| Adapted from Special Feature on Pulse Oximeters: The invention that changed the paradigm of patient safety around the world. (LiSA (1340-8836) vol28 No3 Page237-308, 2021.03 (in Japanese)
Disclaimer: The information provided is for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of APSF. It is not the intention of APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information. |
The invention of the pulse oximeter by Dr. Takuo Aoyagi in 1974 has been viewed as a typical case of medical device development achieved under a plan based on definite goal setting. The favorable outcome of such an approach to development is shown by the fact that the medical device developed thus has been commercialized and adopted widely across the globe. In the medical device industry, characterized by small-lot large-variety production, scarcely any medical device has contributed as significantly to healthcare as the pulse oximeter. Thus, it may be called “a good model of medical device development.”
I have just given the conclusion at the beginning of this paper. When the project led by Dr. Aoyagi was under way, I was also a member of the Development Department of Nihon Kohden Corporation. In those days, I could hardly have imagined that the device development under way at the neighboring section would advance to such a remarkable extent 1), because the missions of different sections of the department differed completely from each other. Neighboring sections were expected to make efforts together for improvement through friendly rivalry. The job that I was in charge of in those days pertained to improving patient monitors (e.g., adoption of wireless monitor systems), which had already advanced to a certain degree, and this mission was different in nature from that of in which Dr. Aoyagi was engaged. In other words, the topic of development assigned to my section, which pertained to improving the practical aspects of existing products, differed in dimension from the innovative development topic assigned to the Aoyagi Project.
Figure 1 shows the front page of the Patent Publication Bulletin on “Optical Oxygen Measuring Device,” application for patent registration of which was filed in 1974 2). This device later served as the prototype of the pulse oximeter. To this document, the memo “Registration NO. 947,714” was later added manually. This memo is a valuable relic written by Dr. Aoyagi himself. A noteworthy phrase used in this bulletin is the “earpiece-type oximeter” as an example of patent embodiment. This was commercialized as the initial product of an “ear oximeter.” Here, the term “oximeter” means “measuring oxygen” and the phrase “earpiece-type” indicates “measurement attaching to the earlobe.”
From the standpoint of engineering, the ear oximeter was characterized by the use of an analog device, with utilization of primitive electric parts like light bulbs as the source of light. It is an undeniable fact that these characteristics of the ear oximeter led to unstable of the measurements, resulting in the development of the domestic impression that the product was “useless.” ☆
Efforts to overcome such a challenge were made by Minolta Camera, Inc. (currently named “Konica Minolta, Inc.”). OXIMET, which was the first oximeter marketed by Minolta Camera, was characterized by the utilization of a “finger sensor” 3). It is noteworthy that this product for routine use was marketed only several years after the ear oximeter was launched in the market. Considering that most pulse oximeters currently on the market adopt this type of sensor, we may say that the OXIMET served as the driving force for establishment of the path towards development and commercialization of pulse oximeters for routine use.
However, the clinical usefulness, in the true sense of the term, of pulse oximeters became apparent only after the products were marketed by two American companies. The term “pulse oximeter” became popular only at in those days. This is because this kind of device was shown to play a significant role in the fields of anesthesiology/critical care and neonatal management.
The table below summarizes the course of pulse oximeter development, focusing on the actions/events in Japan and the USA. It can be seen from this table at a glance that the efforts made in Japan primarily pertained to the principle and development, while those in the USA contributed to implementation of pulse oximeters. On the basis of the efforts made in these two countries under such role assignment, pulse oximeters have so far grown into a group of devices that are used extensively in routine clinical practice around the world.
