| This article was previously published on the APSF online portal. The present version is updated and modified by the author for the present APSF Newsletter. |
Dear RAPID Response:
The global crisis due to COVID-19 has permeated every aspect of our health care systems. Concerns about the biohazard of SARS-CoV-2, spread and contact transmission to patients, health care personnel, environment, and equipment have been widespread, especially with regards to procedures that generate aerosols (AGPs).1-3 Transmission of the virus is primarily respiratory in nature. SARS-CoV-2 virion is approximately 120 nanometers in diameter (0.06–0.14 µm), and travels from person to person in biological carrier particles such as droplets or aerosols.2,3 Recommendations regarding adequate levels of PPE, handwashing, surface cleaning, decontamination, and precautions during airway management procedures have been discussed extensively during the pandemic.4-6 As with other respiratory transmissible diseases, we rely on two important filtering systems: circuit filters when artificial breathing systems are used in the operating room and/or intensive care units (ICU) and face-mask respirators.
However, things are a bit complicated:
- Anesthesia machines and mechanical ventilators require filters for air quality purification and cross-contamination prevention. The efficiency standard of such filters is termed HEPA for high-efficiency particulate air/ high-efficiency particulate absorbing capacity.7 The ASA recommends placement of HEPA filters between the Y-piece of the breathing circuit and the patient’s mask, endotracheal tube or laryngeal mask airway.8
- European and U.S standards to determine filter efficiency are not the same: European standards use 99.95% removal of particles with a diameter of 0.3 µm in diameter, while the U.S uses 99.97%.9
- Face mask efficiency is determined by the level of particle penetration. An N95 mask for example removes at least 95% of 300 nm particles using an airflow rate of 85 liters/min.10 Face mask respirators are regulated according to U.S National Institute for Occupational Safety and Health (NIOSH) and internationally recognized standards and testing.
- Filters in breathing circuits and anesthesia machines are not regulated. There is no national or international standard test for filters in breathing circuits. Since there is no standard testing, are all manufactures reporting the same when discussing the level of efficiency?11
- Are current available filters adequate for COVID-19?
- Because many COVID-19 patients require prolonged mechanical ventilation, how often should these filters be changed in the ICU?
- What should health care professionals do in case of filter shortages?
These are some of the pressing questions with regards to HEPA filters that I would like to see discussed.
Thank you.
Felipe Urdaneta
Clinical Professor of Anesthesiology
University of Florida/NFSGVHS
Gainesville, Florida
The author is a consultant for Medtronic and member of the Advisory Board for Vyaire and has received speaker honoraria on their behalf.
References
- Canova V, Lederer Schlpfer H, Piso RJ, et al. Transmission risk of SARS-CoV-2 to healthcare workers—observational results of a primary care hospital contact tracing. Swiss Medical Weekly. 2020;150:1–5.
- Asadi S, Bouvier N, Wexler AS, et al. The coronavirus pandemic and aerosols: Does COVID-19 transmit via expiratory particles? Aerosol Sci Technol. 2020;0:1–4.
- van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med. 2020;382:1564–1567.
- Asenjo JF. Safer intubation and extubation of patients with COVID-19. Can J Anaesth. 2020:1–3.
- Chia SE, Koh D, Fones C, et al. Appropriate use of personal protective equipment among healthcare workers in public sector hospitals and primary healthcare polyclinics during the SARS outbreak in Singapore. Occup Environ Med. 2005;62:473–477.
- Sorbello M, El-Boghdadly K, Di Giacinto I, et al. The Italian coronavirus disease 2019 outbreak: recommendations from clinical practice. Anaesthesia. 2020;75:724–732.
- First MW. Hepa filters. Appl Biosaf. 1998;3:33–42.
- American Society of Anesthesiologists. Information for health care professionals. Published 2020. https://www.asahq.org/about-asa/governance-and-committees/asa-committees/committee-on-occupational-health/coronavirus. Accessed June, 2020.
- Wikipedia. HEPA. https://en.wikipedia.org/wiki/HEPA. Accessed June 9, 2020.
- Haghighat F, Bahloul A, Lara J, et al. Development of a procedure to measure the effectiveness of N95 respirator filters against nanoparticles. 2012.
