and frequently report impressively low levels of mortality.6 These studies serve as a useful marker of what is achievable in the best hands but are unlikely to be representative of community wide practice. Publication bias is a reasonable fear: centers with poor results are unlikely to publicize their figures, particularly in a competitive market environment. A more representative picture may be obtained from nationally published figures collected for administrative and audit purposes where these are available.
Analysis
The availability of epidemiological data about surgical risk varies widely between different countries. In the US in particular national data are sparse, possibly because of medico-legal and commercial concerns about the publication of potentially sensitive information. In other countries more data are available and, where present, this information frequently conflicts with clinicians’ perceptions and published case series. In the United Kingdom (except Scotland) a national audit of deaths (National Confidential Enquiry into Perioperative Deaths Ð NCEPOD) occurring within 30 days of surgery (19,832) provides a numerator for calculation of national surgical mortality although data collection is known to be incomplete. The denominator, total number of non day-case operations performed per year, may be obtained from nationally available hospital surgical activity data (3,040,149).7 The overall surgical death rate for non day-case procedures, elective and emergency, calculated from these figures is 0.65%. The newly published National Health Service performance indicators in England, which include 30-day in hospital postoperative mortality on a Trust (administrative unit) by Trust basis, report 32,956 deaths from 2,325,636 procedures in the financial year to April 1999 with an overall calculated mortality rate of 1.4% (emergency cases 3.87%, non-emergency cases 0.48%).8 It is arguable that postsurgical mortality between different countries and health care systems may vary by a significant amount. However, it seems unlikely that this variation will even approach a single order of magnitude between broadly comparable developed world healthcare systems. Mortality following surgery, particularly in patients at high-risk has a significant measurable incidence. Clearly the vast majority of this mortality following surgery is not directly attributable to anesthesia. Equally it is not obviously surgical misadventure. The available data from clinical studies and from national data collection exercises (NCEPOD) suggests that much of this mortality is delayed (>50% occurring after the 5th postoperative day). Often the death occurs on the intensive care unit, commonly from multiple-organ failure after a protracted period of illness. There is an increasing body of evidence that this mortality can be reduced.
This risk of death following surgery is related not only to the magnitude and duration of the procedure but also to a number of patient related factors. For a given procedure three key patient variables contribute to the risk of a major surgical procedure: mode of presentation, age and the presence of co-morbidity. Patients presenting for surgery as urgent or emergency cases have a much greater risk of adverse events when compared to those undergoing scheduled or elective procedures (UK-NCEPOD definitions).8,9 The risk of adverse outcome following surgery is also increased with increasing age (NCEPOD).4,10 However, it is unclear whether this association occurs because of the large number of elderly individuals with significant comorbidity or whether age alone is a risk factor. The presence of co-morbidity increases risk in a summative fashion.11,12 The degree of increase in risk for different types and degrees of co-morbidity is, however, difficult to quantify and report without resorting to sophisticated methods of risk description, e.g., weighted scoring systems.
On an institutional level the surgical patient at high-risk of adverse outcome can be identified using subjective assessment or using objective criteria. Formal identification of those patients at high-risk of adverse outcome permits both preoperative risk stratification and meaningful institutional audit of surgical outcome but this is rarely undertaken. Objective criteria may be simple categorical systems or more complex scoring systems13 or more complex scoring systems.14 Scoring systems can be used for case-mix (risk) adjustment. Comparison can then be made between non-matched populations or with historical benchmarking data.15
Adverse outcome following surgery occurs through a combination of patient factors, (e.g. age and co-existing disease) and operation factors (e.g. magnitude and duration of procedure). The high-risk surgical patient can be defined as any patient with a significant chance of a major adverse outcome, death or major morbidity, following an operative procedure. Identification of the high-risk patient requires recognition of risk factors and experienced clinicians integrate this information into a subjective assessment of the risk of an adverse outcome. This may be very effective in individual cases but is difficult to reproduce consistently, and impossible to define clearly in the context of audit, quality-improvement or clinical research. A number of more objective approaches are available and have been used both in research studies and the clinical environment.