| Year | Role of Japan | Role of the USA |
| 1974 | Patent application for the pulse oximetry principle filed by Takuo Aoyagi (Nihon Kohden) | |
| 1977 | Pulse oximeter marketed by Akio Yamanishi et al. (Minolta Camera) | |
| 1981 | Pulse oximeter (the first full-scale product) marketed by Nellcor and Ohmeda | |
| 1992 | International Standard on Pulse Oximeter ISO 9919 enforced | |
| 1994 | Wrist watch-type pulse oximeter marketed by Techna Electronic Industry (currently named “T & RK”) | |
| 1996 | Fingertip-type pulse oximeter marketed by Nonin | |
| 1998 | Software (Signal Extraction Technology) for improving the precision of measurement with pulse oximeters developed by Masimo | |
| 2000 and afterwards | Portable/wearable products developed by many manufacturers, including Nihon Seimitsu-sokki, Inc. | |
| 2005 | Pulse oximeter allowing measurement under the environment for MRI marketed by Nonin | |
| 2011 | International Standard on Pulse Oximeter ISO 800601-2-61 (revision of ISO 9919) enforced | |
| 2014
|
Domestic standard on pulse oximeter JIS T80601-2-61 enforced |
|
| 2020 | “Blood Oxygen Wellness” introduced into common products by Apple Inc. | |
Table 1 Course of pulse oximeter development – Comparison between Japan and the USA
Biased towards the two Extremes (High-functioning products vs. Extensively applicable products)
Pulse oximeters that were successfully commercialized by the two American companies Nellcor and Biox (Ohmeda) as products for routine use were both standalone-type (Type A) products and were intended primarily for use in critical care (intraoperative care in severely sick patients, premature infant/neonate management, and so on). Development of these products was led by Prof. Severinghaus of the University of California, San Francisco (UCSF), who emphasized the significance of continuous monitoring of arterial blood oxygen saturation enabled by pulse oximeters. The contribution of Prof. Severinghaus is particularly consequential, in that he positioned a pulse oximeter as an optimal device for use in intraoperative respiratory management and thus, as an indispensable device for anesthesia management. At that time, he announced to the world that this principle related to a pulse oximeter was actually discovered by Dr. Takuo Aoyagi of Japan 4). It was Dr. Kunio Suwa (Department of Anesthesiology, University of Tokyo, in those days) who actively disseminated this information in Japan. This information, announced ten-odd years after registration of the related patent in Japan, was perceived as “information that had never been heard before” or “information that should prompt re-recognition,” even in Japan, the country where the researcher originated from.
Thereafter, new types of pulse oximeters were marketed one after another, not only by American manufacturers, but also by Japanese and European manufacturers. In the 1990s, pulse oximeters became general-purpose devices, applicable also to the care of moderately severe patients and in general wards. In the 2000s, technological advances in pulse oximeters became accelerated, resulting in the development of compact-sized/portable products and wearable products (Type B), thereby enabling more extensive use of pulse oximeters. Among others, spread of the fingertip sensor-combined type of pulse oximeter generated a new role for the pulse oximeter (a spot checker of oxygen saturation in ordinary patients and in health management in general; Type B), in addition to its previous role of patient monitoring; Type A.
Figure 2 shows the recent trends of the pulse oximeter market in Japan. It can be seen that the portable or wearable type of pulse oximeters, intended primarily for use as spot checkers (Type B), has spread more rapidly as compared to Type A pulse oximeters, which are primarily intended for use as medical monitors. The number of Type A pulse oximeters sold per year in the market has remained steady on the order of 7000 to 8000 in recent years, while the sales for Type B have been growing continuously year after year. At present, Type B pulse oximeter sales have exceeded 250 000 units per year and are expected to continue to increase.