- NIOSH-Approved Particulate Filtering Facepiece Respirators. https://www.cdc.gov/niosh/npptl/topics/respirators/disp_part/default.html. Accessed June 9, 2020.
Reply:
We thank Felipe Urdaneta, MD, for highlighting some confusion about the use of anesthesia breathing system filters in response to the COVID-19 pandemic. The APSF website has a page (https://www.apsf.org/faq-on-anesthesia-machine-use-protection-and-decontamination-during-the-covid-19-pandemic/) that summarizes current strategies for protecting the anesthesia machine from contamination by a potentially infected patient. But it does not provide some of the details behind the recommendations. This article will provide details, such as the risk of patient trans-infection via the breathing system, modes of virus transmission, filtration physics, types of filters, and standardized tests and specifications of filters, in an effort to answer Felipe Urdaneta’s questions and clear up similar confusions among our readership.
Risk of Patient Cross-infection via the Breathing System
Circle breathing systems present a hypothetical risk of patient cross-infection due to rebreathing of previously exhaled gases. Prior to the 1990s, anesthesia breathing system filters were not routinely used and it was thought that cross-infection of patients was prevented by passage of exhaled gas through the alkaline carbon dioxide absorbent.1 However, breathing system filters became increasingly used in the 1990s2 after a report of nine cases of cross-infection by hepatitis C attributed to contaminated anesthesia breathing systems.3 There is conflicting evidence for the potential for cross-infection; almost no cases have been documented, but in-vitro tests demonstrate the possibility.4-6 In any case, breathing system filters are recommended by a number of anesthesia societies, but only when breathing circuits are reused between patients.7,8
Modes of Respiratory Virus Transmission
COVID-19 (SARS-CoV-2) is transmitted primarily via the respiratory route, as are Severe Acute Respiratory Syndrome (SARS-CoV), Middle-East Respiratory Syndrome-associated coronavirus (MERS-CoV), and other coronaviruses. It is transmitted via droplets, which are more than 20 microns in diameter, and via aerosols which are less than 5–10 microns in diameter.9 Droplets tend to fall due to gravity, whereas aerosol particles float in air and follow airstreams. Intermediate-sized particles share some properties of droplets and aerosols. Rapid evaporation