Optimization
Several of the optimization studies have used broadly similar categories to identify high-risk surgical patients. The “Shoemaker criteria” include broad groups of patients e.g. previous severe cardiorespiratory illness, acute abdominal catastrophe with hemodynamic instability.13 The development of scoring systems using weighted scores for multiple categories has significantly improved our ability to predict adverse outcome following major surgery. A well designed and adequately validated scoring system can have great utility for comparing outcome between different units. The APACHE scoring systems which are widely used in the critical care setting provide a good example of this kind of “case-mix” or risk adjustor and allow comparison of standardized mortality rates between units and within units over time.16
The American Society of Anesthesiologists physical status score, originally defined in the 1940s and refined to its present form in 1963, is a five-component morbidity score which has become a routine part of the preoperative anesthetic assessment.14 It has the great benefits of simplicity and almost universal acceptance but suffers from subjective interpretation of loosely defined categories. Despite these limitations it has been demonstrated to function as simple risk-adjustor, a higher score being consistently associated with an increase mortality rate.
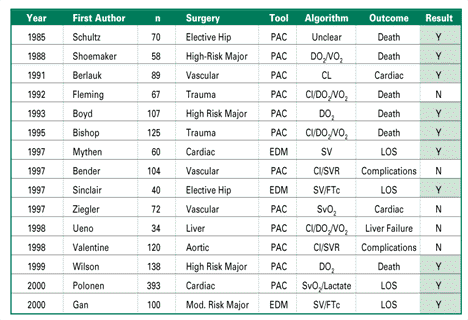
Table 1. Studies on perioperative goal directed hemodynamic management and outcome. PAC = pulmonary artery catheter; EDM = esophageal Doppler monitor; DO2 = oxygen delivery, VO2 = oxygen consumption; CI = cardiac index; SV = stroke volume; SVR = systemic vascular resistance; FTc = corrected flow time; SvO2 = venous oxygen saturation; LOS = length of stay. Y = positive outcome; N = negative outcome.
More recently in an attempt to quantify the risk of cardiac complications following surgery Goldman et al described a Cardiac Risk Index17 that can be used as a predictor of risk of cardiac death. This, and other related scores, is not intended to predict overall mortality and in addition take no account of operative severity. POSSUM (Physiological and Operative Severity Score for the enUmeration of Mortality) is a scoring system developed specifically as a case-mix adjustor for general surgical patients.12 The score is compose of a 12-section physiological score recorded preoperatively and 6-component Operative Severity Score recorded by the operating surgeon. Using reference data from a database of more than 200,000 patients an expected risk of mortality or morbidity can be obtained for a particular score. When looking at populations the observed mortality (or morbidity) can be compared with the expected mortality (or morbidity) and an observed to expected ratio (O:E ratio) obtained. An O:E ratio greater than 1 indicates an outcome worse than the historical benchmark whereas a ratio less than 1 indicates an outcome better than expected from the historical data. The score can therefore be used to compare outcome in nonmatched populations or to provide a historical benchmark to compare a current cohort with. It can therefore be a valuable tool for research and audit. The score is neither designed for nor appropriate to use for making management decisions on an individual patient-by-patient basis.
If we accept that mortality following major surgery has a significant incidence and that patients at high-risk of adverse outcome can be identified then what should we be doing about these patients? Since the late 1950’s a succession of authors have described an association between perioperative cardiac output and survival following major surgery: the survivors exhibiting higher values than the non-survivors.18-20 From these observations, the hypothesis developeed that using the cardiac output and oxygen delivery values exhibited by the survivors, as goals for all patients would reduce overall mortality.20
In 1988 Shoemaker was the first to test this hypothesis in a randomized controlled clinical trial (RCT).13 In a complex study he demonstrated that targeting specific values for cardiac index, oxygen delivery and oxygen consumption, using fluids and inotropes to achieve these goals, resulted in a reduction in mortality, morbidity and, in a retrospective analysis, reduction in overall cost of patient care.
