APSF Technology Education Initiative (TEI) : Low-Flow Anesthesia
Technology is integral to the practice of Low-Flow Anesthesia. The most astute practitioner, well-schooled in the scientific foundations of uptake and distribution as well as the intricacies of the circle system, cannot anesthetize a single patient without an anesthesia machine at their side. Dr. Ralph Waters, who pioneered the introduction of carbon dioxide absorbents and low-flow anesthesia, is to be admired for his skillful low-flow and closed-circuit anesthesia practice despite having very limited equipment capabilities by today’s standards.1 Lowe and Ernst described its theoretical foundations more than 40 years ago, including a recognition of the environmental benefits.2
In modern anesthesia practice, we are fortunate to have access to a variety of anesthesia delivery systems that support the practice of low-flow anesthesia. All of these systems provide the features that are essential to the practice of low-flow anesthesia namely, a circle system and CO2 absorbent. Even with these essential tools, effective low-flow anesthesia practice requires vigilance and engagement to minimize fresh gas flow while avoiding the potential complications of inadequate anesthetic and oxygen concentrations. Manufacturers have developed a variety of different tools to help guide low-flow anesthesia practice. Since these tools require different underlying technology, they are not available on all anesthesia delivery systems, even from the same manufacturer.
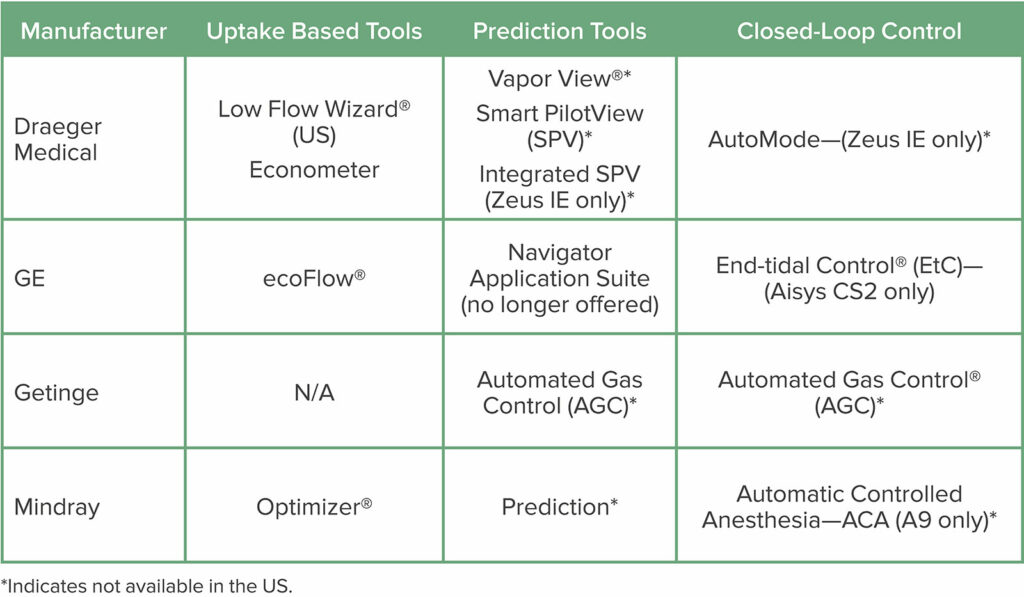
The available features fall broadly into four categories—1) Measurement of inspired and expired oxygen and anesthetic concentrations, 2) Oxygen uptake measurement to guide a minimum flow recommendation, 3) Prediction tools indicating future trends of oxygen and anesthetic concentrations and 4) Closed-loop control of oxygen and anesthetic concentration. The Table lists the tools from various manufacturers that fall into each of these categories. This article will review the functionality of the four categories of tools and highlight their advantages and disadvantages.
OXYGEN AND ANESTHETIC CONCENTRATION MEASUREMENT
Dr. Waters’ feats of low-flow anesthesia practice notwithstanding, a safe and effective practice in modern times requires continuous oxygen and anesthetic concentration monitoring. As fresh gas flow is reduced, the concentrations of oxygen and agent delivered by the anesthesia machine will differ from the concentrations inspired by the patient from the inspiratory limb of the breathing system. This is entirely due to mixing of exhaled gases with the delivered gases. The magnitude of the impact on the inspired concentrations will depend upon the degree of rebreathing and the rate of uptake by the patient which will in turn depend upon the phase of the anesthetic. There are actually three different concentrations of oxygen and anesthetic in a circle system to consider when practicing low-flow anesthesia (See Figure 1):
Delivered concentrations (FD): The concentration in the fresh gas. Oxygen concentration is determined by the oxygen and air or nitrous oxide flow settings, agent concentration is determined by the vaporizer setting.