of small droplets results in even smaller droplet nuclei that also follow airstreams. Droplets, aerosols, and intermediate-sized particles are generated during coughing, sneezing, and talking, whereas aerosols are primarily generated during passive breathing. An important concept in filtering pathogens is that respiratory viruses are not transmitted by isolated virus particles floating in air, but by viruses contained within larger particles. Droplets and some intermediate-sized particles can settle on surfaces, potentially leading to surface transmission.
No studies have estimated how many virus particles SARS-CoV-2 infected patients exhale. However, one study that quantified exhalation of other respiratory virus particles, found that seasonal coronavirus infected patients exhaled and coughed-out 0 to 200,000 virus particles per hour.10
While a single viral particle can theoretically result in systemic infection, the chance of infection increases with the duration and magnitude of viral exposure.11
Physics of Filtration
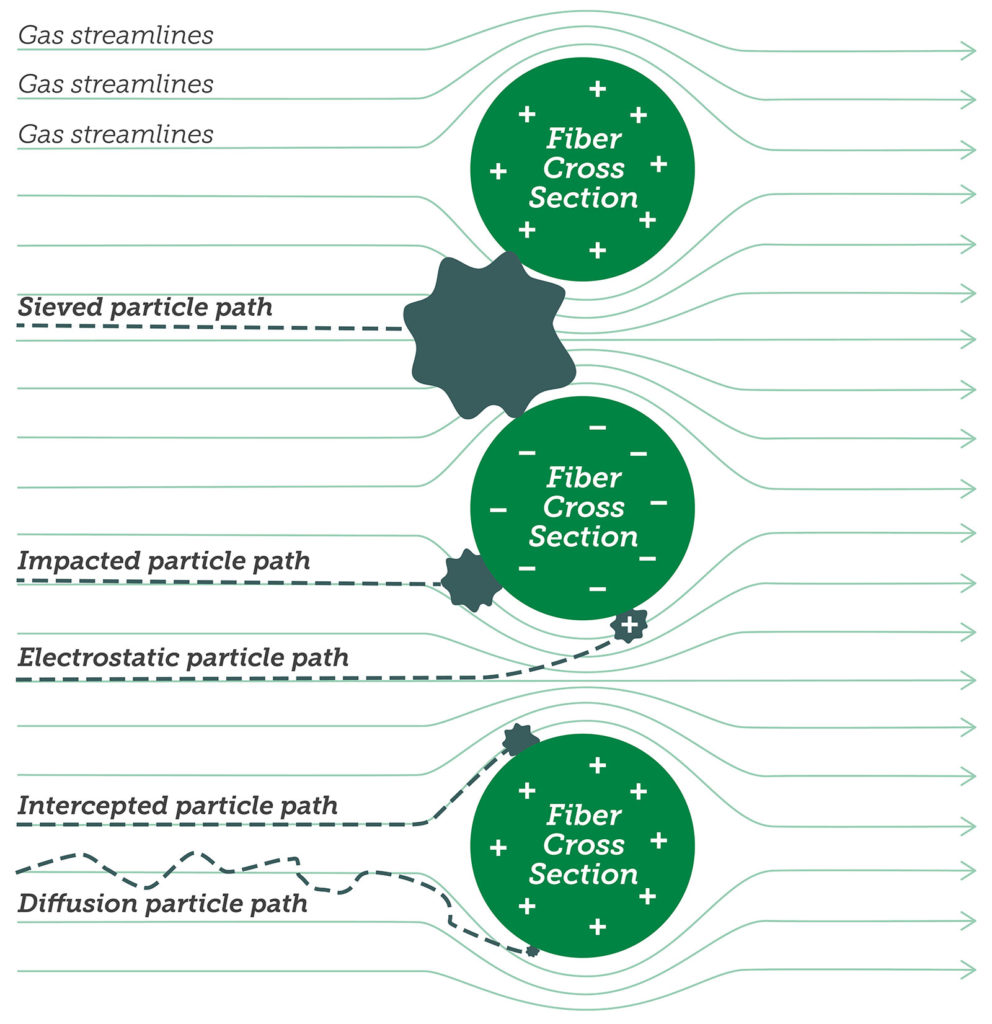
People generally understand the physics of sieve filtration, an observable phenomenon in strainers, whereby a particle that is larger than the smallest holes cannot pass through a filter. However, other forces come into play with very small particles (e.g., diameter < 2 microns).12 Very small particles tend to adhere to the filter material once they make contact, even if they could fit through openings in the filter. There are four basic mechanisms whereby particles make contact with filter material. Particles in the range of 0.11 micron can directly impact filter strands through a process called inertial impaction. Particles in the range of 0.05–1 micron can tangentially contact filter strands through a process called interception. As particles get smaller, they increasingly exhibit Brownian motion in addition to moving with air flow—and can contact filter material as a consequence of this erratic movement through a process called diffusion. Finally, small charged particles can be attracted to the charged surface of filter material through a process called electrostatic attraction. Figure 1 illustrates each of these phenomenon, and Figure 2 shows how the sum of these phenomena affects the overall filter efficiency. Note that for most air filters, particles around 0.3 microns (i.e., 300 nanometers) in size are the most difficult to trap—particles that are larger or smaller are easier to catch.