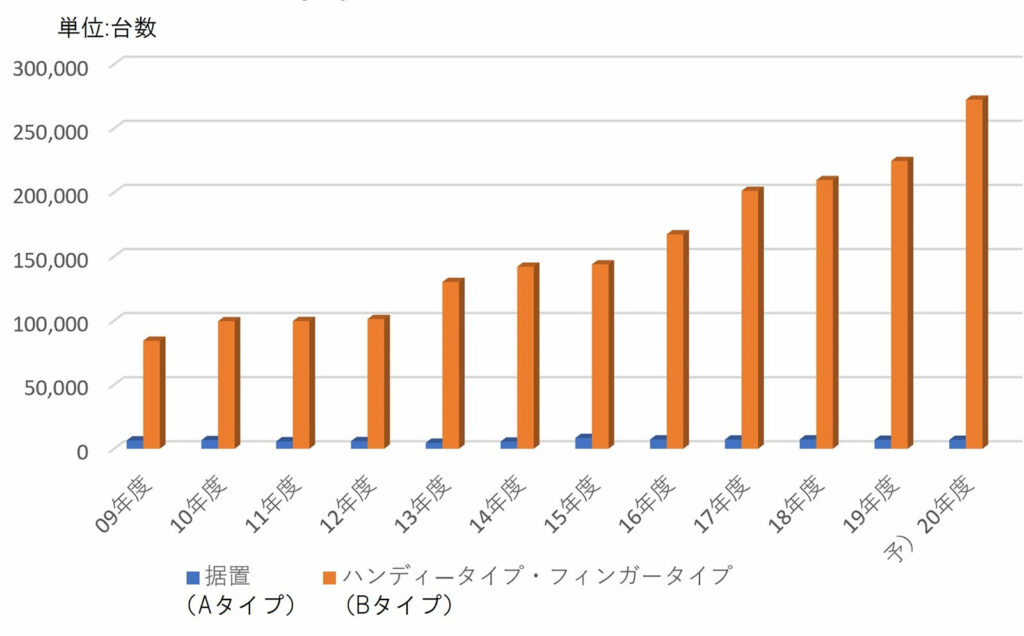
Figure 2: The recent trends of the pulse oximeter market in Japan. (2009 – 2020, Yano Research Institute)
To cite another characteristic of this field, the standalone-type pulse oximeters (Type A) mainly consist of high-functioning products often made overseas (e.g., USA). An example is a pulse oximeters that offers improved precision of measurement and is “capable of dealing also with bodily movements” 5) or “enabling measurement even during hypoperfusion.” There are also products which have been categorized as “pulse photometry” products that are capable of measuring abnormal hemoglobin (carbon monoxide-bound hemoglobin, methemoglobin, etc.), in addition to measuring the oxygen saturation level.
Among the Type B products, Japanese products are predominant. While typically it is difficult for big businesses to enter this market, the entry by venture companies, such as small- and medium-sized enterprises, is rather remarkable; as many as more than 30 such companies have entered this market in Japan, with Japan Precision Instruments, Inc., serving as the market leader. This device is fitted with a wireless communication function and is linkable to smartphones and clouds, reflecting the latest trends in the field of information technology.
Essential Nature of Pulse Oximeters and Its Relationship to International Standards
Now, the term “pulse oximeter” is known well; people hearing this word are likely to imagine a compact-sized Type B device. However, during the stage of development and early phase of commercialization, i.e., until the 1980s, only the full-scale type (Type A) of pulse oximeters were available in the market. After the turn of the century, however, this market changed dramatically, and Type B products have become predominant. Now, I shall refer to open issues related to Type B Pulse pulse oximeters, the currently predominantly used type of oximeter.
My greatest concern pertains to the large discrepancy between the international standards and the current status of Type B pulse oximeters available in the market. The current international standard ISO80601-2-61: 2011 was set forth, covering Type A full-scale pulse oximeters as the main target. For example, the section about precision of measurement in this standard requires implementation of a study involving strict comparison between the arterial oxygen saturation (SaO2) measured by the oximeter and the SaO2 in the patient’s arterial blood (in arterial blood samples) in healthy volunteers under induced hypoxemia. This means that even though a pulse oximeter is “essentially a non-invasive device,” the standard requires that it provides clinical evaluation results equivalent in precision to those of “highly invasive devices” of a different nature.
When viewed from the angle of underlying principle or the design concept during development, the primary principle of a pulse oximeter is “(non-invasive) extracorporeal measurement of oxygen saturation with the use of light.” It is no exaggeration to say that this principle or concept served as the basis for the global spread of this type of device. Considering that the predominantly used type of pulse oximeters changed from Type A monitors to the current compact-sized type monitors, it is understandable that a large discrepancy arose between the international standard and the actual status of type B pulse oximeters. However, a fundamental issue is that no ideal standard criterion device for “non-invasive” calibration of a pulse oximeter is available until date.
There is another factor that needs to be borne in mind. In Japan, the ISO standard mentioned above was translated directly, to yield JIST80601-2-61:2014. * Then, this part of the JIS was cited directly in the pulse oximeter accreditation criteria set forth pursuant to the Pharmaceutical and Medical Devices Act **. This poses a problem. Originally, each provision of the international or domestic standards serves as “a standard” and cannot be interpreted as indicating “must” (mandatory). However, if such a provision of the international or domestic standards is incorporated directly into the accreditation criteria based on the Pharmaceutical and Medical Devices Act, the provision suddenly changes to assume the meaning “must” (mandatory) 6). We see a possibility that those involved in legislation of the law were not aware of this hidden mechanism.