Inspired concentrations (FI): The concentration in the inspiratory limb of the circuit. Depending upon the degree of rebreathing and the phase of the anesthetic, inspired concentrations will be different from the concentrations delivered in the fresh gas. Set values on the vaporizer and flowmeters will not equal what the patient inspires during low-flow anesthesia.
Expired concentrations (FE): The concentration in the expiratory limb of the circuit. For induction and maintenance, FE will generally be less than FI due to uptake.
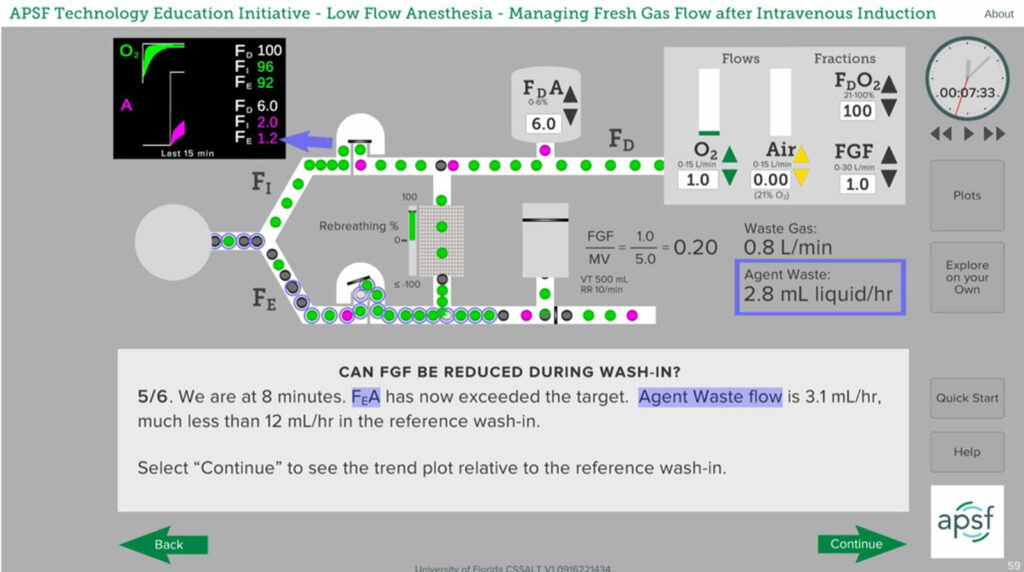
Figure 1: Sample low-flow anesthesia scenario following pre-oxygenation and IV induction. Note the location in the circuit for the three concentrations of oxygen and anesthetic—FD, FI, FE—and the trend values in the minitrend. A one MAC concentration of isoflurane (1.2%) is achieved in the exhaled gas while the vaporizer is set to 6%. Progressive reduction in the vaporizer setting is required to prevent anesthetic overdosage. To explore these concepts in more detail, the APSF course on low-flow anesthesia can be found at www.apsf.org/tei.
A safe and effective low-flow practice requires that the anesthesia professional increase the delivered concentrations of oxygen and anesthetic, in some cases well above the desired inspired values, and make adjustments as the rate of uptake changes. For example, during induction when uptake of anesthetic is high, the vaporizer setting will need to be well above the desired MAC value if FGF is reduced to minimize waste and pollution. (See Figure 1 above) Having a real-time measurement of the oxygen and anesthetic concentrations in the inspired and expired gases is essential to a safe and effective practice.
Fortunately, all modern anesthesia machines are required to be equipped with an inspired oxygen monitor. Analysis of inspired and expired oxygen and anesthetic concentrations is sometimes a feature of the anesthesia machine and sometimes an external device. Unfortunately, anesthetic concentration monitoring is not required by standards for machines and therefore may be lacking when it is not a built-in feature. The ability to monitor both inspired and expired concentrations of oxygen AND volatile anesthetic is HIGHLY RECOMMENDED, if not essential, to the safe practice of low-flow anesthesia.