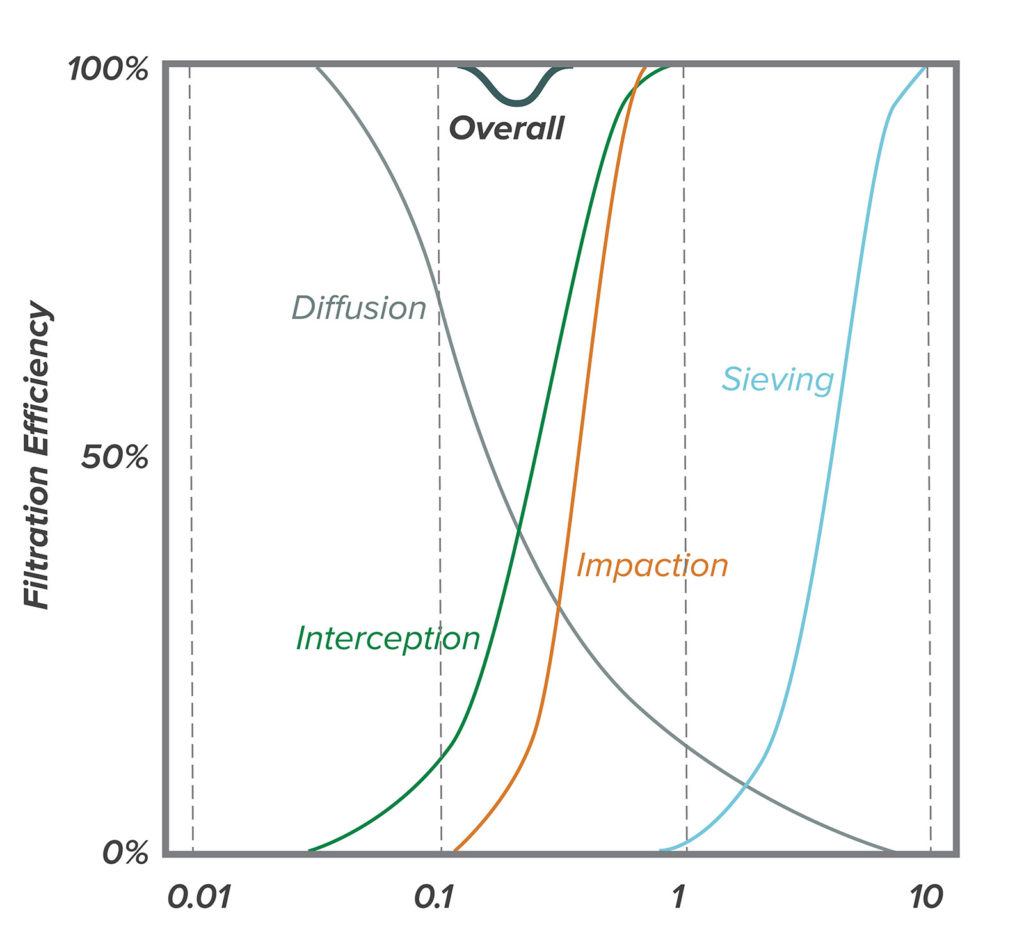
Figure 2: Individual filtration phenomena sum to yield overall filtration for different size particles. Note that the lowest efficiency is around 0.3 microns.
Types of Filters Used in Anesthesia Breathing Systems
Pleated mechanical filters
Pleated mechanical filters contain a thick sheet of tightly packed, randomly oriented, bonded hydrophobic fibers that capture particulates within the depth of the filter. The filter material is pleated to increase the surface area and decrease resistance to airflow. They typically have a very high filtration efficiency and may also provide some heat and moisture exchange when placed close to the airway, at a site of two-way airflow. When used in a humid environment, their filtration efficiency and resistance to airflow can get better or worse; they still tend to be highly effective when damp.13 Liquids do not easily pass through pleated mechanical filters.14 Mechanical filters tend to cost more, and have a higher internal volume than electrostatic filters.
Electrostatic filters
Electrostatic filters contain a thin sheet of less tightly woven electrostatic fibers. Their resistance to airflow is less for a given surface area, so they are not pleated. Electrostatic filters typically have 1000-fold lower filtration efficiencies than pleated mechanical filters.13 Their filtration efficiency and resistance to airflow can get better or worse in a humid environment. Liquids easily pass through an electrostatic filter.14
Heat and moisture exchange filters (HMEF)
By themselves, heat and moisture exchange (HME) devices provide no filtering. HMEs that contain an electrostatic or a pleated mechanical filter are denoted HMEF. HMEs and HMEFs are only effective for humidification when placed close to the airway, at a site of two-way airflow, where they absorb water during exhalation and release it during inhalation.15
Membrane filters
A completely different type of filter is used in respiratory gas analyzers to prevent fluid entry into the analyzer chamber. While not classified as breathing system filters, hydrophobic membrane filters are commonly included in water traps because they allow gas to pass when dry, but become occlusive when wet. Membrane filters have ultra-small pores and channels that can prevent particle passage primarily by sieving.