Thus, the issue has two aspects: (1) the standard available at present does not match up to the current status, and (2) the law has carelessly converted the “standard” into a “mandatory rule.” I fear that although the pulse oximeter has grown in stature to one of the most frequently used medical devices in the world, a lack of sufficient understanding of this device by the regulatory authorities/administrative organs can hamper or markedly delay new development and/or clinical introduction of pulse oximeters for medical use. ***
Relationship to IT and Perspectives for Future
The spread and expansion of the use of pulse oximeters across the world are showing no signs of stopping or slowing down. This can be viewed as an outcome of the efforts made to date by manufacturers around the world towards “commercialization” of their products while adopting new ideas/designs and competing with each other. In other words, “rivalry/free competition” serves as the basis for device improvement, and this should be viewed as a very favorable direction of development. Furthermore, interlinking with the latest IT-related devices in terms of technology and merchandise has also been achieved, resulting in elevation of the value of pulse oximeters as a merchandise.
For example, the new concept of “Blood Oxygen Wellness” was introduced into Japan, in September 2020 to the Apple Watch 6 7). The function and performance of this watch are estimated to be approximately comparable to those of existing pulse oximeters in terms of the technique and principle of measurement. A difference lies in that existing terms such as “pulse oximetry” and “SaO2” are not used in this watch, and this product has been viewed as a product reflecting the manufacturer’s intention of creating the new concept of healthcare or wellness devices derived from the existing concept of medical devices. I have already stated that from a historical point of view, pulse oximeter use is shifting from Type A to Type B devices. Apple Watch 6 should be classified as another new type of device. From an industrial standpoint, this product may be viewed as creating a new field and can serve as a starting point for arguments over the form and nature of devices used for health promotion and management from now on. However, from the legal point of view, no statement has been issued by the regulatory authorities like the Pharmaceutical and Medical Devices Agency (PMDA) in Japan or the approval/accreditation system of Food and Drug Administration (FDA, USA) about approval/accreditation of this type of products. ****
We may expect further sophistication in the software used for pulse oximeters in the future. For example, research is now under way over measurement of the respiratory rate from changes in the pulse wave, and so on. In addition, further efforts will be made to explore the possibility of improving the precision of oxygen saturation measurement by using a multiple wavelength system (a topic to which Dr. Aoyagi dedicated his passion), adding the capability for measuring other parameters, and so on. Contactless sensors for use in pulse oximeters are also under development and their commercialization in the near future is highly probable.
In the end, I would like to add that the heritage left by Dr. Aoyagi (given the shining title of “Father of Pulse Oximeter” 8)) and his guidance on new research and development give us a strong impression that pulse oximeters will continue to evolve in the future.
Comments
☆
In the Japanese industry (particularly in the automobile-related industry) in those days, the world-leading light-emitting diode (LED) technology, characterized by stably low-heat generation, had already become available. Nihon Kohden, however, did not pay close attention to this technology, because its commercialization had not been included in the development goals of the company. Thus, this technology was not adopted in any product made by Nihon Kohden then. At Minolta, on the other hand, commercialization of this technology was included in the development goals and the company succeeded in commercialization of their devices through making full use of the fiberoptic technology, in which also, Japan led the world. However, US venture companies, which place greater emphasis on “clinical applicability” than over precision and theory, were much faster in adopting the Japanese top-notch LED technology into their products. As a result, ironically enough, Japan was left behind by US companies in this field. In hindsight, we may say that the gap between the abilities needed for development and clinical application (presence/absence of the concept of “bio-design”) probably served as a key factor in determining the winner of the competition between Japan and the USA. This is an open issue that we continue to face until date in the field of medical device development.
*
There is an international rule that the ISO standard (international standard) needs to be adopted directly as the JIS standard (domestic standard). This rule is based on the World Trade Organization (WTO)/TBT Agreement (Agreement on Technical Barriers to Trade), which was enforced in 1995, after it was agreed to at the Tokyo Round 1979. Because of this rule, the ISO standard is often translated into the Japanese standard and enforced by the government, without reflecting the views of clinical practitioners, although slight modification of the original standard, tailored to the circumstances in each country, is acceptable under the rule.
