Disclaimer: Viewers of this material should review the information contained within it with appropriate medical and legal counsel and make their own determinations as to relevance to their particular practice setting and compliance with state and federal laws and regulations. The APSF has used its best efforts to provide accurate information. However, this material is provided only for informational purposes and does not constitute medical or legal advice. This response also should not be construed as representing APSF endorsement or policy (unless otherwise stated), making clinical recommendations, or substituting for the judgment of a physician and consultation with independent legal counsel.
Introduction
Concern over droplet and aerosol transmission of SARS-CoV-2 requires that healthcare workers (HCWs) wear PPE and N95 respirators during endotracheal intubation, open suctioning and nebulization treatments1. Improved methods to better protect HCWs, while allowing comprehensive respiratory care for patients, are needed. To increase HCW protection, a number of physical barriers have been designed and developed, most notably the Aerosol Box,2,3 which can circumscribe a patient’s head and upper torso, acting as a physical barrier for respiratory droplets between HCWs and the patient. However, such devices are only effective against larger, inertial respiratory droplets; Micrometer-to-sub micrometer particles cannot be collected or reliably contained by such means and may remain aerosolized for longer periods of time,4,5 potentially contributing to nosocomial spread. To further advance engineering controls and enable greater treatment options, we have developed a portable, negative pressure Aerosol Hood (Fig. 1A,B), which combines the rigid Aerosol Box structure with active suction through a HEPA filter mounted at the top of the device (akin to a fume hood). The Aerosol Hood utilizes an external blower (AC Infinity Cloudline S6, operating and at >10,000 L min-1, single-phase, 110V power source), a 2’x2’ pleated HEPA filter on its top, and 4 custom-made, two-layer iris ports made of chemically-resistant silicone rubber. The initial prototype weight is ~40 lbs., and two HCWs can efficiently position the device on most transport and ICU beds, over a patient. All surface materials are compatible with isopropyl alcohol and other hospital based antiseptic solutions, and the iris ports are easily replaceable. The unit along with the duct system and blower fit easily on routine carts available in most hospitals and operating rooms. Rigorous tests of particle penetration conducted through the Aerosol Hood, suggest that its implementation would allow for successful HCW protection while providing effective respiratory isolation, without the need for an airborne isolation room, while the patient is receiving NIV, during aerosol-generating procedures and if intubated during any required suctioning or other care.

Figure 1. The Aerosol Hood demonstrating use of a high-flow nasal cannula (A). Rear-view (caudal) image of the Aerosol Hood, showing all iris ports and the HEPA filter (B). Images 30–40 seconds after igniting a smoke candle within the hood; left: no active suction (C); right: active suction at >10,000 L min-1 (D). The active suction directs smoke particles towards the HEPA filter and releases clean air into the room.
Published with author permission.
Methods
Aerosol Hood performance was quantified first by utilizing smoke candles to visualize particle penetration through the device. Further quantification was conducted through nebulizing oleic acid particles (50–400 nm size range) within the aerosol hood. Size distribution measurements of the oleic acid droplets were made using a differential mobility analyzer (1) within the hood, near the aerosol source; (2) near the front (caudal) opening designed for the patient’s upper torso (but outside the hood), and (3) outside the hood near the rear (cranial) iris ports. The iris ports are made of durable silicone rubber and may be replaced easily from routine wear and tear over a period of time yet to be determined.
Results
Smoke candle penetration tests (Fig 1C) reveal clearly that in the absence of suction, sub micrometer particles can exit the hood via the iris ports, hence physical barriers with openings alone are not protective against such small particles. Size distribution results (Fig. 2) are used to determine penetration, the ratio of particles detected at the measurement location to the particles detected at the aerosol source in the hood. Penetration values in the sub micrometer size range decrease with size, from 2 × 10–3 (0.20%) near 70 nm to 2 × 10–4 (0.02%) near 300 nm with maximum flow speed applied. Penetrations increase appreciably in the absence of flow, particularly in the caudal region, where the barrier alone leads to little reduction in concentration. While the study focused primarily on sub micrometer aerosol penetration, we remark that the Aerosol Hood provides a similar or better level of protection from inertial droplets as the original Aerosol Box.