OXYGEN UPTAKE MEASUREMENT
Tools based upon oxygen uptake measurement are the simplest to implement and are designed to indicate to the clinician a safe target for reducing fresh gas flow. These tools require a monitor of inspired and expired oxygen concentration and a flow sensor to measure tidal volume and respiratory rate to derive minute ventilation. The difference between inspired and expired oxygen concentrations multiplied by the minute ventilation is an estimate of the patient’s oxygen consumption. This estimate, adjusted for any other losses from the circuit, is used to make a minimum flow recommendation to which the actual flow setting is compared. Additional losses include gas sampled for analysis by a sidestream gas analyzer if the sampled gases are not returned to the circuit, as well as any leaks. Specific algorithms for the uptake based tools may differ between manufacturers but the basic method is the same. All of these tools are approved by the FDA and available in the US.
Tools that fall into this category can be useful as a guide to reducing fresh gas flow, but have very significant limitations since the concentrations of anesthetic and oxygen are not incorporated into all of these tools. Further, there is no accompanying guidance for the vaporizer or flowmeter setting required to achieve a desired clinical endpoint. Inadequate oxygen and anesthetic delivery is possible when using these tools.
During induction, when anesthetic uptake is high, uptake tools will recommend a FGF setting that will, in general, not be sufficient to ensure adequate anesthetic depth, even if the vaporizer is set well above the desired MAC value. Oxygen requirements may not be satisfied since the provider can use any gas in the fresh gas flow to satisfy the minimum flow recommendation when as much as 100% oxygen in the delivered gas may be needed to maintain an adequate inspired oxygen concentration. Furthermore, if concentrations in the circuit need to change quickly, fresh gas flow will need to be increased well above the minimum recommendation.
In general, these tools can be useful during maintenance to guide reducing fresh gas flow when anesthetic and other gas concentrations are relatively stable and do not need to change quickly. They are not particularly useful for guiding FGF during induction, and do not obviate the need for meticulous attention to monitored concentrations of oxygen and anesthetic when setting the fresh gas mixture and the vaporizer.
PREDICTION TOOLS
Tools that predict future oxygen and volatile anesthetic concentrations are a useful type of decision support. They allow the user to set fresh gas flow rates and the vaporizer by seeing, in real time, the future effects of the chosen settings. Such systems allow more accurate and rapid adjustment of oxygen and agent delivery and also provide an important safety device in that the display of future concentrations gives an early, visual warning if O2 and anesthetic concentrations are heading out of the desired range.
At a basic level these tools are similar to the various tools that predict concentrations of iv agents using well described drug models such as StanPump®.3 Modeling inhalational agents is more complex than for intravenous medications, especially during low-flow anesthesia, since the interactions of total fresh gas flow and vaporizer settings, rebreathing and patient uptake all must be considered. However, in contrast to predictive models of iv agents, the end-tidal concentration of volatile anesthetics is measured and used to refine the predictions in realtime.
There is significant clinical experience documenting the utility of prediction tools. Kennedy and French developed and evaluated a prototype prediction system that was used for some years in their hospital with the Datex ADU. This system made it easy to set a desired fresh gas flow and then, by observing the predictions, adjust the vaporizer dial to achieve the desired end-tidal (and effect-site) vapor concentrations.4-5 Similar tools are now commercially available on anesthesia delivery systems from several manufacturers although they are not available in the US. (See Table below).
Table 1: Tools to Support Low-Flow Anesthesia Listed by Manufacturer
Tools are only available on certain models from each manufacturer. Contact the manufacturer for information on which models support the tools and the technology options required for each.
While prediction tools have been proven to be both useful and reliable, they have limitations. Continuous information about total fresh gas flow and mixture as well as agent delivery is required to make accurate predictions. As a result, these systems will typically require an anesthesia machine with electronic flowmeters or electronic measurement of flow settings. In addition, an electronically controlled vaporizer, or one that is modified to electronically record the dial setting is mandatory. Furthermore, while all of these systems reliably predict future anesthetic agent concentrations, they do not all predict oxygen concentrations.
While useful to show the anesthesia professional the implications of the current FGF settings for future anesthetic and oxygen concentrations, prediction tools still require ongoing vigilance and interaction. They do not automate the anesthesia delivery process in any way.
CLOSED LOOP CONTROL
These tools are the most sophisticated, and designed to reduce the burden on the provider by automating oxygen and anesthetic delivery while minimizing fresh gas flow. Once the anesthesia professional selects the target concentrations for in- or expired oxygen and end-expired anesthetic agent, the algorithms can continuously seek to minimize waste while simultaneously ensuring the target concentrations are attained in a timely and accurate manner.