**
The formal name of the Pharmaceutical and Medical Device Act is “Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices.” The “Pharmaceutical Affairs Law,” which had long been in use, was renamed in 2014 to make the law’s objectives clearer (securing the quality, efficacy and safety of not only drugs, but also of medical devices and other products). It has been pointed out that because the safety and efficacy of medical devices are affected more by the method of use than by the device itself, unlike in the case of drugs, it is not completely rational to attempt to cover both drugs and medical devices under a single set of laws. Also concerning the procedure for approval/accreditation of products under such a law, there seems to be a tendency towards applying the requirements for precision of medical devices in the same manner as they are for drugs, without any system of involvement of clinicians that actually use those devices, based on the view that both drugs and medical devices are “materials.” Such a practice has led to a discrepancy between the Act and the actual status of use of medical devices in some cases.
***
This may seem irrelevant to clinicians in anesthesia or critical care where assessment of hypoxia is so important. Since there is no theory of pulse oximetry (although the principle is known), actual invasive testing must be used to confirm the accuracy of a device, even though more than 40 years have passed since its invention. The requirement, recently established by ISO to assure precision, is an improvement that should be welcomed from the standpoint of healthcare professionals primarily using Type A (standalone-type for medical use) pulse oximeters.
The new requirement for all pulse oximeter devices to provide proof of calibration prior to application for approval/accreditation is a heavy burden for manufacturers who make the devices and then sell them outside the OR, ICU, or even to lay people. Those are typically Type B pulse oximeters, which are evidently more popular than Type A devices, and manufactured by small- and medium-sized manufacturers. They all want to market their devices as “medical quality products” for better sales, but the testing requirement is contradictory to their claim that their devices are “non-invasive.” Developers feel the need for inexpensive products that by-pass strict calibration requirements. They argue that it is unnecessary to conduct invasive testing prior to accreditation as pulse oximeters have been in use without problem for years. Unfortunately, this idea disregards the need for knowledge of what the values displayed mean.
Lay people started using pulse oximeters to reassure themselves of their good health without learning the concept underlying the product. Most pulse oximeters display what appears to be normal values, as long as one has a good pulse, and this makes people feel secure. If the device happens to show irrelevant values, such as 60%, people instinctively discard those values and most of the time, there is no problem. However, when pulse oximeters are used to make critical judgements (even though the process may not be called diagnosis), such as detecting silent hypoxia in COVID-19, the user’s inability to correctly interpret the values displayed (as in discarding seemingly irrelevant values as mentioned above) cannot be overlooked, as this can be a life-threatening issue.
It is a sobering possibility that the proliferation of non-medical products (assembly kits, Apple Watch, etc.) that, to the lay person, look similar to medical quality pulse oximeters, could result in decreased investment in the development of high-precision pulse oximeters. This would be an unfavorable development from the viewpoint of healthcare for severely sick patients.
It appears that the regulatory authorities are at a loss about what actions should be taken to deal with the sudden increase in the market of medical devices for use in health promotion or for household use. They loosened restrictions compared to the rules for medical use, but there are still no criteria for precise calibration of these devices. The regulatory authorities allow lay persons to read pulse oximeter values, but prohibit anyone other than medical professionals from interpreting the values!
This is like the logic of the merchant of Venice and works only in the world of fiction. While alive, Dr. Aoyagi was very concerned about the possibility that pulse oximeters of differing quality would become available in the market without education being provided to lay persons about the correct interpretation of the displayed oxygen saturation values. New administrative action on this kind of device should include, but not be limited to, the precision of data and method of use, and should also include the method of data assessment for non-medical users.
****
There is a description that this product is estimated to be approximately comparable in function and performance to existing pulse oximeters. This statement needs to be interpreted carefully. The pulse oximeter principally displays a value close to 100% (normal value) in an overwhelming majority of healthy individuals who can walk or run without a problem. Even when no value is displayed or a seemingly abnormal value is displayed occasionally, healthy users can ignore it and can feel reassured that it could be a faulty reading, and no health hazards would be expected in most cases. On the other hand, even when the device displays a value of 100%, there may be cases that still require attention (such as sedated patients receiving oxygen therapy, patients with chronic pulmonary disease, and so on) and cases that may suffer from unfavorable consequences (patients with retinopathy of prematurity, patients receiving immunosuppressants, and so on). These caveats are not commonly known among lay persons.