Standardized Tests and Specifications of Filters
Particle Filtration
There is one single international standard for testing the filtration efficiency of breathing system filters, ISO 23328-1: Breathing system filters for anaesthetic and respiratory use.16 The standard describes a method, the salt test method, that quantifies the number of 0.1 to 0.3 micron airborne sodium chloride particles that pass through the filter during a short-term challenge at airflow rates likely to be encountered during the intended use. Pediatric and adult filters are challenged with either 0.1 mg or 0.2 mg of sodium chloride particles at 15 L/min or 30 L/min, respectively. Filters are preconditioned in humidified air to simulate a period of clinical use before they are tested. Nonelectrostatically charged dry salt particles are used for the challenge, because they are very difficult to trap. The method does not assess the filtration performance for droplets and aerosols, nor does it proport to test the filtration performance for microorganisms. It is for comparison purposes only and has no proven clinical relevance. The standard contains no threshold for minimum performance of breathing system filter efficiency. The test results are expressed as the percent filtration efficiency, which is the percent of particles in the challenge that do not pass through the filter. For example, if the filter is challenged with 10 million (107) particles, and 1000 (103) particles are detected on the other side, then the percent filtration efficiency is 100 * (1 – 103/ 107) = 99.99%.
Entirely different standards are used for testing and rating other types of filters. Notably, the National Institute for Occupational Health and Safety, developed NIOSH 42 CFR Part 84: Respiratory Protective Devices17 as a method to test and rate nonpowered air-purifying respirators. N-series respirators that are used in health care are challenged with 200 mg of nonelectrostatically charged dry sodium chloride particles that are 0.1 to 0.3 microns in size at a flow rate of 85 L/min.18 This is a similar but more challenging test than ISO 23328-1 due to the higher particle mass and flow rate. Another notable filtration testing standard is IEST-RP-CC001: HEPA and ULPA Filters,19 which tests the performance of filters used in clean air devices and clean rooms. HEPA refers to high-efficiency particle air filters that remove 99.97% of particles whose diameter is equal to 0.3 microns. However, it is not appropriate to apply this term to breathing system filters because the test methods are different.
Microorganism Filtration
Some breathing system filter product literature contains statements about bacterial and or viral filtration efficiency. There is no standard test for determining the bacterial and or viral filtration efficiency of breathing system filters, but there are standard methods for determining this for other types of filters. One of these is ASTM F2101–19: Standard test method for evaluating the bacterial filtration efficiency (BFE) of medical face mask materials, using a biological aerosol of Staphylococcus aureus.20 A similar procedure using biologic aerosols of Bacillus subtilis or MS-2 coliphage to test breathing system filters is described by Wilkes et al.21 and is the same as that in Draft BS EN 13328-1 (which never got past the draft stage). In both procedures, suspensions of bacteria or viruses are aerosolized to a mean liquid particle size of 3.0 microns and drawn through the filter material by a downstream vacuum. Anything that passes through the filter is captured either into nutrient broth or onto culture plates. Percent filtration efficiency is calculated by dividing the number of cultured particles downstream of the filter by the number in the upstream challenge. At face value, these methods might seem more clinically relevant than the salt test method. They use larger sized fluid particles than the salt test method. The fluid particles may be electrostatically charged. Only viable microorganisms are counted. However, these methods are less reproduceable. In general, the same filter will have greater percent filtration efficiencies for bacteria, than for viruses, than for salt particles.
Bubble Point Testing
Membrane filters are rated by pore size, which is indirectly determined using the bubble point test. The bubble point test is based on the principle that liquid is held in the pores of the filter by surface tension and capillary forces, and that the minimum pressure required to force liquid out of the pores is related to the pore diameter. However, pore size cannot be used as a surrogate for particle or pathogen filtration efficiency. Hydrophilic 0.22-micron membrane filters are commonly used to sterilize pharmaceuticals, and to maintain sterility of epidural infusions, but their efficiency at filtering airborne particles has not been tested. Some 0.2-micron hydrophobic membrane filters (e.g., those in the GE D-Fend Pro, Dräger WaterLock® 2, and Covidien FilterLine® water traps) have been independently tested, and have an airborne viral filtration efficiency of 99.99% or greater.
Clinical Recommendations
In 2003, the United States Center for Disease Control stated, “No recommendation can be made for placing a bacterial filter in the breathing system or patient circuit of anesthesia equipment,” citing now 40-year old studies that showed failure of sterile breathing circuits or breathing system filters to reduce the incidence of postoperative pneumonia.22 There is no current regulation to use breathing system filters on anesthesia machines. However, it seems prudent to prevent, as best as possible, the cross-infection of patients with SARS-CoV-2 during this COVID-19 pandemic. There are sparse reports of cross-infection from contaminated anesthesia machines prior to SARS, MERS, and COVID-19, but the risk from these pathogens is not currently known. Out of an abundance of caution and informed by existing knowledge, the APSF and ASA recommend using breathing system filters, recognizing that the science is incomplete. (https://www.apsf.org/faq-on-anesthesia-machine-use-protection-and-decontamination-during-the-covid-19-pandemic/).