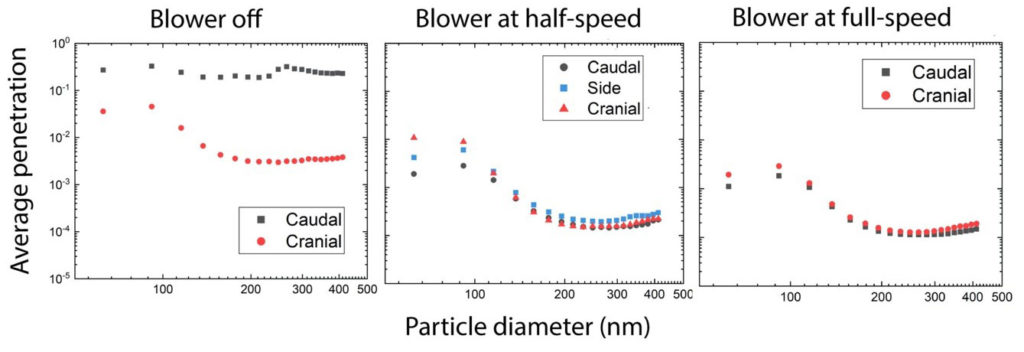
Figure 2. Penetration values calculated for the Aerosol Hood based upon differential mobility analyzer measurements (caudal refers to the chest and abdominal end of the hood and cranial refers to the head-end of the hood)
Discussion

Figure 3. An aerosol hood with limited negative pressure and HEPA filtration.
Published with author permission.
A recent editorial, pertaining to the COVID pandemic, in JAMA by Weissman and associates suggested “Improved engineering controls providing barrier protection against droplet sprays and protection against small airborne particles generated during intubation procedures would be especially valuable”6. The Aerosol Hood can substantially reduce aerosol dispersal (by orders of magnitude) while also providing HCW access. With the Aerosol Hood all modes of NIV with minimal risk of aerosol release may be employed without risk to HCWs. Both, patients and HCWs will benefit from the use of Aerosol Hoods. HEPA filters are much better than N95 respirators. HEPA filters at the very least allow 3 out of 10,000 particles to passing through, while N95 respirators allow 1 out of every 20 particles to pass through. . The particular hood prototype we developed and tested is unique in its use of an external blower capable of providing suction flow rates exceeding 10,000 L min-1, and a high surface area HEPA filter. Both elements are critical to limiting aerosol penetration out of the device. Vacuum systems typically available in hospital rooms lack the pumping capacity needed to achieve such high flows, and the flow rate drawn in by the hood needs to greatly exceed the flow rate of any and all NIV procedures, in order to ensure protection during such procedures. In addition, HEPA filters commonly used in hospital settings are significantly smaller in area, and for the same flow rate the pressure drop across them would be prohibitive, even when using external blower units. Although using a large HEPA filter unit in a medical ward with 6 beds has been described7, its efficiency to successfully provide individual isolation was not possible. About the same time we were designing and testing our prototype unit, the engineers at Bon Secours Health System in Greenville, SC described a system similar to our prototype but with only a small surface area HEPA filter and absence of data to describe its efficiency8. As seen in figure 3, the Greenville box has only a small filter occupying only a small portion of the box. No data about its particle penetration efficacy has been reported to our knowledge.
Another unit, referred to as the helmet, initially described for non-invasive ventilation9 and recommended for care of patients with the COVID respiratory illness10 was customized in Italy during care of their large number of patients with COVID (Figure 4)11. The helmet was adapted with a small HEPA filter in their COVID wards during the pandemic experience in Italy. These helmets although useful, do not provide the necessary active suction to create an adequate negative pressure and also significantly restrict access to patients and are limited in their ability to protect HCWs. Our prototype unit has been placed over healthy human volunteers. There is no discomfort with the blower turned on at full power and it did not interfere during the use of saline nebulization nor during high-flow nasal canula application.