The judgment by a physician as to whether or not a given person is healthy (i.e., the diagnosis by a physician) involves also the step of interpretation of the displayed value by a pulse oximeter. Lay persons, on the other hand, tend to make self-diagnosis about whether they are healthy or not, without being aware of the difficulty in the interpretation of oxygen saturation values. The package inserts for the product, however, states: “The product should be used under a physician’s instructions,” or “Seek a physician’s advice when the displayed value looks abnormal.” If a pulse oximeter is used by lay persons who have received no education about how to interpret the values, it is only natural to expect that a displayed value 100% is interpreted by them as “full marks,” and that even values of 60% may be ignored unless the person feels short of breath. In practice, such instances have been reported among patients with COVID-19.
Pulse oximeters have been designated as medical devices requiring specific maintenance and control even when designed solely for household use. Depending on the purpose of use, this type of device “requires appropriate maintenance and control because of its potential to affect human life and health.” It is problematic when these devices are used by lay persons who have not received prior education about how to use it. Social education should be provided for correct use of such devices, rather than lifting the restriction as controlled medical devices or devising other easier methods to avoid difficulties. Medical devices are classified into three classes according to the risk to the human body: general medical devices (Class I), controlled medical devices (Class II) and high-level controlled medical devices (Class III). Class III devices have the potential to seriously affect human life and health, and are strictly reviewed before they are granted approval/accreditation, consuming much time and money prior to their being launched in the market much time and money prior to their being launched in the market. The review pertains only to the mechanical safety of the device and the potential for disordered functioning, and does not take into consideration about how it is/should be used or how the displayed values should be interpreted (such a policy for the review of medical devices is a global practiced policy). Extracorporeal membrane oxygenation (ECMO), a class III device), which are used only by pre-trained healthcare professionals, are quite safe, while pulse oximeters (Class II) can be life-threatening if the displayed data are erroneously interpreted, even when the device itself is functioning well.
The regulatory authority gives tacit approval to such inappropriate way of use so long as the device is distributed as a medical device. The device distributor is exempted from responsibility if the package insert includes a statement “not for diagnostic use.” Users have to assume all responsibilities for troubles arising from inappropriate use of such devices. The device marketer uses a sales promotional expression suggesting the nature of the device as being close to a controlled medical device, in an attempt to elevate the product’s value. The only solution to overcome such a cat-and-mouse game is education by healthcare professionals of lay persons who use the devices. During the COVID-19 pandemic, close attention was paid to occult hypoxemia, which probably contributed to a large expansion of the market for Type B pulse oximeters, without provision of education on how these devices should be used to lay users. It is considered important henceforth to evaluate the role actually played by the device in such cases.
Hironami Kubota
Medical devices market research consultant
K and K Japan Corp.
References
- Kubota H: Ch13 Crossing with Pulse Oximetry Development. In 50 years of Developing Vital Sign Monitors (ed) Kubota H) Yakuji Nippo, Tokyo 2016. P179-97 (In Japanese)
- Japanese Patent Gazette. Optical Blood Measurement Device. Japan Patent Office. 1978;181-92 (Showa 49-36229; March 29, 1987) (In Japanese)
- Yamanishi A. Dawn of Pulse Oximetry. In: Ikaikai Gakkai-shi 2005; 75:12 852-62 (In Japanese)
- Severinghaus JW, Y Honda. VII. Pulse oximetry In: History of blood gas analysis. 1987; 3:2 135-8.
- Baker SJ and Shah NK. The Effects of Motion on the Performance of Pulse Oximeters in Volunteers In: Anesthesiology: 1997; 86: 1 101-8
- Kubota H: Problems of approval process of medical devices in Japan, in the case of pulse oximetry. In Digital Health. Nikkei x-Tech (Nikkei.com) April 3, 2019 ( In Japanese)
- Kubota H: Impact of “Blood Oxygen Wellness” by Apple Watch. In Digital Health. Nikkei x-Tech (Nikkei.com) Sept 6, 2020 ( In Japanese)
- Togawa T: Father of the Pulse Oximeter. JSMBE 2011; 49:310-312 (In Japanese)
| Read more articles from this special collection hosted by the APSF on Pulse Oximetry and the Legacy of Dr. Takuo Aoyagi. |