Adding breathing system filters is not without risk.23,24 Depending on placement, they can add dead space which increases carbon dioxide rebreathing and slows inhalation induction and emergence. They increase resistance to inspiratory and/or expiratory flow, which increases spontaneous work of breathing, and affects pulmonary mechanics (testing methods are described in international standard ISO 9360-1).25 Filters can become obstructed leading to life-threatening hypoventilation and barotrauma. They add weight to the breathing circuit and add sites for accidental disconnection.
The filtration efficiency required to prevent infection from exhaled viruses via the breathing system is not known. If a patient exhales 200,000 virus particles per hour, then an electrostatic filter that traps 99.9% of them will allow only 200 to pass. Placing two of these filters in series (e.g., one at the airway and one on the expiratory limb) will multiply the filtration efficiency to 99.9999%, making the risk of virus passage almost nil, but will double the resistance to flow. Using a single higher efficiency (e.g., 99.9999%) pleated mechanical breathing system filter at the airway will capture the same number of viruses and introduce less airway resistance than two electrostatic filters in series, but may increase the dead space.
Clinicians should know the specifications for the breathing system filters available to them. These can be found from the manufacturer’s web site or help line, in product literature, online, and in journal articles.13,14 Important specifications are:
- bacterial and viral filtration efficiency (%—higher is better),
- NaCl or salt filtration efficiency (%—higher is better),
- resistance to flow (pressure drop in Pa or cmH2O at a given airflow rate in L/min—lower is better),
- how the former specifications are affected by filter conditioning in humidity,
- internal volume (ml—lower is better), and
- humidification
- (moisture loss in mgH2O/L of air—lower is better), or
- (moisture output in mgH2O/L of air—higher is better).
Note that some publications list evaluations that were done 10 or 20 years ago, and that products may change, or be manufactured or distributed by different companies.
Robert G. Loeb, MD, is clinical professor of anesthesiology at the University of Florida College of Medicine, Gainesville, FL.
He is employee of the University of Florida, chair of the American Society of Anesthesiologists Committee on Equipment and Facilities, and he is on the Masimo, Inc. Technical Advisory Board.
References
- Murphy PM, Fitzgeorge RB, Barrett RF. Viability and distribution of bacteria after passage through a circle anaesthetic system. Br J Anaesth. 1991;66:300–304.
- Atkinson MC, Girgis Y, Broome IJ. Extent and practicalities of filter use in anaesthetic breathing circuits and attitudes towards their use: a postal survey of UK hospitals. Anaesthesia. 1999;54:37–41.
- Chant K, Kociuba K, Munro R, et al. Investigation of possible patient-to-patient transmission of hepatitis C in a hospital. New South Wales Public Health Bulletin. 1994;5:47–51.
- Spertini V, Borsoi L, Berger J, et al. Bacterial contamination of anesthesia machines’ internal breathing-circuit-systems. GMS Hyg Infect Control. 2011;6(1).
- Lloyd G, Howells J, Liddle C, et al. Barriers to hepatitis C transmission within breathing systems: efficacy of a pleated hydrophobic filter. Anaesth Intensive Care. 1997;25:235–238.
- Heinsen A, Bendtsen F, Fomsgaard A. A phylogenetic analysis elucidating a case of patient-to-patient transmission of hepatitis C virus during surgery. J Hosp Infect. 2000;46: 309–313.
- Australian & New Zealand College of Anaesthetists. PS28 Guideline on infection control in anaesthesia 2015. Available at: https://www.anzca.edu.au/safety-advocacy/standards-of-practice/policies,-statements,-and-guidelines Accessed July 6, 2020.
- Association of Anaesthetists of Great Britain & Ireland. Guidelines: infection prevention and control 2020. https://anaesthetists.org/Portals/0/PDFs/Guidelines%20PDFs/Infection_Control_Guideline_FINAL%202020.pdf?ver=2020-01-20-105932-143 Accessed July 6, 2020.