Hemodynamic Goals
Since then a number of single center randomized controlled trials have been conducted, the majority of which support this original positive result. Five studies have used the same hemodynamic goals as the original study by Shoemaker. Two of these were large (>100 patient) studies on high-risk general surgical and vascular patients and both demonstrated a statistically significant reduction in mortality in the protocol groups.21,22 Two were studies of major trauma surgery and these were both conducted by the same group.23,24 The first smaller study showed a trend towards reduction in mortality in the protocol group and this was confirmed by a statistically significant reduction in protocol group mortality in the second, larger, trial. The fifth study in this group was a small trial focusing on surgery for hepatobiliary carcinoma and demonstrated a reduction in liver failure and hyperbilirubinemia although this was not their specified primary outcome variable.25 An older study using a similar philosophy, but with less clearly defined goals, in patients undergoing hip fracture surgery also demonstrated a significant mortality reduction.26
Somewhat different results have been obtained in a group of papers in which patients presenting for major vascular or aortic surgery were studied.27-29 The goals for cardiac index and oxygen delivery used in these trials were significantly lower and the overall mortality for each trial was also low. These studies did not demonstrate a significant reduction in mortality, or in some cases complications, however in only one of these studies were there more deaths in the protocol than control groups.28
Targeting mixed venous oxygen saturation (SvO2) as an indirect index of oxygen delivery has also been studied in two trials. The first studied patients having aortic or lower limb arterial surgery and failed to demonstrate a significant morbidity or mortality difference between control and protocol groups.30 More recently a large Scandinavian study of patients undergoing elective coronary revascularization with cardiopulmonary bypass demonstrated a significant reduction in length of stay in those randomized to maintenance of SvO2 > 70% and lactate ² 2 mmoláL-1 when compared with controls.31
Two published studies using intra-operative esophageal Doppler monitoring of cardiac output compared a stroke volume optimization algorithm with standard fluid management. In the first study patients with normal left ventricular function undergoing coronary artery revascularization had a statistically significant reduction in length of both ICU and overall hospital stay in the protocol group.32 The second study, of elderly patients having hip prosthesis surgery, also demonstrated a reduction in hospital length of stay in patients managed in the protocol group.33 Neither of these studies was designed to demonstrate a mortality difference. A recent study using a similar optimization algorithm also demonstrated a reduction in hospital stay and earlier return to tolerating solid food in the protocol group.34
The application of this management approach to patients with established critical illness has been much less successful. A number of single center studies35,36 and one large multi-center RCT37 have failed to show an outcome benefit for patients in the protocol group. Indeed in some studies the intervention group mortality exceeded that of the control group.36
Taken together the perioperative trials present an interesting picture. Many of the studies were not designed with mortality as a primary outcome goal and several of those that were did not have adequate statistical power to detect a mortality reduction. However if all the trials are grouped together and the control group mortality compared with the intervention group the results are striking. The crude death rates are 96/753 for the combined control groups and 36/814 for the combined protocol groups. In only two of 15 studies was there greater mortality in the protocol group: in one study 5/72 patients died: 2/40 control and 3/32 protocol and in the other 4/120 patients died: 1/60 control and 3/60 protocol.28,30
The accumulated body of evidence relating to goal directed hemodynamic perioperative management now includes at least 14 RCTS (Table 1) and 2 supportive meta analyses.38,39 A third meta-analysis using the Cochrane methodology and further clinical trials are currently underway. In the world of evidence-based medicine this is Level 1 evidence supporting a change in clinical practice (Class A recommendation). However anecdotal evidence suggests that this philosophy of care is rarely followed in practice. There are probably three main reasons for this. Firstly for many there is no recognition that a problem exists – it is a commonly stated position that “I don’t see that level of mortality and morbidity in my practice”. Lack of data to support the existence of the problem is very different from data demonstrating there is no problem. In the absence of data demonstrating that a problem does not exist such a position is untenable in the face of the available epidemiological data. Secondly the idea that anesthetic management might be a key determinant of longer-term outcome (i.e. outside of the immediate perioperative period) is an anathema to many anesthesiologists and surgeons. Finally the practical conduct of optimization requires a significant investment of time and resources and this may not be possible without significant investment adjustments.