While these algorithms differ among manufacturers, all maintain selected targets within a narrow range, reduce waste to closed circuit or near closed circuit conditions and help to reduce the provider workload.6-9
At the same time, the risk of inadequate oxygen or anesthetic delivery is managed automatically. The clinician no longer has to continually adjust FGF and vaporizer settings, freeing up time to devote to other tasks, especially during the busy induction period. Additional features help the clinician to titrate down agent delivery in anticipation of emergence while maintaining low-flow conditions.
While tools for closed-loop control of oxygen and anesthetic delivery are clinically proven to be reliable, they are completely dependent upon a sophisticated, and costly, anesthesia workstation. Without continuous real-time measurement of inspired and expired oxygen and agent concentrations and computer control of flowmeters and vaporizers, closed-loop control is not possible. All of these machines have failure modes that return to manual control if any of the sensors fail. Only one closed-loop control solution is available in the US.
CONCLUSIONS
The technique of low-flow inhaled anesthetic delivery became possible almost 100 years ago with the introduction of carbon dioxide absorption. For quite some time, all anesthesia workstations have been designed to use a circle breathing system with CO2 absorbent. However, the technique of low-flow anesthesia has yet to be universally adopted. Admittedly, the cognitive burden of continuously monitoring oxygen and anesthetic concentrations as fresh gas flow is reduced, along with the mental models required to understand the complex real time interactions of rebreathing and uptake, remain barriers to adoption. Fortunately, tools for helping the clinician are available and understanding and adopting these tools will make it easier to pursue a low-flow anesthesia practice.10 Hopefully this primer is helpful for understanding the benefits and limitations of these tools.
Many anesthesia workstations configured with electronic vaporizers will indicate the anesthetic agent usage. This information is very useful as you embark upon the practice of low-flow anesthesia. The ultimate goal of the low-flow practice is to learn how to achieve the same clinical results while using less anesthetic agent. If the anesthetic agent usage is available, it becomes a convenient metric for measuring the impact of reducing fresh gas flow.
Technology notwithstanding, understanding the principles of low-flow anesthesia and the importance of continuous oxygen and anesthetic monitoring remains essential. For more information the APSF has developed a MOCA Safety CME accredited course on low-flow anesthesia which is available at www.apsf.org/tei.
REFERENCES
- Waters RM. Carbon dioxide absorption from anaesthetic atmospheres: (Section of Anaesthetics). Proc R Soc Med. 1936;30:11–22. PMID: 19990907.
- Lowe HJ, Ernst EA. The quantitative practice of anesthesia use of closed circuit. Williams & Wilkins, Baltimore, 1981.
- Shafer S. stanpumpR: Pharmacokinetic simulation of anethestic drugs. https://steveshafer.shinyapps.io/stanpumpr. Accessed October 12, 2022.
- Kennedy RR, French RA. The development of a system to guide volatile anaesthetic administration. Anaesth Intensive Care. 2011;39:182–190. PMID: 21485665.
- Kennedy RR, French RA, Gilles S. The effect of a model-based predictive display on the control of end-tidal sevoflurane concentrations during low-flow anesthesia. Anesth Analg. 2004;99:1159–1163. PMID: 1538536.
- De Cooman S, Hendrickx JF, Peyton PJ, Demeere JL, De Wolf AM. Agent consumption with the Zeus® in the automated closed-circuit anesthesia mode with O2/air mixtures. BMC Res Notes. 2014 ;Jul 23;7:469. doi: 10.1186/1756-0500-7-469. PMID: 25056252.
- Carette R, De Wolf AM, Hendrickx JF. Automated gas control with the Maquet FLOW-i. J Clin Monit Comput. 2016;30:341–346. PMID: 26072157.
- De Cang M, Hendrickx JFA, De Wolf AM. Sevoflurane usage with different O2/air fresh gas flows during end-expired target control administration with the Aisys. Presented at the ESA 2014 meeting, Stockholm, Sweden ESA 2014, 3AP1–7.
- Lucangelo U, Garufi G, Marras E et al. End-tidal versus manually-controlled low-flow anaesthesia. J Clin Monit Comput. 2014;28:117–121. PMID: 24114079.
- Jan F A Hendrickx, Andre M De Wolf. The Anesthesia Workstation: Quo Vadis? Anesth Analg. 2018;Sep;127:671–675. doi: 10.1213/ANE.0000000000002688. PMID:29239956.

 Articles
Articles  PDF
PDF