- Tellier R, Li Y, Cowling BJ, Tang JW. Recognition of aerosol transmission of infectious agents: a commentary. BMC Infect Dis. 2019;19:101.
- Leung NH, Chu DK, Shiu EY, et al. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med. 2020;26:676–680.
- Nicas M, Hubbard AE, Jones RM, Reingold AL. The infectious dose of variola (smallpox) virus. Appl Biosaf. 2004;9:118–127.
- Hakobyan NA. Introduction to basics of submicron aerosol particles filtration theory via ultrafine fiber media. Armen J Phys. 2015;8:140–151.
- Wilkes A. Breathing system filters: an assessment of 104 breathing system filters. MHRA Evaluation 04005. March 2004 https://www.psnetwork.org/wp-content/uploads/2018/01/An-assessment-of-104-breathing-system-filters-MHRA-Evaluation-04005-2004-.pdf Accessed July 6, 2020.
- Wilkes AR. The ability of breathing system filters to prevent liquid contamination of breathing systems: a laboratory study* APPARATUS. Anaesthesia. 2002;57:33–39.
- Wilkes AR. Heat and moisture exchangers and breathing system filters: their use in anaesthesia and intensive care. Part 1–history, principles and efficiency. Anaesthesia. 2011;66:31–9.
- International Organization for Standardization. Breathing system filters for anaesthetic and respiratory use — Part 1: Salt test method to assess filtration performance. (ISO 23328-1:2003) https://www.iso.org/standard/35330.html Accessed July 6, 2020.
- Department of Health and Human Services. 42 CFR Part 84 Respiratory protective devices; final rules and notice. Federal Register Volume 60, Number 110 (Thursday, June 8, 1995). https://www.govinfo.gov/content/pkg/FR-1995-06-08/html/95-13287.htm Accessed July 6, 2020.
- National Institute for Occupational Safety and Health. Determination of particulate filter efficiency level for N95 series filters against solid particulates for non-powered, air-purifying respirators standard testing procedure (STP). https://www.cdc.gov/niosh/npptl/stps/pdfs/TEB-APR-STP-0059-508.pdf Accessed July 6, 2020.
- Institute for Environmental Sciences and Technology. HEPA and ULPA Filters. (IEST-RP-CC001) https://www.iest.org/Standards-RPs/Recommended-Practices/IEST-RP-CC001 Accessed July 6, 2020.
- ASTM International. Standard test method for evaluating the bacterial filtration efficiency (BFE) of medical face mask materials, using a biological aerosol of Staphylococcus aureus. (F2101 – 19) https://compass.astm.org/EDIT/html_annot.cgi?F2101+19 Accessed July 6, 2020.
- Wilkes AR, Benbough JE, Speight SE, et al. The bacterial and viral filtration performance of breathing system filters. Anaesthesia. 2000;55:458–465.
- CDC Healthcare Infection Control Practices Advisory Committee. Guidelines for preventing health-care–associated pneumonia, 2003. MMWR 53(RR03); 1–36, 2004.
- Lawes EG. Hidden hazards and dangers associated with the use of HME/filters in breathing circuits. Their effect on toxic metabolite production, pulse oximetry and airway resistance. Br J Anaesth. 2003;91:249–264.
- Wilkes AR. Heat and moisture exchangers and breathing system filters: their use in anaesthesia and intensive care. Part 2–practical use, including problems, and their use with paediatric patients. Anaesthesia. 2011;66:40–51.
- International Standards Organization. Anaesthetic and respiratory equipment—heat and moisture exchangers (HMEs) for humidifying respired gases in humans—part 1: HMEs for use with tracheostomized patients having minimal tidal volume of 250 ml (ISO 9360-2: 2001).
The information provided is for safety-related educational purposes only, and does not constitute medical or legal advice. Individual or group responses are only commentary, provided for purposes of education or discussion, and are neither statements of advice nor the opinions of APSF. It is not the intention of APSF to provide specific medical or legal advice or to endorse any specific views or recommendations in response to the inquiries posted. In no event shall APSF be responsible or liable, directly or indirectly, for any damage or loss caused or alleged to be caused by or in connection with the reliance on any such information.


 Issue PDF
Issue PDF