Conclusion
The amount and quality of literature supporting goal directed perioperative hemodynamic management is considerably greater than that supporting many commonly accepted interventions. However, there is profound resistance to change. The Cochrane Meta-analysis and subsequent multi-center RCTs are keenly awaited. In the face of increasing evidence demonstrating that outcome can be improved, a conservative approach will become increasingly hard to defend.
Dr. Mike Grocott is Clinical Research Fellow, Surgical Outcomes Research Centre, UCLH, London, UK and Dr. Gan is Associate Professor and Director of Clinical Research in the Department of Anesthesiology, and Medical Director of Peri-Anesthesia Care Unit, Duke University Medical Center.
References
1. Cooper, J. B., Newbower, R. S., and Kitz, R. J. An Analysis of Major Errors and Equipment Failures in Anesthesia Management: Considerations for Prevention and Detection. Anesthesiology 1984;60(1):34-42.
2. Caplan, R. A., Posner, K. L., Ward, R. J., and Cheney, F. W. Adverse Respiratory Events in Anesthesia: a Closed Claims Analysis. Anesthesiology 1990;72(5):828-33.
3. American Society of Anesthesiologists Closed Claims Project. 2001.Ref Type: Internet Communication:http://depts.washington.edu/asaccp/
4. Lunn, J. N. and Mushin, W. W. Mortality Associated With Anesthesia. Anaesthesia 1982;37(8):856.
5. American Society of Anesthesiologists (Patient Safety). 2001. Ref Type: Internet Communication: http://www.asahq.org/what_is_anesth.html#anchor20748154
6. Michalopoulos, A., Tzelepis, G., Dafni, U., and Geroulanos, S. Determinants of Hospital Mortality After Coronary Artery Bypass Grafting. Chest 1999;115(6):1598-603.
7. Hospital Episode Statistics (UK). 2001. Ref Type: Internet Communication: http://www.doh.gov.uk/hes
8. NHS Performances Indicators. 2001. Ref Type: Internet Communication: http://www.doh.gov.uk/nhsperformanceindicators/hlpi2000/index2000.html
9. The 2000 National Confidential Enquiry into Peri-Operative Deaths. 2001. Ref Type: Internet Communication: http://www.ncepod.org.uk/20001.htm
10. Daley, J., Khuri, S. F., Henderson, W., Hur, K., Gibbs, J. O., Barbour, G., Demakis, J., Irvin, G., III, Stremple, J. F., Grover, F., McDonald, G., Passaro E Jr, Fabri, P. J., Spencer, J., Hammermeister, K., Aust, J. B., and Oprian, C. Risk Adjustment of the postoperative Morbidity Rate for the Comparative Assessment of the Quality of Surgical Care: Results of the National Veterans Affairs Surgical Risk Study. Journal of the American College of Surgeons 1997;185(4):328-40.
11. Dripps, RD, Lamont, A, and Eckhardt, M. The Role of Anesthesia in Surgical Mortality. JAMA 1963;178(3):261-6.
12. Copeland, G. P., Jones, D., and Walters, M. POSSUM: a Scoring System for Surgical Audit. British Journal of Surgery 1991;78(3):355-60.
13. Shoemaker, W. C., Appel, P. L., Kram, H. B., Waxman, K., and Lee, T. S. Prospective Trial of Supranormal Values of Survivors As Therapeutic Goals in High-Risk Surgical Patients. Chest 1988;94(6):1176-86.
