Many safety checks are required prior to administering an anesthetic, one of which is to test the integrity and function of the pilot balloon and cuff on the endotracheal tube. In this letter, we review a case in which a manufacturing defect in an endotracheal tube was not detected prior to use.
DEAR RAPID RESPONSE:
A 2-year-old undergoing dental rehab was intubated orally with a Covidien (Mansfield, MA) Shiley Hi-Lo Oral Tracheal 4.0 cuffed ETT. After confirmation of ETT placement, we attempted to adjust the leak to 20 cmH20. Despite over-inflating the pilot balloon, a large leak remained. After confirming with video laryngoscopy that the cuff was below the vocal cords, we concluded that the most likely culprit was a faulty cuff. The decision was made to re-intubate, which was done with a new 4.0 cuffed ETT without complication. Air was added to the cuff until there was a small leak at 20 cmH20.
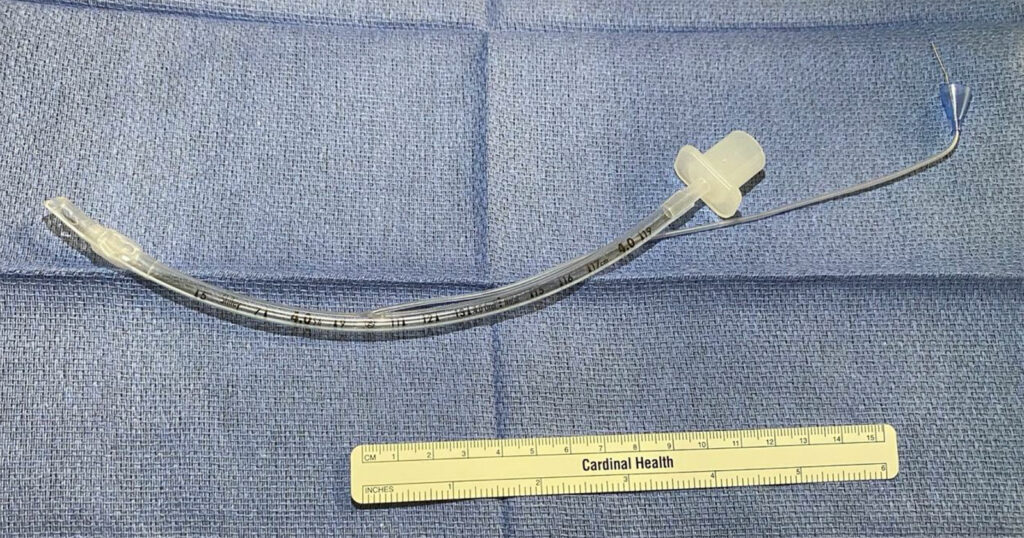
Upon later inspection of the original ETT, we noted that regardless of volume pushed into the pilot balloon, the cuff did not inflate (Figure 1). Unsure about where the disconnect or obstruction was, we incrementally dissected the ETT to see if we could isolate the problem. In order to determine the patency of the inflation tube, we bisected the pilot balloon and passed a wire through the inflation tube, past the cuff, to the tip of the ETT (Figure 2). Since the inflation tube was patent, we then removed the cuff and discovered that the groove which should connect the inflation lumen in the wall of the ETT and the interior space of the ETT cuff was not sufficiently deep as to allow for patency (Figures 3 and 4). We injected methylene blue into the inflation lumen to demonstrate the lack of an opening into the cuff space (Figure 5).

Figure 3 – (a) Left ETT is a normal tube with a groove that measures almost 3mm (Black arrow). (b) Right ETT, which was our problematic tube, is measured at less than 2mm (Red arrow).
