We have discovered what we believe to be a flaw in the Dräger Fabius Tiro® anesthetic machines, which we use in our same day surgery operating rooms. We have observed a problem with the leak test, which does not detect a cracked, broken, or absent carbon dioxide absorbent canister. We discovered this problem a few weeks ago when the Fabius Tiro® machine passed all the checks in the morning, but shortly after beginning our first case we realized that the carbon dioxide absorbent was missing from the circuit. By the time a canister was placed, the patient’s inspired pCO2 was 30 mmHg. Fortunately the patient experienced no lasting sequelae from this event.
After investigating the incident, we discovered that the Dräger Fabius Tiro® leak test uses positive pressure, which closes the valve to the carbon dioxide absorbent, sealing off any leaks caused by abnormalities in the carbon dioxide absorbent canister. Furthermore, there is no prompt to remind clinicians to check the carbon dioxide absorbent during the machine checkout. This is a particularly dangerous situation as many clinicians at our institution are under the impression that the absence of a carbon dioxide absorbent canister would cause an alert during the leak test. We would like to advise others of this potential error and suggest that Dräger incorporate the carbon dioxide absorbent canister into the Fabius Tiro® machine’s leak test.
Karen C. Nanji, MD, MPH
Edward A. Bittner, MD, PhD
Boston, MA
In Response:
The leak test performed by the Dräger Fabius Tiro (also applies to Fabius GS) does not detect the absence of a CO2 absorbent canister when using the CLIC adapter. That adapter is designed to allow for the canister to be removed and changed during patient care by closing a valve and preventing any leaks from the patient circuit when the canister is removed. Since the leak test was performed without a CO2 absorbent canister in place, no leak was detected.
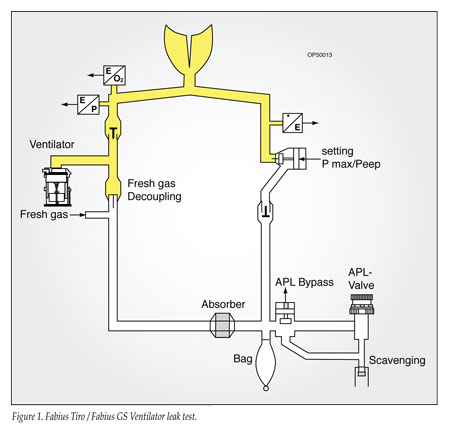
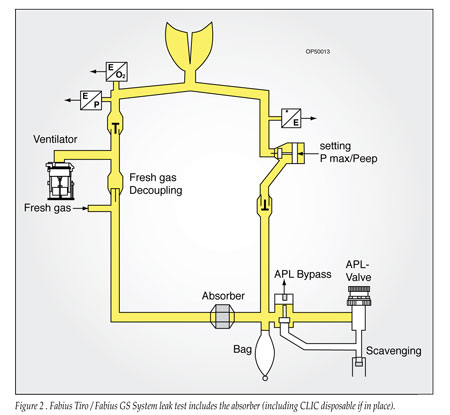
The supposition that the machine leak test does not identify leaks in the absorbent canister is not correct. The Fabius Tiro and GS incorporate a semi-automatic leak and compliance test as part of the recommended pre-use check. The test is divided into 2 parts to assist the user with troubleshooting, one for the patient circuit during mechanical ventilation, and one for the complete system back to the flow control valves (Figures 1 and 2). The complete system test includes the CO2 absorbent canister, and will identify leaks in the canister assuming the canister is properly placed and the CLIC adapter closed.
The recommended checkout procedure in the Tiro and Fabius manuals includes a step for “Checking the Condition of the CO2 Absorbent.” This is a visual inspection intended to identify that the absorbent is present and the amount of indicator is not excessive. If this visual step is performed and a CO2 canister properly placed, the leak test will identify a leak if the integrity of the CO2 absorbent canister is compromised.
The Fabius Tiro and GS anesthesia delivery systems are also available with a loose fill CO2 absorbent canister option instead of the CLIC absorber. The loose fill canister must be in place and secured during the leak test; otherwise, a leak will be detected.
The concerns expressed above underscore the challenge of automating pre-anesthesia checkout procedures. No anesthesia system on the market has completely automated all aspects of the checkout procedures and eliminated the need for manual checkout. The current Pre-Anesthesia Checkout Guidelines published by the American Society of Anesthesiologists recognizes the need for both automated and manual procedures as well as the differences between anesthesia delivery systems. These guidelines are a useful resource for evaluating checkout procedures used for individual delivery systems (see
http://www.asahq.org/clinical/fda.htm).
Users of the Dräger CLIC absorber must be aware of the potential disadvantages of that system. One disadvantage is highlighted in this report and can be eliminated by noting the presence of a properly placed absorbent canister before starting the automated leak test. The other disadvantage will be apparent if a cracked or leaking absorbent canister is replaced during patient care. The leak test cannot be performed during patient care without interrupting anesthesia delivery and ventilation. If a leak should become apparent after changing the canister during a case, the integrity of the canister should be suspected as the cause of the leak.
The value of capnography is also highlighted by this report. Increased inspired carbon dioxide concentration resulted from the absence of an absorbent canister and helped to identify the problem. It should be noted that CO2 absorbent is present in a circle system to deliver vapor efficiently by allowing rebreathing of exhaled gas, and is not required to prevent hypercarbia. When inspired CO2 results from absent or exhausted CO2 absorbent material, inspired CO2 can be eliminated by increasing fresh gas flow to exceed minute ventilation until the problem can be rectified.
Robert Clark
CareArea™ Director, Perioperative Care
Dräger Medical, Inc.
Jeffrey Feldman, MD, MSE
APSF Committee on Technology
| The information in this column is provided for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of APSF. It is not the intention of APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information. |


 Issue PDF
Issue PDF