14. Anonymous. New Classification of Physical Status. Anesthesiology 1963;24(1):111.
15. Sagar, P. M., Hartley, M. N., MacFie, J., Taylor, B. A., and Copeland, G. P. Comparison of Individual Surgeon’s Performance. Risk-Adjusted Analysis With POSSUM Scoring System. Diseases of the Colon & Rectum 1996;39(6):654-8.
16. Knaus, W. A., Wagner, D. P., Draper, E. A., Zimmerman, J. E., Bergner, M., Bastos, P. G., Sirio, C. A., Murphy, D. J., Lotring, T., and Damiano, A. The APACHE III Prognostic System. Risk Prediction of Hospital Mortality for Critically Ill Hospitalized Adults. Chest 1991;100(6):1619-36.
17. Goldman, L., Caldera, D. L., Nussbaum, S. R., Southwick, F. S., Krogstad, D., Murray, B., Burke, D. S., O’Malley, T. A., Goroll, A. H., Caplan, C. H., Nolan, J., Carabello, B., and Slater, E. E. Multifactorial Index of Cardiac Risk in Noncardiac Surgical Procedures. New England Journal of Medicine 1977;297(16):845-50.
18. Boyd, A. R., Tremblay, R. E., Spencer, F. C., and Bahnson, H. T. Estimation of Cardiac Output Soon After Cardiac Surgery With Cardiopulmonary Bypass. Annals of Surgery 1959;150:613-25.
19. Clowes, GHA and Del Guerico, LRM. Circulatory Response to Trauma of Surgical Patients. Metabolism 1960;9(1):67-81.
20. Shoemaker, W. C., Montgomery, E. S., Kaplan, E., and Elwyn, D. H. Physiological Patterns in Surviving and Non-Surviving Shock Patients. Use of Sequential Cardiorespiratory Parameters in Defining Criteria for Therapeutic Goals and Early Warning of Death. Archives of Surgery 1973;106:630-6.
21. Boyd, O., Grounds, R. M., and Bennett, E. D. A Randomized Clinical Trial of the Effect of Deliberate Perioperative Increase of Oxygen Delivery on Mortality in High-Risk Surgical Patients. JAMA 1993;270(22):2699-707.
22. Wilson, J., Woods, I., Fawcett, J., Whall, R., Dibb, W., Morris, C., and McManus, E. Reducing the Risk of Major Elective Surgery: Randomised Controlled Trial of preoperative Optimisation of Oxygen Delivery. BMJ 1999;318(7191):1099-103.
23. Fleming, A., Bishop, M., Shoemaker, W., Appel, P., Sufficool, W., Kuvhenguwha, A., Kennedy, F., and Wo, C. J. Prospective Trial of Supranormal Values As Goals of Resuscitation in Severe Trauma. Archives of Surgery 1992;127(10):1175-9.
24. Bishop, M. H., Shoemaker, W. C., Appel, P. L., Meade, P., Ordog, G. J., Wasserberger, J., Wo, C. J., Rimle, D. A., Kram, H. B., Umali, R., and et al. Prospective, Randomized Trial of Survivor Values of Cardiac Index, Oxygen Delivery, and Oxygen Consumption As Resuscitation Endpoints in Severe Trauma. Journal of Trauma-Injury Infection & Critical Care 1995;38(5):780-7.
25. Ueno, S., Tanabe, G., Yamada, H., Kusano, C., Yoshidome, S., Nuruki, K., Yamamoto, S., and Aikou, T. Response of Patients With Cirrhosis Who Have Undergone Partial Hepatectomy to Treatment Aimed at Achieving Supranormal Oxygen Delivery and Consumption. Surgery 1998;123(3):278-86.