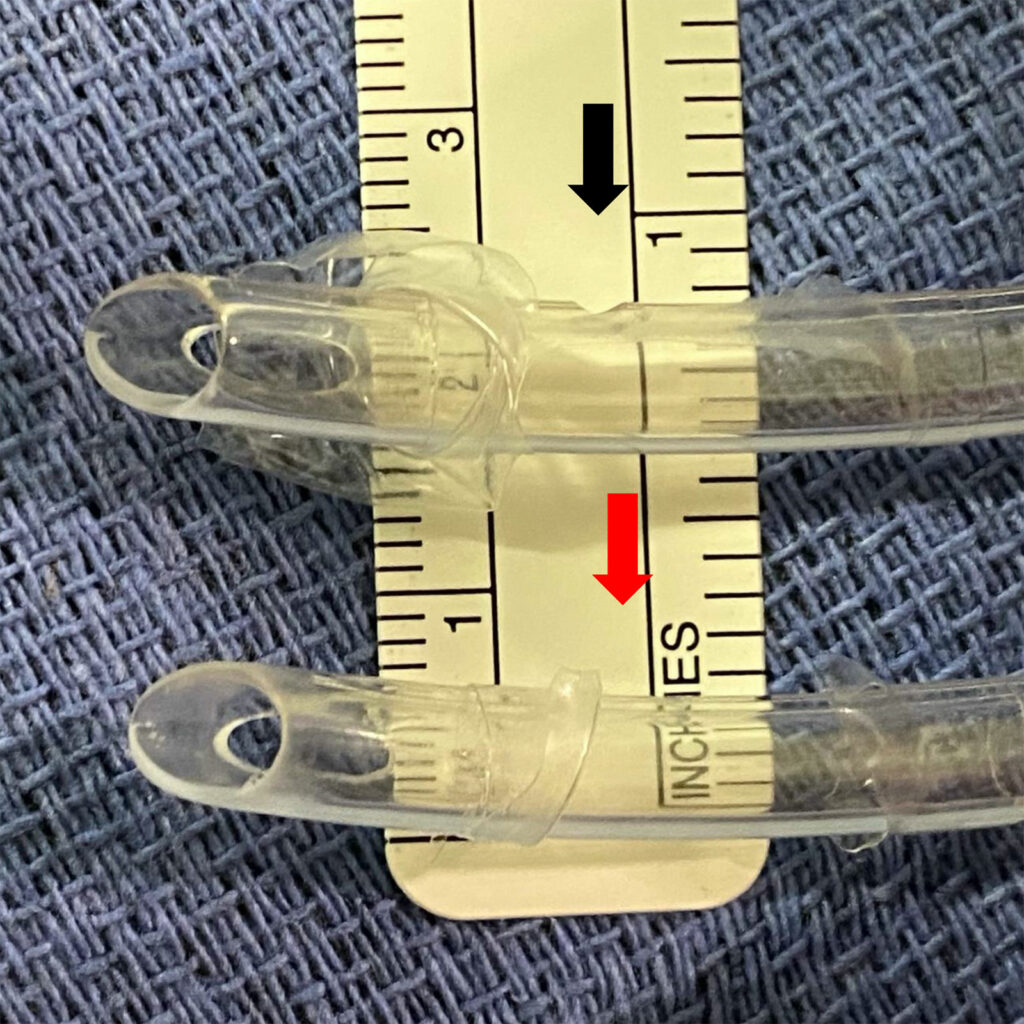
Figure 4 – Side by side comparison of a normal groove (above, black arrow) and that in our ETT (below, red arrow)
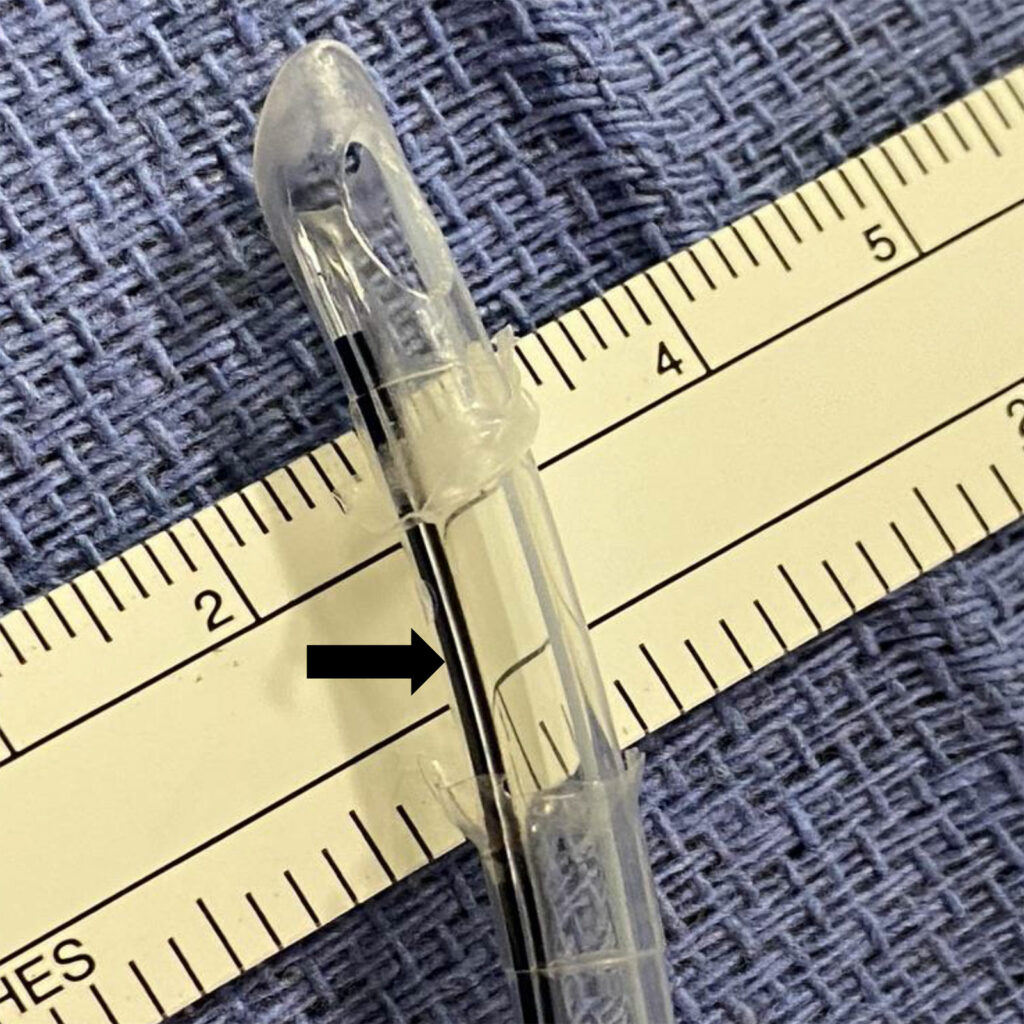
Figure 5 – Methylene blue injected showing no flow from inflation lumen into the now opened cuff space (black arrow)
DISCUSSION
Air leaks are a common part of airway management in anesthesia, but the use of cuffed ETTs has made it easier to secure and protect the trachea without the need for reintubations. Many leaks can be attributed to a mal-positioned ETT, but it is not uncommon for cuffs to be punctured by teeth or equipment like Magill forceps, compromising the integrity of the ETT and ability to ventilate adequately.1 Underinflated cuffs, cephalad head movement, and mis-sized ETTs can also lead to air leaks. Less common are manufacturing defects that result in the ETT not functioning as designed. Although not always the case, these defects can be identified prior to the use of the ETT if an appropriate check of the cuff is performed.2 This entails inflation of the cuff via the pilot balloon, disconnection of the syringe and palpation of the pilot balloon and cuff to assure that they are inflated. After de-briefing, it was recognized that only the pilot balloon had been palpated as part of the initial check, so the lack of a connection between the pilot balloon and cuff was not detected prior to the ETT’s use.
This case highlights the importance of a complete pre-intubation check in order to avoid the inadvertent use of a defective ETT and the possible need for multiple intubations.
Christopher L. Heine, MD, MBA is an Associate Professor in the Department of Anesthesia & Perioperative Medicine at the Medical University of South Carolina
Cory M. Furse, MD, MPH, FAAP is a Professor in the Department of Anesthesia & Perioperative Medicine at the Medical University of South Carolina
The authors have no conflicts of interest.
REFERENCES
- Kearl RA, Hooper RG ; Massive airway leaks: An analysis of the role of endotracheal tubes. Critical Care Medicine. 1993;21(4):518-521
- Gettelman TA, Morris GN; Endotracheal tube failure: Undetected by routine testing. Anesthesia & Analgesia. 1995;81:1313
![]()
Respiratory Interventions Operating Unit