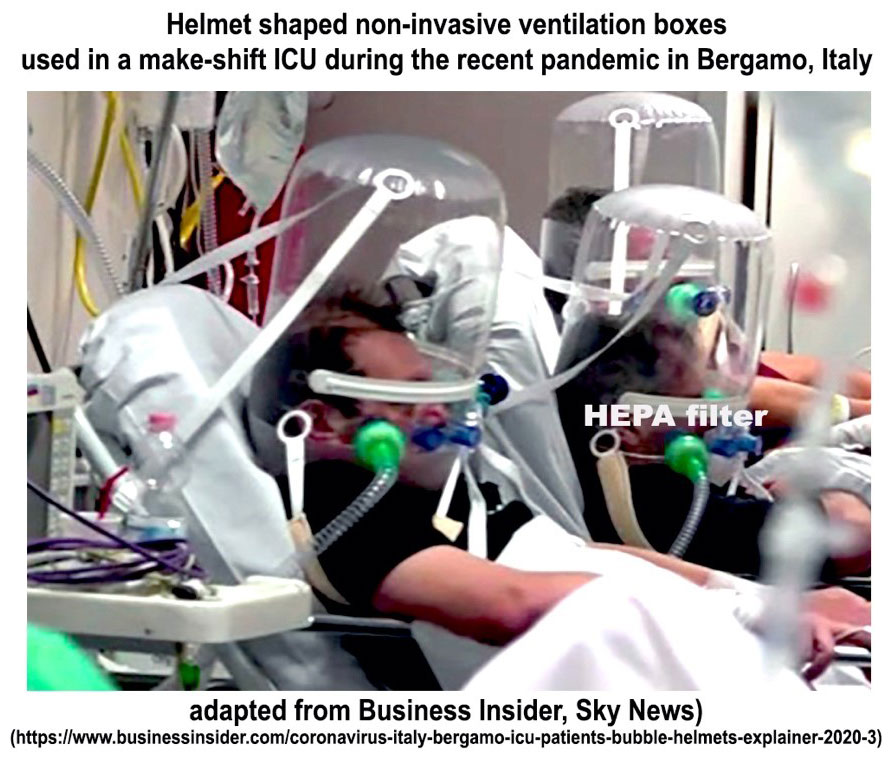
Figure 4. Use of the helmet to provide non-invasive ventilation during COVID respiratory care. The helmet used here was adapted to include a small HEPA filter to clean exhaled air during use.
Published with author permission.
Author Affiliation: Medical School and Mechanical Engineering, University of Minnesota, Minneapolis, MN.
1Corresponding Author: Dr. Kumar Belani, MD, Department of Anesthesiology, 420 Delaware Street SE, MMC 8294, Mayo B-574, Minneapolis, MN 55455. Email: [email protected]; mobile phone: 952-465-6643
References
- POSTON JPB, DAVIS AM. Management of Critically Ill AdultsWith COVID-19. JAMA. 2020;323(18):1839-1841.
- Lai HY. Aerosol Box Protects Healthcare Providers During Endotracheal Intubation. https://sites.google.com/view/aerosolbox/home. Published 2020. Accessed.
- Canelli R, Connor CW, Gonzalez M, Nozari A, Ortega R. Barrier Enclosure during Endotracheal Intubation. New England Journal of Medicine. 2020.
- van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. New England Journal of Medicine. 2020;382(16):1564-1567.
- Liu Y, Ning Z, Chen Y, et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature. 2020.
- Weissman DN, de Perio MA, Radonovich LJ, Jr. COVID-19 and Risks Posed to Personnel During Endotracheal Intubation. JAMA. 2020.
- QIAN HL, Y, SUN, H; NIELSEN, PV; HUANG X; ZHENG, X. . PARTICLE REMOVAL EFFICIENCY OF THE PORTABLE HEPA AIR CLEANER IN A SIMULATED HOSPITAL WARD. BUILD SIMUL. 2010;3:215-224.
- PUTNAM J. the local people & the upstate businesses that you need to know GREENVILLE JOURNAL. 2020(https://greenvillejournal.com/health/bon-secours-st-francis-develops-negative-pressure-intubation ).
- Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP. Effect of Noninvasive Ventilation Delivered by Helmet vs Face Mask on the Rate of Endotracheal Intubation in Patients With Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA. 2016;315(22):2435-2441.
- Innovative Helmet May Help COVID-19 Patients Avoid Intubation. Houston Methodist Leading Medicine. April 14, 2020(https://www.houstonmethodist.org/research/covid19/helmet/).
- Bostock B SH. Critical coronavirus patients in Italy are being treated with bubble-shaped containers over their heads. Here’s what they do. Business Insider.https://www.businessinsider.com/coronavirus-italy-bergamo-icu-patients-bubble-helmets-explainer-2020-3.

 Articles
Articles