26. Schultz, R. J., Whitfield, G. F., LaMura, J. J., Raciti, A., and Krishnamurthy, S. The Role of Physiologic Monitoring in Patients With Fractures of the Hip. Journal of Trauma-Injury Infection & Critical Care 1985;25(4):309-16.
27. Berlauk, J. F., Abrams, J. H., Gilmour, I. J., O’Connor, S. R., Knighton, D. R., and Cerra, F. B. preoperative Optimization of Cardiovascular Hemodynamics Improves Outcome in Peripheral Vascular Surgery. A Prospective, Randomized Clinical Trial. Annals of Surgery 1991;214(3):289-97.
28. Valentine, R. J., Duke, M. L., Inman, M. H., Grayburn, P. A., Hagino, R. T., Kakish, H. B., and Clagett, G. P. Effectiveness of Pulmonary Artery Catheters in Aortic Surgery: a Randomized Trial. Journal of Vascular Surgery 1998;27(2):203-11.
29. Bender, J. S., Smith-Meek, M. A., and Jones, C. E. Routine Pulmonary Artery Catheterization Does Not Reduce Morbidity and Mortality of Elective Vascular Surgery: Results of a Prospective, Randomized Trial. Annals of Surgery 1997;226(3):229-36.
30. Ziegler, D. W., Wright, J. G., Choban, P. S., and Flancbaum, L. A Prospective Randomized Trial of preoperative “Optimization” of Cardiac Function in Patients Undergoing Elective Peripheral Vascular Surgery. Surgery 1997;122(3):584-92.
31. Polonen, P., Ruokonen, E., Hippelainen, M., Poyhonen, M., and Takala, J. A Prospective, Randomized Study of Goal-Oriented Hemodynamic Therapy in Cardiac Surgical Patients. Anesthesia & Analgesia 2000;90(5):1052-9.
32. Mythen, M. G. and Webb, A. R. Perioperative Plasma Volume Expansion Reduces the Incidence of Gut Gut Mucosal Hypoperfusion During Cardiac Surgery. Arch Surg 1995;130:423-9.
33. Sinclair, S., James, S., and Singer, M. Intra-operative Intravascular Volume Optimisation and Length of Hospital Stay After Repair of Proximal Femoral Fracture: Randomised Controlled Trial. BMJ 1997;315(7113):909-12.
34. Gan, T. J.; Soppitt, A. J.; Maroof, M.; El-Moalem, H.; Glass, P. S. A. Intra-operative volume expansion guided by esophageal Doppler improves postoperative outcome and shortens hospital stay. Anesthesiology 2001;91:A537.
35. Yu, M., Levy, M. M., Smith, P., Takiguchi, S. A., Miyasaki, A., Myers, and SA. Effect of Maximizing Oxygen Delivery on Morbidity and Mortality Rates in Critically Ill Patients: a Prospective, Randomized, Controlled Study. Critical Care Medicine 1993;21(6):830-8.
36. Hayes, M. A., Timmins, A. C., Yau, E. H., Palazzo, M., Hinds, C. J., and Watson, D. Elevation of Systemic Oxygen Delivery in the Treatment of Critically Ill Patients. New England Journal of Medicine 1994;330(24):1717-22.
37. Gattinoni, L., Brazzi, L., Pelosi, P., Latini, R., Tognoni, G., Pesenti, A., and Fumagalli, R. A Trial of Goal-Oriented Hemodynamic Therapy in Critically Ill Patients. SvO2 Collaborative Group. New England Journal of Medicine 1995;333(16):1025-32.
38. Boyd, O. and Hayes, M. The Oxygen Trail-the Goal. Br.Med.Bull. 1999;55(1):125-39.
39. Heyland, D. K., Cook, D. J., King, D., Kernerman, P., Brun, Buisson, and C. Maximizing Oxygen Delivery in Critically Ill Patients: a Methodologic Appraisal of the Evidence. [Review] [42 Refs]. Critical Care Medicine 1996;24(3):517-24.