2101 Faraday Ave
Carlsbad, CA 92008
United States
www.medtronic.com
In Response: Pilot Balloon Occlusion
Thank you for the opportunity to respond to the report by Dr. Heine regarding his experience with a Shiley Hi-Lo Orotracheal 4.0 Cuffed Endotracheal Tube (ETT), submitted for publication in the APSF Newsletter.
Background Information: The authors describe a case of a 2-yr old patient intubated with the above-mentioned ETT for dental rehabilitation surgery. After confirmation of ETT placement it was noted that a large leak persisted despite ‘over-inflation of the pilot balloon’. The patient was re-intubated and the original, removed ETT was examined; dissection of the tube and cuff revealed a lack of patency between the pilot line and the ETT cuff.
Reported Incidents: Our PMV team have confirmed that over the past 5 years Medtronic sold roughly 28.2 million Shiley™ endotracheal tubes. The rate for any inflation system complaints is 3.4 complaints per million ETTs sold during that time period.
Root Cause Discussion: This report arose after the author demonstrated a lack of patency between the pilot line and the cuff. In this case, it appears that an error occurred within the manufacturing process, which also survived the rigid quality inspections to which all our products are subjected. We can confirm that, as with all manufacturing-related issues, review and improvements have been instituted at the manufacturing site to capture any defective devices which leave the production line.
This event highlights the importance of adhering to the pre-insertion cuff inflation procedure, as highlighted in the Instructions for Use of the device.
The Medtronic Mission guides us to strive without reserve for the greatest possible reliability and quality in our products. In order to achieve that outcome, we rely heavily on physicians such as Dr. Heine, and organizations such as the APSF, to ensure that we remain true to this statement. We are grateful for the efforts and thorough investigation of the defective ETT by Dr. Heine, and to the APSF for continuing to partner with us as we strive for the best in patient safety and device quality.
Sincerely,
Dr. Karen A Phillips, MD, FCA, MBA, CPPS
Chief Medical Officer, Respiratory Interventions
Medtronic